Abstract
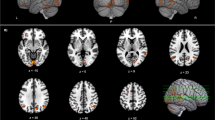
Ample evidence has suggested that individuals with subjective memory complaints are at a higher risk for cognitive decline. Nevertheless, the significance of subjective memory complaints in Parkinson’s disease has not been studied until now. We investigated whether the patterns of cognitive profiles and gray matter density differed in cognitively normal patients with Parkinson’s disease based on the presence of subjective memory complaints. Using a single question with a yes or no answer, cognitively normal patients with Parkinson’s disease were classified as with (n = 20) or without subjective memory complaints (n = 15). Cognitive profiles and gray matter density were examined using standardized neuropsychological tests and voxel-based morphometry. No significant differences in demographic characteristics were observed between groups. The detailed neuropsychological tests demonstrated that Parkinson’s disease patients with subjective memory complaints had significantly decreased verbal fluency and slightly lower scores on the backward digit-span test compared with those without subjective memory complaints. A voxel-based morphometry analysis revealed that Parkinson’s disease patients with subjective memory complaints had significantly decreased gray matter density in the anterior cingulate gyrus and right inferior parietal lobule compared with those without subjective memory complaints. Our data demonstrated that Parkinson’s disease patients with subjective memory complaints showed a poorer performance on tasks related to verbal fluency and attention with more severe cortical atrophy compared to those without subjective memory complaints, suggesting that subjective memory complaints in patients with Parkinson’s disease may represent an early manifestation of underlying Parkinson’s disease-related pathological changes.

Similar content being viewed by others
References
Reid LM, Maclullich AM (2006) Subjective memory complaints and cognitive impairment in older people. Dement Geriatr Cogn Disord 22:471–485
Striepens N, Scheef L, Wind A, Popp J, Spottke A, Cooper-Mahkorn D, Suliman H, Wagner M, Schild HH, Jessen F (2010) Volume loss of the medial temporal lobe structures in subjective memory impairment. Dement Geriatr Cogn Disord 29:75–81
Rodda J, Dannhauser T, Cutinha DJ, Shergill SS, Walker Z (2010) Subjective cognitive impairment: functional MRI during a divided attention task. Eur Psychiatry 26:457–462
Barnes LL, Schneider JA, Boyle PA, Bienias JL, Bennett DA (2006) Memory complaints are related to Alzheimer disease pathology in older persons. Neurology 67:1581–1585
Emre M (2003) Dementia associated with Parkinson’s disease. Lancet Neurol 2:229–237
Biggins CA, Boyd JL, Harrop FM, Madeley P, Mindham RH, Randall JI, Spokes EG (1992) A controlled, longitudinal study of dementia in Parkinson’s disease. J Neurol Neurosurg Psychiatry 55:566–571
de Lau LM, Schipper CM, Hofman A, Koudstaal PJ, Breteler MM (2005) Prognosis of Parkinson disease: risk of dementia and mortality: the Rotterdam Study. Arch Neurol 62:1265–1269
Hobson P, Meara J (2004) Risk and incidence of dementia in a cohort of older subjects with Parkinson’s disease in the United Kingdom. Mov Disord 19:1043–1049
Levy G, Tang MX, Louis ED, Cote LJ, Alfaro B, Mejia H, Stern Y, Marder K (2002) The association of incident dementia with mortality in PD. Neurology 59:1708–1713
Caviness JN, Driver-Dunckley E, Connor DJ, Sabbagh MN, Hentz JG, Noble B, Evidente VG, Shill HA, Adler CH (2007) Defining mild cognitive impairment in Parkinson’s disease. Mov Disord 22:1272–1277
Janvin CC, Larsen JP, Aarsland D, Hugdahl K (2006) Subtypes of mild cognitive impairment in Parkinson’s disease: progression to dementia. Mov Disord 21:1343–1349
Aarsland D, Bronnick K, Williams-Gray C, Weintraub D, Marder K, Kulisevsky J, Burn D, Barone P, Pagonabarraga J, Allcock L, Santangelo G, Foltynie T, Janvin C, Larsen JP, Barker RA, Emre M (2010) Mild cognitive impairment in Parkinson disease: a multicenter pooled analysis. Neurology 75:1062–1069
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 55:181–184
Kang Y, Na DL (2003) Seoul Neuropsychological Screening Battery. Human Brain Research and Consulting Co, Incheon
Lee JE, Park HJ, Park B, Song SK, Sohn YH, Lee JD, Lee PH (2010) A comparative analysis of cognitive profiles and white-matter alterations using voxel-based diffusion tensor imaging between patients with Parkinson’s disease dementia and dementia with Lewy bodies. J Neurol Neurosurg Psychiatry 81:320–326
Lee JE, Park HJ, Song SK, Sohn YH, Lee JD, Lee PH (2010) Neuroanatomic basis of amnestic MCI differs in patients with and without Parkinson disease. Neurology 75:2009–2016
Cho H, Yang DW, Shon YM, Kim BS, Kim YI, Choi YB, Lee KS, Shim YS, Yoon B, Kim W, Ahn KJ (2008) Abnormal integrity of corticocortical tracts in mild cognitive impairment: a diffusion tensor imaging study. J Korean Med Sci 23:477–483
Yue NC, Arnold AM, Longstreth WT Jr, Elster AD, Jungreis CA, O’Leary DH, Poirier VC, Bryan RN (1997) Sulcal, ventricular, and white matter changes at MR imaging in the aging brain: data from the cardiovascular health study. Radiology 202:33–39
Beck AT, Ward CH, Mendelson M, Mock J, Erbaugh J (1961) An inventory for measuring depression. Arch Gen Psychiatry 4:561–571
Sollinger AB, Goldstein FC, Lah JJ, Levey AI, Factor SA (2010) Mild cognitive impairment in Parkinson’s disease: subtypes and motor characteristics. Parkinsonism Relat Disord 16:177–180
Innis RB, Cunningham VJ, Delforge J, Fujita M, Gjedde A, Gunn RN, Holden J, Houle S, Huang SC, Ichise M, Iida H, Ito H, Kimura Y, Koeppe RA, Knudsen GM, Knuuti J, Lammertsma AA, Laruelle M, Logan J, Maguire RP, Mintun MA, Morris ED, Parsey R, Price JC, Slifstein M, Sossi V, Suhara T, Votaw JR, Wong DF, Carson RE (2007) Consensus nomenclature for in vivo imaging of reversibly binding radioligands. J Cereb Blood Flow Metab 27:1533–1539
Levy G, Jacobs DM, Tang MX, Cote LJ, Louis ED, Alfaro B, Mejia H, Stern Y, Marder K (2002) Memory and executive function impairment predict dementia in Parkinson’s disease. Mov Disord 17:1221–1226
Jacobs DM, Marder K, Cote LJ, Sano M, Stern Y, Mayeux R (1995) Neuropsychological characteristics of preclinical dementia in Parkinson’s disease. Neurology 45:1691–1696
Dubois B, Burn D, Goetz C, Aarsland D, Brown RG, Broe GA, Dickson D, Duyckaerts C, Cummings J, Gauthier S, Korczyn A, Lees A, Levy R, Litvan I, Mizuno Y, McKeith IG, Olanow CW, Poewe W, Sampaio C, Tolosa E, Emre M (2007) Diagnostic procedures for Parkinson’s disease dementia: recommendations from the movement disorder society task force. Mov Disord 22:2314–2324
Williams-Gray CH, Evans JR, Goris A, Foltynie T, Ban M, Robbins TW, Brayne C, Kolachana BS, Weinberger DR, Sawcer SJ, Barker RA (2009) The distinct cognitive syndromes of Parkinson’s disease: 5 year follow-up of the CamPaIGN cohort. Brain 132:2958–2969
Kehagia AA, Barker RA, Robbins TW (2010) Neuropsychological and clinical heterogeneity of cognitive impairment and dementia in patients with Parkinson’s disease. Lancet Neurol 9:1200–1213
Rinne JO, Portin R, Ruottinen H, Nurmi E, Bergman J, Haaparanta M, Solin O (2000) Cognitive impairment and the brain dopaminergic system in Parkinson disease: [18F]fluorodopa positron emission tomographic study. Arch Neurol 57:470–475
Sawamoto N, Piccini P, Hotton G, Pavese N, Thielemans K, Brooks DJ (2008) Cognitive deficits and striato-frontal dopamine release in Parkinson’s disease. Brain 131:1294–1302
Jokinen P, Bruck A, Aalto S, Forsback S, Parkkola R, Rinne JO (2009) Impaired cognitive performance in Parkinson’s disease is related to caudate dopaminergic hypofunction and hippocampal atrophy. Parkinsonism Relat Disord 15:88–93
Abrahams S, Goldstein LH, Simmons A, Brammer MJ, Williams SC, Giampietro VP, Andrew CM, Leigh PN (2003) Functional magnetic resonance imaging of verbal fluency and confrontation naming using compressed image acquisition to permit overt responses. Hum Brain Mapp 20:29–40
Pihlajamaki M, Tanila H, Hanninen T, Kononen M, Laakso M, Partanen K, Soininen H, Aronen HJ (2000) Verbal fluency activates the left medial temporal lobe: a functional magnetic resonance imaging study. Ann Neurol 47:470–476
Pereira JB, Junque C, Marti MJ, Ramirez-Ruiz B, Bartres-Faz D, Tolosa E (2009) Structural brain correlates of verbal fluency in Parkinson’s disease. Neuroreport 20:741–744
Phelps EA, Hyder F, Blamire AM, Shulman RG (1997) FMRI of the prefrontal cortex during overt verbal fluency. Neuroreport 8:561–565
Raichle ME, Fiez JA, Videen TO, MacLeod AM, Pardo JV, Fox PT, Petersen SE (1994) Practice-related changes in human brain functional anatomy during nonmotor learning. Cereb Cortex 4:8–26
Pardo JV, Pardo PJ, Janer KW, Raichle ME (1990) The anterior cingulate cortex mediates processing selection in the Stroop attentional conflict paradigm. Proc Natl Acad Sci USA 87:256–259
Corbetta M, Shulman GL (2002) Control of goal-directed and stimulus-driven attention in the brain. Nat Rev Neurosci 3:201–215
Dujardin K, Duhamel A, Delliaux M, Thomas-Anterion C, Destee A, Defebvre L (2010) Cognitive complaints in Parkinson’s disease: its relationship with objective cognitive decline. J Neurol 257:79–84
Petrova M, Raycheva M, Zhelev Y, Traykov L (2010) Executive functions deficit in Parkinson’s disease with amnestic mild cognitive impairment. Am J Alzheimers Dis Other Demen 25:455–460
Mattos P, Lino V, Rizo L, Alfano A, Araujo C, Raggio R (2003) Memory complaints and test performance in healthy elderly persons. Arq Neuropsiquiatr 61:920–924
Acknowledgments
This study was supported by a grant of the Korea Healthcare Technology R&D Project, Ministry for Health, Welfare and Family Affairs, Republic of Korea (A091159).
Conflicts of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hong, J.Y., Lee, J.E., Sohn, Y.H. et al. Neurocognitive and atrophic patterns in Parkinson’s disease based on subjective memory complaints. J Neurol 259, 1706–1712 (2012). https://doi.org/10.1007/s00415-011-6404-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-011-6404-3