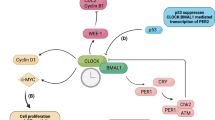
Abstract
For life forms to survive, they must adapt to their environmental conditions. One such factor that impacts on both prokaryotic and eukaryotic organisms is the light–dark cycle, a consequence of planetary rotation in relation to our sun. In mammals, the daily light cycle has affected the regulation of many cellular processes such as sleep–wake and calorific intake activities, hormone secretion, blood pressure and immune system responses. Such rhythmic behaviour is the consequence of circadian rhythm/biological clock (BC) systems which are controlled in a light stimulus-dependent manner by a master clock called the suprachiasmatic nucleus (SCN) situated within the anterior hypothalamus. Peripheral clocks located in other organs such as the liver and kidneys relay signals from the SCN, which ultimately leads to tightly controlled expression of several protein families that in turn act on a broad range of cellular functions. Work in lower organisms has demonstrated a link between aging processes and BC factors, and studies in both animal models and clinical trials have postulated a role for certain BC-associated proteins in tumourigenesis and cancer progression. Recent exciting data reported within the last year or so have now established a molecular link between specific BC proteins and factors that control the mammalian cell cycle and DNA damage checkpoints. This mini review will focus on these discoveries and emphasise how such BC proteins may be involved, through their interplay with cell cycle/DNA damage response pathways, in the development of human disease such as cancer.


Similar content being viewed by others
References
Ahmed S, Alpi A, Hengartner MO, Gartner A (2001) C. elegans RAD-5/CLK-2 defines a new DNA damage checkpoint protein. Curr Biol 11:1934–1944
Andreassen PR, D’Andrea AD, Taniguchi T (2004) ATR couples FANCD2 monoubiquitination to the DNA-damage response. Genes Dev 18:1958–1963
Antoch MP, Kondratov RV, Takahashi JS (2005) Circadian clock genes as modulators of sensitivity to genotoxic stress. Cell Cycle 4:901–907
Barnes JW, Tischkau SA, Barnes JA, Mitchell JW, Burgoon PW, Hickok JR, Gillette MU (2003) Requirement of mammalian Timeless for circadian rhythmicity. Science 302:439–442
Bartek J, Lukas C, Lukas J (2004) Checking on DNA damage in S phase. Nat Rev Mol Cell Biol 5:792–804
Bekker-Jensen S, Lukas C, Kitagawa R, Melander F, Kastan MB, Bartek J, Lukas J (2006) Spatial organization of the mammalian genome surveillance machinery in response to DNA strand breaks. J Cell Biol 173:195–206
Benard C, McCright B, Zhang Y, Felkai S, Lakowski B, Hekimi S (2001) The C. elegans maternal-effect gene clk-2 is essential for embryonic development, encodes a protein homologous to yeast Tel2p and affects telomere length. Development 128:4045–4055
Brown EJ, Baltimore D (2003) Essential and dispensable roles of ATR in cell cycle arrest and genome maintenance. Genes Dev 17:615–628
Chen ST, Choo KB, Hou MF, Yeh KT, Kuo SJ, Chang JG (2005) Deregulated expression of the PER1, PER2 and PER3 genes in breast cancers. Carcinogenesis 26:1241–1246
Chini CC, Wood J, Chen J (2006) Chk1 is required to maintain claspin stability. Oncogene 25(30):4165–4171
Chou DM, Elledge SJ (2006) Tipin and Timeless form a mutually protective complex required for genotoxic stress resistance and checkpoint function. Proc Natl Acad Sci USA 103:18143–18147
Collis SJ, Barber LJ, Ward JD, Martin JS, Boulton SJ (2006) C. elegans FANCD2 responds to replication stress and functions in interstrand cross-link repair. DNA Repair (Amst) 5:1398–1406
Collis SJ, Barber LJ, Clark AJ, Martin JS, Ward JD, Boulton SJ (2007) HCLK2 is essential for the mammalian S-phase checkpoint and impacts on Chk1 stability. Nat Cell Biol 9:391–401
Cuadrado M, Martinez-Pastor B, Murga M, Toledo LI, Gutierrez-Martinez P, Lopez E, Fernandez-Capetillo O (2006) ATM regulates ATR chromatin loading in response to DNA double-strand breaks. J Exp Med 203:297–303
Dengg M, Garcia-Muse T, Gill SG, Ashcroft N, Boulton SJ, Nilsen H (2006) Abrogation of the CLK-2 checkpoint leads to tolerance to base-excision repair intermediates. EMBO Rep 7:1046–1051
Doi M, Hirayama J, Sassone-Corsi P (2006) Circadian regulator CLOCK is a histone acetyltransferase. Cell 125:497–508
Filipski E, King VM, Li X, Granda TG, Mormont MC, Liu X, Claustrat B, Hastings MH, Levi F (2002) Host circadian clock as a control point in tumor progression. J Natl Cancer Inst 94:690–697
Freire R, van Vugt MA, Mamely I, Medema RH (2006) Claspin: timing the cell cycle arrest when the genome is damaged. Cell Cycle 5:2831–2834
Fu L, Pelicano H, Liu J, Huang P, Lee C (2002) The circadian gene Period2 plays an important role in tumor suppression and DNA damage response in vivo. Cell 111:41–50
Garcia-Muse T, Boulton SJ (2005) Distinct modes of ATR activation after replication stress and DNA double-strand breaks in Caenorhabditis elegans. Embo J 24:4345–4355
Gery S, Komatsu N, Baldjyan L, Yu A, Koo D, Koeffler HP (2006) The circadian gene per1 plays an important role in cell growth and DNA damage control in human cancer cells. Mol Cell 22:375–382
Gewurz BE, Harper JW (2006) DNA-damage control: claspin destruction turns off the checkpoint. Curr Biol 16: R932–934
Gorbacheva VY, Kondratov RV, Zhang R, Cherukuri S, Gudkov AV, Takahashi JS, Antoch MP (2005) Circadian sensitivity to the chemotherapeutic agent cyclophosphamide depends on the functional status of the CLOCK/BMAL1 transactivation complex. Proc Natl Acad Sci U S A 102:3407–3412
Gotter AL, Suppa C, Emanuel BS (2007) Mammalian TIMELESS and Tipin are evolutionarily conserved replication fork-associated factors. J Mol Biol 366:36–52
Granda TG, Liu XH, Smaaland R, Cermakian N, Filipski E, Sassone-Corsi P, Levi F (2005) Circadian regulation of cell cycle and apoptosis proteins in mouse bone marrow and tumor. Faseb J 19:304–306
Hardin PE (2005) The circadian timekeeping system of Drosophila. Curr Biol 15:R714–722
Harrison JC, Haber JE (2006) Surviving the breakup: the DNA damage checkpoint. Annu Rev Genet 40:209–235
Hartman PS, Herman RK (1982) Radiation-sensitive mutants of Caenorhabditis elegans. Genetics 102:159–178
Hastings MH, Herzog ED (2004) Clock genes, oscillators, and cellular networks in the suprachiasmatic nuclei. J Biol Rhythms 19:400–413
Hirano Y, Sugimoto K (2006) ATR homolog Mec1 controls association of DNA polymerase zeta-Rev1 complex with regions near a double-strand break. Curr Biol 16:586–590
Hoeijmakers JH (2001) Genome maintenance mechanisms for preventing cancer. Nature 411:366–374
Hurley PJ, Wilsker D, Bunz F (2007) Human cancer cells require ATR for cell cycle progression following exposure to ionizing radiation. Oncogene 26:2535–2542
Jazayeri A, Falck J, Lukas C, Bartek J, Smith GC, Lukas J, Jackson SP (2006) ATM- and cell cycle-dependent regulation of ATR in response to DNA double-strand breaks. Nat Cell Biol 8:37–45
Kastan MB, Bartek J (2004) Cell-cycle checkpoints and cancer. Nature 432:316–323
Kennedy RD, D’Andrea AD (2005) The Fanconi anemia/BRCA pathway: new faces in the crowd. Genes Dev 19: 2925–2940
Lakowski B, Hekimi S (1996) Determination of life-span in Caenorhabditis elegans by four clock genes. Science 272:1010–1013
Larocque JR, Jaklevic BR, Su TT, Sekelsky J (2007) Drosophila ATR in double-strand break repair. Genetics 175(3):1023–1033
Lee CC (2006) Tumor suppression by the mammalian Period genes. Cancer Causes Control 17:525–530
Levitus M, Joenje H, de Winter JP (2006) The Fanconi anemia pathway of genomic maintenance. Cell Oncol 28:3–29
Li XM, Metzger G, Filipski E, Boughattas N, Lemaigre G, Hecquet B, Filipski J, Levi F (1997) Pharmacologic modulation of reduced glutathione circadian rhythms with buthionine sulfoximine: relationship with cisplatin toxicity in mice. Toxicol Appl Pharmacol 143:281–290
Lim CS, Mian IS, Dernburg AF, Campisi J (2001) C. elegans clk-2, a gene that limits life span, encodes a telomere length regulator similar to yeast telomere binding protein Tel2p. Curr Biol 11:1706–1710
Mailand N, Bekker-Jensen S, Bartek J, Lukas J (2006) Destruction of claspin by SCFbetaTrCP restrains Chk1 activation and facilitates recovery from genotoxic stress. Mol Cell 23:307–318
Mamely I, van Vugt MA, Smits VA, Semple JI, Lemmens B, Perrakis A, Medema RH, Freire R (2006) Polo-like kinase-1 controls proteasome-dependent degradation of claspin during checkpoint recovery. Curr Biol 16:1950–1955
Matsuo T, Yamaguchi S, Mitsui S, Emi A, Shimoda F, Okamura H (2003) Control mechanism of the circadian clock for timing of cell division in vivo. Science 302:255–259
Mori T, Saveliev SV, Xu Y, Stafford WF, Cox MM, Inman RB, Johnson CH (2002) Circadian clock protein KaiC forms ATP-dependent hexameric rings and binds DNA. Proc Natl Acad Sci USA 99:17203–17208
Myers JS, Cortez D (2006) Rapid activation of ATR by ionizing radiation requires ATM and Mre11. J Biol Chem 281:9346–9350
Niedernhofer LJ, Lalai AS, Hoeijmakers JH (2005) Fanconi anemia (cross)linked to DNA repair. Cell 123:1191–1198
O’Driscoll M, Jeggo PA (2006) The role of double-strand break repair—insights from human genetics. Nat Rev Genet 7:45–54
Ohdo S, Makinosumi T, Ishizaki T, Yukawa E, Higuchi S, Nakano S, Ogawa N (1997) Cell cycle-dependent chronotoxicity of irinotecan hydrochloride in mice. J Pharmacol Exp Ther 283:1383–1388
Petermann E, Caldecott KW (2006) Evidence that the ATR/Chk1 pathway maintains normal replication fork progression during unperturbed S phase. Cell Cycle 5:2203–2209
Pogue-Geile KL, Lyons-Weiler J, Whitcomb DC (2006) Molecular overlap of fly circadian rhythms and human pancreatic cancer. Cancer Lett 243:55–57
Pregueiro AM, Liu Q, Baker CL, Dunlap JC, Loros JJ (2006) The Neurospora checkpoint kinase 2: a regulatory link between the circadian and cell cycles. Science 313:644–649
Reppert SM, Weaver DR (2002) Coordination of circadian timing in mammals. Nature 418:935–941
Rothschild LJ, Cockell CS (1999) Radiation: microbial evolution, ecology, and relevance to mars missions. Mutat Res 430:281–291
Rutter J, Reick M, Wu LC, McKnight SL (2001) Regulation of Clock and NPAS2 DNA binding by the redox state of NAD cofactors. Science 293:510–514
Schernhammer ES, Laden F, Speizer FE, Willett WC, Hunter DJ, Kawachi I, Colditz GA (2001) Rotating night shifts and risk of breast cancer in women participating in the nurses’ health study. J Natl Cancer Inst 93:1563–1568
Schernhammer ES, Laden F, Speizer FE, Willett WC, Hunter DJ, Kawachi I, Fuchs CS, Colditz GA (2003) Night-shift work and risk of colorectal cancer in the nurses’ health study. J Natl Cancer Inst 95:825–828
Schibler U, Ripperger J, Brown SA (2003) Peripheral circadian oscillators in mammals: time and food. J Biol Rhythms 18:250–260
Shiloh Y (2003) ATM and related protein kinases: safeguarding genome integrity. Nat Rev Cancer 3:155–168
Sorensen CS, Hansen LT, Dziegielewski J, Syljuasen RG, Lundin C, Bartek J, Helleday T (2005) The cell-cycle checkpoint kinase Chk1 is required for mammalian homologous recombination repair. Nat Cell Biol 7:195–201
Stiff T, Reis C, Alderton GK, Woodbine L, O’Driscoll M, Jeggo PA (2005) Nbs1 is required for ATR-dependent phosphorylation events. EMBO J 24:199–208
Takeda DY, Dutta A (2005) DNA replication and progression through S phase. Oncogene 24:2827–2843
Tauber E, Last KS, Olive PJ, Kyriacou CP (2004) Clock gene evolution and functional divergence. J Biol Rhythms 19:445–458
Tercero JA, Diffley JF (2001) Regulation of DNA replication fork progression through damaged DNA by the Mec1/Rad53 checkpoint. Nature 412:553–557
Unsal-Kacmaz K, Mullen TE, Kaufmann WK, Sancar A (2005) Coupling of human circadian and cell cycles by the timeless protein. Mol Cell Biol 25:3109–3116
Unsal-Kacmaz K, Chastain PD, Qu PP, Minoo P, Cordeiro-Stone M, Sancar A, Kaufmann WK (2007) The human Tim/Tipin complex coordinates an intra-S checkpoint response to UV that slows replication fork displacement. Mol Cell Biol 27(8):3131–3142
Wang X, Kennedy R, Ray K, Stuckert P, Ellenberger T, D’Andrea AD (2007) Chk1-mediated phosphorylation of FANCE is required for the Fanconi anemia/BRCA pathway. Mol Cell Biol 27(8):3098–4108
Yeh KT, Yang MY, Liu TC, Chen JC, Chan WL, Lin SF, Chang JG (2005) Abnormal expression of period 1 (PER1) in endometrial carcinoma. J Pathol 206:111–120
Yoshizawa-Sugata N, Masai H (2007) Human Tim/Timeless-interacting protein, Tipin, is required for efficient progression of S phase and DNA replication checkpoint. J Biol Chem 282(4):2729–2740
You S, Wood PA, Xiong Y, Kobayashi M, Du-Quiton J, Hrushesky WJ (2005) Daily coordination of cancer growth and circadian clock gene expression. Breast Cancer Res Treat 91:47–60
Zhang R, Lu Z, Liu T, Soong SJ, Diasio RB (1993) Relationship between circadian-dependent toxicity of 5-fluorodeoxyuridine and circadian rhythms of pyrimidine enzymes: possible relevance to fluoropyrimidine chemotherapy. Cancer Res 53:2816–2822
Zhang YW, Otterness DM, Chiang GG, Xie W, Liu YC, Mercurio F, Abraham RT (2005) Genotoxic stress targets human Chk1 for degradation by the ubiquitin-proteasome pathway. Mol Cell 19:607–618
Zhou BB, Elledge SJ (2000) The DNA damage response: putting checkpoints in perspective. Nature 408:433–439
Zou L, Elledge SJ (2003) Sensing DNA damage through ATRIP recognition of RPA-ssDNA complexes. Science 300:542–1548
Acknowledgments
S.J.C. and S.J.B. are funded by Breast Cancer Campaign (GA3221) and Cancer Research UK.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E.A. Nigg
Rights and permissions
About this article
Cite this article
Collis, S.J., Boulton, S.J. Emerging links between the biological clock and the DNA damage response. Chromosoma 116, 331–339 (2007). https://doi.org/10.1007/s00412-007-0108-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-007-0108-6