Abstract
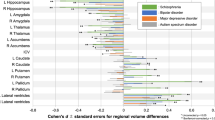
The septal nuclei are assumed to play a significant role in the pathophysiology of schizophrenia and affective disorders. The aim of this study was to morphometrically characterize the septal nuclei in patients with schizophrenia, bipolar disorder, and major depressive disorder, when compared with healthy control subjects. We analyzed the septal nuclei by determining the density and size of the neurons in postmortem brains in 17 patients with schizophrenia, 8 patients with bipolar disorder, 7 patients with major depressive disorder, and 14 control subjects matched for age and gender. There was a significant reduction in the neuronal density, but not in the mean cross-sectional area, in the lateral septal nucleus (P = 0.013) in patients with bipolar disorder when compared with control subjects. There were no significant changes in the neuronal density of the septal nuclei of the medial and lateral cell groups in patients with schizophrenia and major depressive disorder when compared with control subjects. There was a significant negative correlation between neuronal density in the lateral septal nucleus and disease duration in patients with major depressive disorder (P = 0.037, r = −0.9). The histopathological abnormality of the decreased neuronal density in the lateral septal nucleus, which is an important limbic region involved in emotions, might be a neuropathological correlate of bipolar disorder.




Similar content being viewed by others
References
McNaughton N, Corr PJ (2004) A two-dimensional neuropsychology of defence: fear/anxiety and defensive distance. Neurosci Biobehav Rev 28:285–305
Sheehan TP, Chambers RA, Russell DR (2004) Regulation of affect by the lateral septum: implications for neuropsychiatry. Brain Res Brain Res Rev 46:71–117
Lubar JF, Numan R (1973) Behavioral and physiological studies on septal function and related medial cortical structures. Behav Biol 8:1–25
Danner H, Pfister C (1981) Investigation on the cytoarchitecture of the nucleus accumbens septi of rat. Anat Anz 150:264–280
Smith KS, Berridge KC (2007) Opioid limbic circuit for reward: interaction between hedonic hotspots of nucleus accumbens and ventral pallidum. J Neurosci 27:1594–1605
Kreczmanski P, Heinsen H, Mantua V, Woltersdorf F, Masson T, Ulfig N, Schmidt-Kastner R, Korr H, Steinbusch HWM, Hof PR, Schmitz C (2007) Volume, neuron density and total neuron number in five subcortical regions in schizophrenia. Brain 130:678–692
Brady JV, Nauta WJH (1953) Subcortical mechanisms in emotional behaviour: affective changes following septal forebrain lesions in the albino rat. J Comp Physiol Psych 46:339–346
Turgeon SM, Kegel G, Davis MM (2001) Electrolytic lesions of the medial septum enhance latent inhibition in a conditioned taste aversion paradigm. Brain Res 890:333–337
van der Staay FJ, Bouger P, Lehmann O, Lazarus C, Cosquer B, Koenig J, Stump V, Cassel JC (2006) Long-term effects of immunotoxicy cholinergic lesions in the septum on acquisition of the cone-field task and noncognitive measures in rats. Hippocampus 16:1061–1079
Brockhaus H (1942) Zur feineren Anatomie des Septums und des Striatums. J Psychol Neurol 51:1–56
Horvath S, Palkovits M (1987) Morphology of the human septal area: a topographic atlas. Acta Morphol Hung 35:157–174
Andy OJ, Stephan H (1969) The septum in the human brain. J Comp Neurol 133:383–410
Ulfig N, Braack H (1989) Neuronal types and their percent distribution within the magnocellular nuclei of the human basal forebrain. Acta Anat 134:237–241
Zeman W, King FA (1958) Tumour of the septum pellucidum and adjacent structures with abnormal affective behavior: an anterior midline structure syndrome. J Nerv Ment Dis 127:490–502
Arendt T, Bigl V, Arendt A, Tennstedt A (1983) Loss of neurons in the nucleus basalis of Meynert in Alzheimer’s disease, paralysis agitans, and Korsakoff’s disease. Acta Neuropathol (Berl) 61:101–108
Averback P (1981) Structural lesions of the brain in young schizophrenics. Can J Neurol Sci 8:73–76
Beck E, Gajdusek DC (1966) Variable size of the septal nuclei in man. Nature 210:1338–1340
Heath RG, Walker CF (1985) Correlation of deep and surface electroencephalograms with psychosis and hallucinations in schizophrenics: a report of two cases. Biol Psychiatry 20:669–674
Heath RG, Dempsey CW, Fontana CJ, Fitzjarelli AT (1980) Feedback loop between cerebellum and septo-hippocampal sites: its role in emotion and epilepsy. Biol Psychiatry 15:541–546
Bogerts B (1997) The temporolimbic system theory of positive schizophrenic symptoms. Schizophr Bull 23:423–435
Rajkowska G (2002) Cell pathology in mood disorder. Sem Clin Neuropsychiatry 7:281–292
Danos P, Baumann B, Krämer A, Bernstein H-G, Stauch R, Krell D, Falkai P, Bogerts B (2003) Volumes of association thalamic nuclei in schizophrenia: a postmortem study. Schizophr Res 60:141–155
Mai JK, Assheuer J, Paxinos G (1997) Atlas of the human brain. Academic Press Harcourt Brace & Company, San Diego, London, Boston, New York, Sydney, Tokyo, Toronto, pp 154–185
Schumann CM, Amaral DG (2005) Stereological estimation of the number of neurons in the human amygdaloid complex. J Comp Neurol 491:320–329
Brisch R, Bernstein H-G, Krell D, Dobrowolny H, Bielau H, Steiner J, Gos T, Funke S, Stauch R, Knüppel S, Bogerts B (2009) Dopamine-glutamate abnormalities in the frontal cortex associated with the catechol-O-methyltransferase (COMT) in schizophrenia. Brain Res 1269:166–175
Gundersen HJ, Jensen EB, Kieu K, Nielsen J (1999) The efficiency of systematic sampling in stereology-reconsidered. J Microsc 151:3–21
Brisch R, Bernstein H-G, Krell D, Stauch R, Dowbrowolny H, Trübner K, Kropf S, Bielau H, Bogerts B (2007) Volumetric analysis of septal region in schizophrenia and affective disorder. Eur Arch Psychiatry and Clin Neurosci 257:140–148
Beyer JL, Krishnan KRR (2002) Volumetric brain imaging findings in mood disorders. Bipolar Disord 4:89–104
Campbell S, MacQueen G (2006) An update on regional brain volume differences associated with mood disorders. Curr Opin Psychiatry 19:25–33
Takahashi T, Malhi GS, Wood SJ, Yücel M, Walterfang M, Nakamura K, Suzuki M, Pantelis C (2010) Midline brain abnormalities in established bipolar affective disorder. J Affect Disord 25:140–148
Radenbach K, Flaig V, Schneider-Axmann T, Usher J, Reith W, Falkai P, Gruber O, Scherk H (2010) Thalamic volumes in patients with bipolar disorder. Eur Arch Psychiatry Clin Neurosci. doi:10.1007/s00406-010-0100-7
Dahabra S, Ashton CH, Bahrainian M, Britton PG, Ferrier IN, McAllister VA, Marsh VR, Moore PB (1998) Structural and functional abnormalities in elderly patients clinically recovered from early- and late-onset depression. Biol Psychiatry 44:34–46
Sheline YI, Gado MH, Price JL (1998) Amygdala core nuclei are decreased in recurrent major depression. Neuroreport 9:2023–2027
Bremner JD, Narayan M, Anderson ER, Staib LH, Miller HL, Charney DS (2000) Hippocampal volume reduction in major depression. Am J Psychiatry 157:115–117
Frodl T, Meisenzahl EM, Zetsche T, Born C, Groll C, Jäger M, Leinsinger G, Bottlender R, Hahn K, Möller H-J (2002) Hippocampal changes in patients with a first episode of major depression. Am J Psychiatry 159:1112–1118
Posener JA, Wang L, Price JL, Gado MH, Province MA, Miller MI, Babb CM, Csernansky JG (2003) High-dimensional mapping of the hippocampus in depression. Am J Psychiatry 160:83–89
Selemon LD, Rajkowska G (2003) Cellular pathology in the dorsolateral prefrontal cortex distinguishes schizophrenia from bipolar disorder. Curr Mol Med 3:427–438
Benes FM, Vincent SL, Todtenkopf M (2001) The density of pyramidal and nonpyramidal neurons in anterior cingulate cortex of schizophrenic and bipolar subjects. Biol Psychiatry 50:395–406
Todtenkopf MS, Vincent SL, Benes FM (2005) A cross-study meta-analysis and three- dimensional comparison of cell counting in the anterior cingulate cortex of schizophrenia and bipolar brain. Schizophr Res 73:79–89
Benes FM, Kwok EW, Vincent SL, Todtenkopf MS (1998) A reduction of nonpyramidal cells in sector CA2 of schizophrenics and manic depressives. Biol Psychiatry 44:88–97
Bouras C, Kövari E, Hof PR, Riederer BM, Giannakopoulos P (2001) Anterior cingulate cortex pathology in schizophrenia and bipolar disorder. Acta Neuropathol 102:373–379
Beretta S, Pantazopoulos H, Lange N (2007) Neuron numbers and volume of the amygdala in subjects diagnosed with bipolar disorder or schizophrenia. Biol Psychiatry 62:884–893
Chana G, Landau S, Beasley C, Everall IP, Cotter D (2003) Two-dimensional assessment of cytoarchitecture in the anterior cingulate cortex in major depressive disorder, bipolar disorder, and schizophrenia: evidence for decreased neuronal somal size and increased density. Biol Psychiatry 53:1086–1098
Cotter D, Mackay D, Landau S, Kerwin R, Everall I (2001) Reduced glial cell density and neuronal size in the anterior cingulate cortex in major depressive disorder. Arch Gen Psychiatry 58:545–553
Cotter D, Mackay D, Frangou S, Hudson L, Landau S (2004) Cell density, cortical thickness in Heschl`s gyrus in schizophrenia, major depression, bipolar disorder. Br J Psychiatry 185:258–259
Öngür D, Drevets WC, Price JL (1998) Glial reduction in the subgenual prefrontal cortex in mood disorders. Proc Natl Acad Sci USA 95:13290–13295
Cotter D, Hudson L, Landau S (2005) Evidence for orbitofrontal pathology in bipolar disorder and major depression, but not in schizophrenia. Bipolar Disord 7:358–369
Liu L, Schulz CS, Lee S, Reutiman TJ, Fatemi SH (2007) Hippocampal CA1 pyramidal cell size is reduced in bipolar disorder. Cell Mol Neurobiol 27:351–358
Bezchlibnyk YB, Sun X, Wang J-F, MacQueen GM, McEwen BS, Young LT (2007) Neuron somal size is decreased in the lateral amygdalar nucleus of subjects with bipolar disorder. J Psychiatry Neurosci 32:203–210
Young KA, Holcomb LA, Yazdani U, Hicks PB, German DC (2004) Elevated neuron number in the limbic thalamus in major depression. Am J Psychiatry 161:1270–1277
Stockmeier CA, Mahajan GJ, Konick LC, Overholser JC, Jurius GJ, Meltzer HY, Uylings HBM, Friedman L, Rajkowska G (2004) Cellular changes in the postmortem hippocampus in major depression. Biol Psychiatry 56:640–650
Amunts K, Zilles K (2007) Funktionelle Neuroanatomie. In: Schneider F, Fink GR (eds) Funktionelle MRT in Psychiatrie und Neurologie. Springer Medizin Verlag Heidelberg, 1, Auflage, pp 1–58
Grossman SP (1977) An experimental dissection of the septal syndrome. Ciba Found Symp 58:227–273
Martin MM, Horn KL, Kusman KJ, Wallace DG (2007) Medial septum lesions disrupt exploratory trip organization: evidence for septohippocampal involvement in dead reckoning. Physiol Behav 90:412–424
Cavazos JE, Wang C-J, Sitoh Y-Y, Ng SES, Tien RD (1997) Anatomy and pathology of the septal region. Neuroimaging Clin N Am 7:67–78
Baumann B, Bielau H, Krell D, Agelink MW, Diekmann S, Wurthmann C, Trübner K, Bernstein H-G, Danos P, Bogerts B (2002) Circumscribed numerical deficit of dorsal raphe neurons in mood disorders. Psychol Med 32:93–103
Bernstein H-G, Stanarius A, Baumann B, Henning H, Krell D, Falkai P, Bogerts B (1998) Nitric oxide synthase-containing neurons in the human hypothalamus: reduced number of immunoreactive cells in the paraventricular nucleus of depressive patients and schizophrenics. Neuroscience 83:867–875
Baumann B, Bogerts B (2001) Neuroanatomical studies of bipolar disorder. Br J Psychiatry Suppl 178:s142–s147
Bernstein H-G, Heinemann A, Krell D, Mawrin C, Bielau H, Danos P, Diekmann S, Keilhoff G, Bogerts B, Baumann B (2002) Further immunohistochemical evidence for impaired NO signalling in the hypothalamus of depressed patients. Ann NY Acad Sci 973:91–93
Bielau H, Mawrin C, Krell D, Agelink MW, Trübner K, Davis R, Gos T, Bogerts B, Bernstein H-G, Baumann B (2005) Differences in activation of the dorsal raphe nucleus depending on performance of suicide. Brain Res 255:401–412
Manaye KF, Lei D-L, Tizabi Y, Davila-Garcia MI, Mouton PR, Kelly PH (2005) Selective neuron loss in the paraventricular nucleus of hypothalamus in patients suffering from major depression and bipolar disorder. J Neuropathol Exp Neurol 64:224–229
Ranft K, Dobrowolny H, Krell D, Bielau H, Bogerts B, Bernstein H-G (2010) Evidence for structural abnormalities of the human habenular complex in affective disorders but not in schizophrenia. Psychol Med 40:557–567
Watson S, Gallagher P, Ritchie JC, Ferrier N, Young AH (2004) Hypothalamic- pituitary-adrenal-axis function in patients with bipolar disorder. Br J Psychiatry 184:496–502
Daban C, Vieta E, Mackin P, Young AH (2005) Hypothalamic-pituitary-adrenal axis and bipolar disorder. Psychiatr Clin North Am 28:469–480
Jokinen J, Nordström P (2009) HPA axis hyperactivity and attempted suicide in young adult mood disorder patients. J Affect Disord 116:117–120
Pompili M, Serafini G, Innamorati M, Möller-Leimkühler AM, Giupponi G, Girardi P, Tatarelli R, Lester D (2010) The hypothalamic-pituitary-adrenal axis and serotonin abnormalities: a selective overview for the implications of suicide prevention. Eur Arch Psychiatry Clin Neurosci. doi:10.1007/s00406-010-0108-z
Murakami S, Imbe H, Morikawa Y, Kuobo C, Senba E (2005) Chronic stress, as well as acute stress, reduces BDNF mRNA expression but less robustly. Neurosci Res 53:129–139
Das GD, Altman J (1970) Postnatal neurogenesis in the caudate nucleus and nucleus accumbens septi. Brain Res 21:122–127
Raisman G (1969) Neuronal plasticity in the septal nuclei of the adult rat. Brain Res 14:25–48
Steciuk M, Kram M, Kramer GL, Petty F (1999) Decrease in stress-induced c-Fos-like immunoreactivity in the lateral septal nucleus of learned helpless rats. Brain Res 822:256–259
Huang YH, Cheng C-Y, Hong C-J, Tsai SJ (2004) Expression of c-Fos-like immunoreactivity in the brain of mice with learned helplessness. Neurosci Lett 363:280–283
Zhang J-H, Pimenta AF, Levitt P, Zhou R (1997) Dynamic expression suggests multiple roles of the EPH family receptor brain-specific kinase (Bsk) during mouse neurogenesis. Mol Brain Res 47:202–214
Bernier PJ, Vinet J, Cossette M, Parent A (2000) Characterization of the sub ventricular zone of the adult human brain: evidence for the involvement of Bcl-2. Neurosci Res 37:67–78
Pencea V, Bingaman KD, Wiegand SJ, Luskin MB (2001) Infusion of brain-derived neurotrophic factor into the lateral ventricle of the adult rat leads to new neurons in the parenchyma of the striatum, septum, thalamus, and hypothalamus. J Neurosci 21:6706–6717
Wakade CG, Mahadik SP, Waller JL, Chiu FC (2002) Atypical neuroleptics stimulate neurogenesis in adult rat brain. J Neurosci Res 69:72–79
Kodama M, Fujioka T, Duman RS (2004) Chronic olanzapine or fluoxetine administration increases cell proliferation in hippocampus and prefrontal cortex of adult rat. Biol Psychiatry 56:570–580
Chen Z, Xu H, Haimano S, Li X, Li XM (2005) Quetiapine and venlafaxine synergically regulate heme oxygenase-2 protein expression in the hippocampus of stressed rats. Neurosci Lett 389:173–177
Luo C, Xu H, Li XM (2005) Quetiapine reverses the suppression of hippocampal neurogenesis caused by repeated restraint stress. Brain Res 1063:32–39
Csernansky JG, Martin MV, Czeisler B, Meltzer MA, Ali Z, Dong H (2006) Neuroprotective effects of olanzapine in a rat model of neurodevelopment injury. Pharmacol Biochem Behav 83:208–213
Xu H, Chen Z, He J, Haimanot S, Li X, Dyck L, Li XM (2006) Synergetic effects of quetiapine and venlafaxine in preventing the chronic restraint stress-induced decrease in cell proliferation and BDNF expression in rat hippocampus. Hippocampus 16:551–559
Weinberger DR (1987) Implications of normal brain development for the pathogenesis of schizophrenia. Arch Gen Psychiatry 44:660–669
Lipska BK (2004) Using animal models to test a neurodevelopmental hypothesis of schizophrenia. J Psychiatry Neurosci 29:282–289
Brisch R, Bernstein H-G, Stauch R, Dobrowolny H, Krell D, Truebner K, Meyer-Lotz G, Bielau H, Steiner J, Kropf S, Gos T, Danos P, Bogerts B (2008) The volumes of the fornix in schizophrenia and affective disorders: A post-mortem study. Psychiatry Res 164:265–273
Acknowledgments
This study was supported by the Stanley Foundation and NBL-3/BMBF (Förderkennzeichen 01ZZ0407).
Conflict of interest statement
All authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brisch, R., Bernstein, HG., Dobrowolny, H. et al. A morphometric analysis of the septal nuclei in schizophrenia and affective disorders: reduced neuronal density in the lateral septal nucleus in bipolar disorder. Eur Arch Psychiatry Clin Neurosci 261, 47–58 (2011). https://doi.org/10.1007/s00406-010-0119-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00406-010-0119-9