Abstract
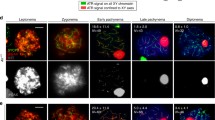
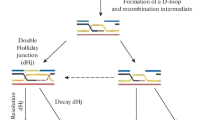
Meiotic recombination in yeast is initiated at DNA double-strand breaks (DSBs), processed into 3′ single-strand overhangs that are active in homology search, repair and formation of recombinant molecules. Are 3′ overhangs recombination intermediaries in mouse germ cells too? To answer this question we developed a novel approach based on the properties of the Klenow enzyme. We carried out two different, successive in situ Klenow enzyme-based reactions on sectioned preparations of testicular tubules. Signals showing 3′ overhangs were observed during wild-type mouse spermatogenesis, but not in Spo11 −/− males, which lack meiotic DSBs. In Atm −/− mice, abundant positively stained spermatocytes were present, indicating an accumulation of non-repaired DSBs, suggesting the involvement of ATM in repair of meiotic DSBs. Thus the processing of DSBs into 3′ overhangs is common to meiotic cells in mammals and yeast, and probably in all eukaryotes.



Similar content being viewed by others
References
Barlow C, Hirotsune S, Paylor R, Liyanage M, Eckhaus M, Collins F et al (1996) Atm-deficient mice: a paradigm of ataxia telangiectasia. Cell 86:159–171
Barlow C, Liyanage M, Moens P, Deng C, Ried T, Wynshaw-Boris A (1997) Partial rescue of the prophase I defects of Atm-deficient mice by p53 and p21 null alleles. Nat Genet 17:462–466
Barlow C, Liyanage M, Moens P, Tarsounas M, Nagashima K, Brown K et al (1998) Atm deficiency results in severe meiotic disruption as early as leptonema of prophase I. Development 125:4007–4017
Baudat F, Manova K, Yuen J, Jasin M, Keeney S (2000) Chromosome synapsis defects and sexually dimorphic meiotic progression in mice lacking spo11. Mol Cell 6:989–998
Bergerat A, de Massy B, Gadelle D, Varoutas PC, Nicolas A, Forterre P (1997) An atypical topoisomerase II from archaea with implication for meiotic recombination. Nature 386:414–417
Bishop DK, Park D, Xu L, Kleckner N (1992) DMC1: a meiosis-specific yeast homolog of E.coli recA required for recombination, synaptonemal complex formation, and cell cycle progression. Cell 69:439–456
Brown K, Barlow C, Wynshaw-Boris A (1999) Multiple ATM-dependent pathways: an explanation for pleiotropy. Am J Hum Genet 64:46–50
Cao L, Alani E, Kleckner N (1990) A pathway for generation and processing of double-strand breaks during meiotic recombination in S. cerevisiae. Cell 61:1089–1101
Cervantes M, Farah J, Smith G (2000) Meiotic DNA breaks associated with recombination in S. pombe. Mol Cell 5:883–888
Chen G, Yuan S, Liu W, Xu Y, Trujillo K, Song B et al (1999) Radiation-induced assembly of Rad51 and Rad52 recombination complex requires ATM and c-Abl. J Biol Chem 274:12748–12752
Clark R, Chen M, Kochanek P, Watkins S, Jin K, Draviam R et al (2001) Detection of single- and double-strand DNA breaks after traumatic brain injury in rats: comparison of in situ labeling techniques using DNA polymerase I, the Klenow fragment of DNA polymerase I, and terminal deoxynucleotidyl transferase. J Neurotrauma 18:675–689
de Massy B, Rocco V, Nicolas A (1995) The nucleotide mapping of DNA double-strand breaks at the CYS3 initiation site of meiotic recombination in Saccharomyces cerevisiae. EMBO J 14:4589–4598
de Vries S, Baart E, Dekker M, Siezen A, de Rooij D, de Boer P et al (1999) Mouse MutS-like protein Msh5 is required for proper chromosome synapsis in male and female meiosis. Genes Dev 13:523–531
Gavrieli Y, Sherman Y, Ben-Sasson S (1992) Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol 119:493–501
Gu Y, Jow G, Moulton B, Lee C, Sensibar J, Park-Sarge O et al (1994) Apoptosis in decidual tissue regression and reorganization. Endocrinology 135:1272–1279
Hawley R, Friend S (1996) Strange bedfellows in even stranger places: the role of ATM in meiotic cells, lymphocytes, tumors, and its functional links to p53. Genes Dev 10:2383–2388
Kassir Y, Simchen G (1991) Monitoring meiosis and sporulation in Saccharomyces cerevisiae. Methods Enzymol 194:94–110
Keeney S (2001) The mechanism and control of meiotic recombination initiation. Curr Top Dev Biol 52:1–53
Kneitz B, Cohen P, Avdievich E, Zhu L, Kane M, Hou HJ et al (2000) MutS homolog 4 localization to meiotic chromosomes is required for chromosome pairing during meiosis in male and female mice. Genes Dev 14:1085–1097
Kornberg A, Baker TA (1991) DNA replication, 2nd edn. WH Freeman, NY
Liu J, Wu T, Lichten M (1995) The location and structure of double-strand DNA breaks induced during yeast meiosis: evidence for a covalently linked DNA-protein intermediate. EMBO J 14:4599–4608
Lydall D, Nikolsky Y, Bishop D, Weinert T (1996) A meiotic recombination checkpoint controlled by mitotic checkpoint genes. Nature 383:840–843
Mahadevaiah S, Turner J, Baudat F, Rogakou E, de Boer P, Blanco-Rodriguez J et al (2001) Recombinational DNA double-strand breaks in mice precede synapsis. Nat Genet 27:271–276
Morrison C, Sonoda E, Takao N, Shinohara A, Yamamoto K, Takeda S (2000) The controlling role of ATM in homologous recombinational repair of DNA damage. EMBO J 19:463–471
Odorisio T, Rodriguez T, Evans E, Clarke A, Burgoyne P (1998) The meiotic checkpoint monitoring synapsis eliminates spermatocytes via p53-independent apoptosis. Nat Genet 18:257–261
Paques F, Haber JE (1999) Multiple pathways of recombination induced by double-strand breaks in Saccharomyces cerevisiae. Microbiol Mol Biol Rev 63:349–404
Pittman D, Cobb J, Schimenti K, Wilson L, Cooper D, Brignull E et al (1998) Meiotic prophase arrest with failure of chromosome synapsis in mice deficient for Dmc1, a germline-specific RecA homolog. Mol Cell 1:697–705
Roeder G, Bailis J (2000) The pachytene checkpoint. Trends Genet 16:395–403
Rogakou E, Boon C, Redon C, Bonner W (1999) Megabase chromatin domains involved in DNA double-strand breaks in vivo. J Cell Biol 46:905–916
Romanienko P, Camerini-Otero R (2000) The mouse spo11 gene is required for meiotic chromosome synapsis. Mol Cell 6:975–987
Rotman G, Shiloh Y (1999) ATM: a mediator of multiple responses to genotoxic stress. Oncogene 18:6135–6144
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Sanchez Y, Desany B, Jones W, Liu Q, Wang B, Elledge S (1996) Regulation of RAD53 by the ATM-like kinases MEC1 and TEL1 in yeast cell cycle checkpoint pathways. Science 271:357–360
Shinohara A, Ogawa H, Ogawa T (1992) Rad51 protein involved in repair and recombination in S. cerevisiae is a RecA-like protein. Cell 69:457–470
Smith KN, Nicolas A (1998) Recombination at work for meiosis. Curr Opin Genet Dev 8:200–211
Sun H, Treco D, Schultes NP, Szostak JW (1989) Double-strand breaks at an initiation site for meiotic gene conversion. Nature 338:87–90
Sun H, Treco D, Szostak JW (1991) Extensive 3′-overhanging, single-stranded DNA associated with the meiosis-specific double-strand breaks at the ARG4 recombination initiation site. Cell 64:1155–1161
Tsuzuki T, Fujii Y, Sakumi K, Tominaga Y, Nakao K, Sekiguchi M et al (1996) Targeted disruption of the Rad51 gene leads to lethality in embryonic mice. Proc Natl Acad Sci U S A 93: 6236–6240
Usui T, Ogawa H, Petrini J (2001) A DNA damage response pathway controlled by Tel1 and the Mre11 complex. Mol Cell 7:1255–1266
Xu Y, Ashley T, Brainerd E, Bronson R, Meyn M, Baltimore D (1996) Targeted disruption of ATM leads to growth retardation, chromosomal fragmentation during meiosis, immune defects, and thymic lymphoma. Genes Dev 10:2411–2422
Yoshida K, Kondoh G, Matsuda Y, Habu T, Nishimune Y, Morita T (1998) The mouse RecA-like gene Dmc1 is required for homologous chromosome synapsis during meiosis. Mol Cell 1:707–718
Zenvirth D, Simchen G (2000) Meiotic double-strand breaks in Schizosaccharomyces pombe. Curr Genet 38:33–38
Zenvirth D, Arbel T, Sherman A, Goldway M, Klein S, Simchen G (1992) Multiple sites for double-strand breaks in whole meiotic chromosomes of Saccharomyces cerevisiae. EMBO J 11:3441–3447
Zickler D, Kleckner N (1998) The leptotene-zygotene transition of meiosis. Annu Rev Genet 32:619–697
Acknowledgements
We thank Yossi Shiloh for useful discussions and helpful comments on the manuscript. This work was supported by grants from the USA/Israel Binational Science Foundation (BSF). A.B. was supported by a Lady Davis postdoctoral fellowship and F.B. by a fellowship from the Charles H. Revson Foundation and by grants to the laboratories of Maria Jasin and Scott Keeney.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by: P. Moens
The first three authors contributed equally to this work
Rights and permissions
About this article
Cite this article
Zenvirth, D., Richler, C., Bardhan, A. et al. Mammalian meiosis involves DNA double-strand breaks with 3′ overhangs. Chromosoma 111, 369–376 (2003). https://doi.org/10.1007/s00412-002-0223-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-002-0223-3