Abstract
Objectives
Apomorphine is a potent dopamine agonist useful in the treatment of Parkinson’s disease patients with disabling motor fluctuations and ‘off’ periods, not responding to oral medication. It can also be of benefit in reducing dyskinesia by providing more constant dopaminergic stimulation and permitting lower levodopa dosage. However, there is a paucity of information on long-term benefits of apomorphine, including no large-scale phase III trial. We have examined our experience of apomorphine over the last 10 years, to assess indications, pattern of use, efficacy, and side effect profile.
Methods
All patients requiring apomorphine were identified through the Parkinson’s disease Nurse Specialist’s records. An audit form was produced so that the same information was gathered from all case-notes.
Results
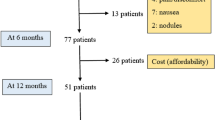
There were 107 patients (61 males and 46 females). Mean age of disease onset was 50.9 years, SD±9.3 (range 29–78). The mean duration of disease at start of apomorphine treatment was 10 years (SD±4.8, range2–29). The most common indications for apomorphine were severe unpredictable ‘off’ periods (75.7 %), motor fluctuations (18.7 %) and dyskinesia (5.6 %). Most patients (63.6 %) used both intermittent subcutaneous injections and infusion via pump; 25.2% were on intermittent injection, and 11.2 % infusion alone. Mean dose per injection was 3.7mg. Mean infusion dose 69.8mg, running over a mean of 13.5 hours. The mean duration of intermittent apomorphine use was 48.2 months. The mean duration of infusion was 25.1 months. Complications included skin problems in 16 patients, 2 had symptomatic hypotension, 2 worsening confusion, 1 new confusion and 5 new hallucinations (after sometime on apomorphine). Sixteen patients have stopped using apomorphine completely. Thirteen have stopped the pump, but continue on intermittent injections.
Conclusion
Subcutaneous apomorphine is easy for patients to use, is well tolerated and has a low incidence of side effects, especially confusion.
Similar content being viewed by others
References
Anden NE, Rubenson A, Fuxe K, et al. (1967) Evidence for dopamine receptor stimulation by apomorphine. J Pharmac Pharmacol 19:627–629
Cantello R, Gilli M, Riccio A, Bergamasco B (1986) Mood changes associated with “end of dose deterioration” in Parkinson’s disease: a controlled study. J Neurol Neurosurg Psychiatry 49:1182–1190
Colzi A, Turner K, Lees AJ (1998) Continuous subcutaneous waking day apomorphine in the long-term treatment of levodopa induced interdose dyskinesias in Parkinson’s disease. J Neurol Neurosurg Psychiatry 64:573–576
Corsini GU, Del Zompo M, Gessa GL, Mangoni A (1979) Therapeutic efficacy of apomorphine combined with an extracerebral inhibitor of dopamine receptors in Parkinson’s disease. Lancet 1:954–956
Cotzias GC, Papavasiliou PS, Tolosa ES, Mendez JS, Bell-Midura M (1976) Treatment of Parkinson’s disease with aporphines. Possible role of growth hormone. N Engl J Med 294(11):567–572
Dewey Jr RB, Hutton JT, LeWitt PA, Factor SA (2001) A randomized double-blind, placebo controlled trial of subcutaneously injected apomorphine for Parkinsonian off states. Arch Neurol 58:1385–1392
Ernst AM (1967) Mode of action of apomorphine and dexamphetamine in gnawing compulsion in rats. Psychopharmacologia 10(4):316–323
Frankel JP, Lees AJ, Kempster PA, Stern GM (1990) Subcutaneous apomorphine in the treatment of Parkinson’s disease. J Neurol Neurosurg Psychiatry 53(2):96–101
Hardie RJ, Lees AJ, Stern GM (1984) On-Off fluctuations in Parkinson’s disease. A clinical and neuropharmacological study. Brain 107:487–506
Kumar R, Lozano A, Kim YJ, et al. (1998) Double-blind evaluation of subthalamic nucleus deep brain stimulation in advanced Parkinson’s disease. Neurology 51:850–855
Manson AJ, Turner K, Lees AJ (2002) Apomorphine monotherapy in the treatment of refractory motor complications of Parkinson’s disease: long-term follow-up study of 64 patients. Mov Disord 17:1235–1241
Marsden CD, Parkes JD (1976) “On-Off” effects in patients with Parkinson’s disease on chronic levodopa therapy Lancet 1:292–296
Marsden CD, Parkes JD (1977) Success and problems of long-term levodopa therapy in Parkinson’s disease. Lancet i:345–349
Quinn NP, Koller WC, Lang AE, Marsden CD (1986) Painful Parkinson’s disease. Lancet 1:1366–1369
Schwab RS, Amador LV, Lettvin JY (1951) Apomorphine in Parkinson’s disease. Transf Am Neurol Assoc 76:251–253
Shaw KM, Lees AJ, Stern GM (1980) The impact of treatment with levodopa on Parkinson’s disease. QJM 49:283–293
Steiger MJ, Quinn NP, Marsden CD (1992) The clinical use of apomorphine in Parkinson’s disease. J Neurol 239:389–393
Stibe CM, Lees AJ, Kempster PA, Stern GM (1988) Subcutaneous apomorphine in parkinsonian on-off oscillations. Lancet 1:403–406
Stocchi F, Vacca L, De Pandis MF, Barbato L, Valente M, Ruggieri S (2001) Subcutaneous continuous apomorphine infusion in fluctuating patients with Parkinson’s disease: long-term results. Neurol Sci 22:93–94
Varma TR, Fox SH, Eldridge PR, et al. (2003) Deep brain stimulation of the subthalamic nucleus: effectiveness in advanced Parkinson’s disease patients previously reliant on apomorphine. J Neurol Neurosurg Psychiatry 74:170–174
Weil E (1884) De l’apomorphine dans certain troubles nerveux. Lyon Med 48:411–419
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tyne, H.L., Parsons, J., Sinnott, A. et al. A 10 year retrospective audit of long-term apomorphine use in Parkinson’s disease. J Neurol 251, 1370–1374 (2004). https://doi.org/10.1007/s00415-004-0547-4
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00415-004-0547-4