Abstract
Objective
Auditory neuropathy spectrum disorder (ANSD) is a condition where cochlear amplification function (involving outer hair cells) is normal but neural conduction in the auditory pathway is disordered. This study was done to investigate the cortical representation of speech in individuals with ANSD and to compare it with the individuals with normal hearing.
Design
Forty-five participants including 21 individuals with ANSD and 24 individuals with normal hearing were considered for the study. Individuals with ANSD had hearing thresholds ranging from normal hearing to moderate hearing loss. Auditory cortical evoked potentials—through odd ball paradigm—were recorded using 64 electrodes placed on the scalp for /ba/-/da/ stimulus. Onset cortical responses were also recorded in repetitive paradigm using /da/ stimuli. Sensitivity and reaction time required to identify the oddball stimuli were also obtained.
Result
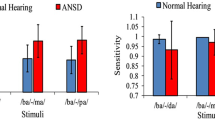
Behavioural results indicated that individuals in ANSD group had significantly lower sensitivity and longer reaction times compared to individuals with normal hearing sensitivity. Reliable P300 could be elicited in both the groups. However, a significant difference in scalp topographies was observed between the two groups in both repetitive and oddball paradigms. Source localization using local auto regressive analyses revealed that activations were more diffuses in individuals with ANSD when compared to individuals with normal hearing sensitivity.
Conclusion
Results indicated that the brain networks and regions activated in individuals with ANSD during detection and discrimination of speech sounds are different from normal hearing individuals. In general, normal hearing individuals showed more focused activations while in individuals with ANSD activations were diffused.








Similar content being viewed by others
References
Starr A, Picton T, Sininger Y, Hood L, Berlin C (1996) Auditory neuropathy. Brain 119:741–753. https://doi.org/10.1093/brain/119.3.741
Starr A, Sininger YS, Pratt H (2000) The varieties of auditory neuropathy. J Basic Clin Physiol Pharmacol 11:215–230. https://doi.org/10.1515/jbcpp.2000.11.3.215
Berlin C, Hood L, Wilensky D, John P, Montgomery E, Thibodaux M (2005) Absent or elevated middle ear muscle reflexes in the presence of normal otoacoustic emissions: a universal finding in 136 cases of auditory neuropathy/dys-synchrony. J Am Acad Audiol 16:546–553
Zeng F, Oba S, Garde S et al (2001) Psychoacoustic and speech perception in auditory neuropathy. In: Sininger Y, Starr A (eds) Auditory neuropathy: a new perspective on hearing disorder. Singular publishing group, Canada, pp 141–164
Sininger Y, Oba S (2001) Patients with auditory neuropathy: who are they and what can they hear? In: Sininger Y, Starr A (eds) Auditory neuropathy: a new perspective on hearing disorder. Singular Publishing Group, Montreal, pp 15–36
Narne VK, Vanaja CS (2008) Speech identification and cortical potentials in individuals with auditory neuropathy. Behav Brain Funct 4(15):2–9
Kraus N, Bradlow A, Cheatham M, Cunningham J, King C, Koch D et al (2000) Consequences of neural asynchrony: a case of auditory neuropathy. JARO 1:33–45
Abdeltawwab M (2014) Auditory N1-P2 cortical event related potentials in auditory neuropathy spectrum disorder patients. J Int Adv Otol 10:270–274. https://doi.org/10.5152/iao.2014.104
Kumar UA, Jayaram M (2005) Auditory processing in individuals with auditory neuropathy. Behav Brain Funct 1:21. https://doi.org/10.1186/1744-9081-1-21
Rance G, Cone-wesson B, Wunderlich J, Dowell R (2002) Speech perception and cortical event related potentials in children with auditory neuropathy. Ear Hear 23:239–253
Narne VK, Prabhu P, Chandan H, Deepthi M (2014) Audiological profiling of 198 individuals with auditory neuropathy spectrum disorder. Hear Balanc Commun 12:112–120. http://informahealthcare.com/doi/abs/10.3109/21695717.2014.938481. Accessed 13 Jan 2015 (Internet)
Mcmaster ML, Kristinsson SY, Turesson I, Bjorkholm M, Landgren O, Husain FT et al (2011) Neuroanatomical changes due to hearing loss and chronic tinnitus: a combined VBM and DTI study. Brain Res 1369:74–88
Peelle JE, Troiani V, Grossman M, Wingfield A (2011) Hearing loss in older adults affects neural systems supporting speech comprehension. J Neurosci 31:12638–12643. https://doi.org/10.1523/JNEUROSCI.2559-11.2011
Boyen K, Langers D, de Kleine E, Van Dijk P (2013) Gray matter in the brain: differences associated with tinnitus and hearing loss. Hear Res 295:67–78. https://doi.org/10.1016/j.heares.2012.02.010 (Internet, Elsevier B.V.)
Lin Y, Wang J, Wu C, Wai Y, Yu J, Ng S (2008) Diffusion tensor imaging of the auditory pathway in sensorineural hearing loss: changes in radial diffusivity and diffusion anisotropy. J Magn Reson Imaging 28:598–603
Profant O, Škoch A, Balogová Z, Tintěra J, Hlinka J, Syka J (2014) Diffusion tensor imaging and MR morphometry of the central auditory pathway and auditory cortex in aging. Neuroscience 260:87–97
Cardin V (2016) Effects of aging and adult-onset hearing loss on cortical auditory regions. Front Neurosci 10:1–12
Venkatesan S (2009) Ethical guidelines for bio-behavioral research involving human subjects. All India Institute of Speech and Hearing, Mysore
Semlitsch HV, Anderer P, Schuster P, Presslich O (1986) A solution for reliable and valid reduction of ocular artifacts, applied to the P300 ERP. Psychophysiology 23(6):695–703
Brunet D, Murray M, Michel C (2011) Spatiotemporal analysis of multichannel EEG: CARTOOL. Comput Intell Neurosci 2011:1–15
de Peralta Menendez RG, Andino SG, Lantz G, Michel CM, Landis T (2001) Noninvasive localization of electromagnetic epileptic activity. I. Method descriptions and simulations. Brain Topogr 14(2):131–137
Murray MM, Michel CM, Grave de Peralta R, Ortigue S, Brunet D, Gonzalez Andino S et al (2004) Rapid discrimination of visual and multisensory memories revealed by electrical neuroimaging. Neuroimage 21(1):125–135
De Santis L, Clarke S, Murray M (2007) Automatic and intrinsic auditory “what” and “where” processing in humans revealed by electrical neuroimaging. Cereb Cortex 17(1):9–17
Spierer L, Tardif E, Sperdin H, Murray MM, Clarke S (2007) Learning-induced plasticity in auditory spatial representations revealed by electrical neuroimaging. J Neurosci 27(20):5474–5483
Lehmann D, Skrandies W (1980) Reference-free identification of components of checkerboard-evoked multichannel potential fields. Electroencephalogr Clin Neurophysiol 48(6):609–621
Krzanowski WJ, Lai YT (1988) A criterion for determining the number of groups in a data set using sum of squares clustering. Biometrics 44(1):23–34
Mazziotta J, Toga A, Evans A, Fox P, Lancaster J, Zilles K et al (2001) A probabilistic atlas and reference system for the human brain: international consortium for brain mapping (ICBM). Philos Trans R Soc London Ser B 356(1412):1293–1322
Michel CM, Koenig T, Brandeis D, Gianotti LRR, Wackermann J (2009) Electrical neuroimaging, 1st edn. Cambridge University Press, Cambridge, p 250
Murray MM, Brunet D, Michel CM (2008) Topographic ERP analyses: a step-by-step tutorial review. Brain Topogr 20(4):249–264
McMahon CM, Patuzzi RB, Gibson WP, Sanli H (2008) Frequency-specific electrocochleography indicates that presynaptic and postsynaptic mechanisms of auditory neuropathy exist. Ear Hear 29:314–325. https://doi.org/10.1097/AUD.0b013e3181662c2a
Polish J (2003) Theoretical overview of P3a and P3b. In: Polish J (eds) Detection of change. Springer, Boston, MA, pp 89–98
Picton TW (1992) The P300 wave of the human event-related potential. J Clin Neurophysiol 9(4):456–479
Swick D, Kutas M, Neville H (1994) Localizing the neural generators of event-related brain potentials. Localization Neuroimaging Neuropsychol 73–121
Johnson R (1993) On the neural generators of the P300 component of the event-related potential. Psychophysiology 30:90–97
Menon V, Uddin LQ (2010) Saliency, switching, attention and control: a network model of insula function. Brain Struct Funct 214:655–667
Tops M, Boksem MAS (2011) A potential role of the inferior frontal gyrus and anterior insula in cognitive control, brain rhythms, and event-related potentials. Front Psychol 2:1–14
Ranganath C, Rainer G (2003) Neural mechanisms for detecting and remembering novel events. Nat Rev Neurosci 4(3):193–202
Acknowledgements
We sincerely thank Director, All India Institute of Speech and Hearing and Head, Department of Audiology. We like to thank all the participants of this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Apeksha, K., Kumar, U.A. Cortical processing of speech in individuals with auditory neuropathy spectrum disorder. Eur Arch Otorhinolaryngol 275, 1409–1418 (2018). https://doi.org/10.1007/s00405-018-4966-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-018-4966-8