Abstract
Purpose
The objective of this study was to achieve uniform reporting of complications and failures in cochlear implantation, to analyze complications and failures and to identify risk factors for complications in a series of over 1300 cochlear implantations.
Methods
In a retrospective chart review and observational study, data from all cochlear implantations from 1987 to 2015 were entered in a custom-made database. Complications were classified using the contracted form of the Clavien–Dindo system and risk factors were identified by statistical analysis.
Results
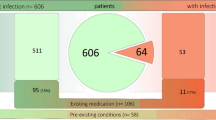
A complication rate of 18.4% and a device failure rate of 2.9% were found. There was a higher rate of hematoma in patients with a clotting disorder and when a subtotal petrosectomy was performed, a higher rate of wound infections in patients who were not vaccinated against Streptococcus pneumoniae and a higher rate of meningitis in patients with an inner ear malformation.
Conclusions
The use of a strict definition of a medical complication and device failure—in combination with the Clavien–Dindo classification system—enables uniform and objective registration of adverse events and prevents any tendency to downgrade complications. Complication and failure rates in this series are comparable to those reported in the literature. These results stress the need for pneumococcal vaccination, which may prevent general wound infections, but is especially important for patients with inner ear malformation, who have an increased risk of (postoperative) meningitis.


Similar content being viewed by others
References
Cohen NL, Hoffman RA (1991) Complications of cochlear implant surgery in adults and children. Ann Otol Rhinol Laryngol 100(9 Pt 1):708–711
Venail F, Sicard M, Piron JPet al (2008) Reliability and complications of 500 consecutive cochlear implantations. Arch Otolaryngol Head Neck Surg 134(12):1276–1281
Hansen S, Anthonsen K, Stangerup S-E, Jensen JH, Thomsen J, Cayé-Thomasen P (2010) Unexpected findings and surgical complications in 505 consecutive cochlear implantations: a proposal for reporting consensus. Acta Otolaryngol 130(5):540–549
Loundon N, Blanchard M, Roger G, Denoyelle F, Garabedian EN (2010) Medical and surgical complications in pediatric cochlear implantation. Arch Otolaryngol Head Neck Surg 136(1):12–15
Jeppesen J, Faber CE (2013) Surgical complications following cochlear implantation in adults based on a proposed reporting consensus. Acta Otolaryngol 133(10):1012–1021
Ernst A, Todt I, Wagner J (2015) Safety and effectiveness of the Vibrant Soundbridge in treating conductive and mixed hearing loss: a systematic review. Laryngoscope 126(6):1451–1457
Zwartenkot JW, Mulder JJS, Snik AFM, Cremers CWRJ., Mylanus EAM (2016) Active middle ear implantation: long-term medical and technical follow-up, implant survival, and complications. Otol Neurotol 37(5):513–519
Smyth GD, Kerr AG, Goodey RJ (1971) Current thoughts on combined approach tympanoplasty. IV. Results and complications. J Laryngol Otol 85(10):1021–1029
Walker PC, Mowry SE, Hansen MR, Gantz BJ (2014) Long-term results of canal wall reconstruction tympanomastoidectomy. Otol Neurotol 35(1):e24-30
Cohen N, Ramos A, Ramsden R et al (2009) International consensus on meningitis and cochlear implants. Acta Otolaryngol 125(9):916–917
Balkany TJ, Hodges AV, Buchman CAet al (2005) Cochlear implant soft failures consensus development conference statement. Otol Neurotol 26(4):815–818
(2005) European consensus statement on cochlear implant failures and explantations. Otol Neurotol 26(6):1097–1099
Clavien PA, Sanabria JR, Strasberg SM (1992) Proposed classification of complications of surgery with examples of utility in cholecystectomy. Surgery 111(5):518–526
Dindo D, Demartines N, Clavien P-A (2004) Classification of surgical complications. Ann Surg 240(2):205–213
Theunisse HJ, Mulder JJ, Pennings RJE, Kunst HPM, Mylanus EAM (2014) A database system for the registration of complications and failures in cochlear implant surgery applied to over 1000 implantations performed in Nijmegen, The Netherlands. J Laryngol Otol 128(11):952–957
Reefhuis J, Honein MA, Whitney CG et al (2003) Risk of bacterial meningitis in children with cochlear implants. N Engl J Med 349(5):435–445
Cunningham C 3rd, Slattery W 3rd, Luxford W (2004) Postoperative infection in cochlear implant patients. Otolaryngol Head Neck Surg 131(1):109–114
Hopfenspirger MT, Levine SC, Rimell FL (2007) Infectious complications in pediatric cochlear implants. Laryngoscope 117(10):1825–1829
Mheen Marang-van de PJ, Kievit J (2003) Automated registration of adverse events in surgical patients in the Netherlands: the current status. Ned Tijdschr Geneeskd 147(26):1273–1277
Buijs EFM, Theunisse HJ, Mulder JJ et al (2015) The use of gentamicin-impregnated collagen sponges (Garacol(®) /Duracoll(®)) in cochlear implant infections: our experience in four cases. Clin Otolaryngol 40(5):492–495
Clavien PA, Barkun J, de Oliveira MLet al (2009) The Clavien–Dindo classification of surgical complications: five-year experience. Ann Surg 250(2):187–196
Muzzi E, Battelino S, Gregori M, Pellegrin A, Orzan E (2015) Life-threatening unilateral hearing impairments. Review of the literature on the association between inner ear malformations and meningitis. Int J Pediatr Otorhinolaryngol 79(12):1969–1974
Korol E, Johnston K, Waser N et al (2013) A systematic review of risk factors associated with surgical site infections among surgical patients. PLoS One 8(12):e83743
Anne S, Ishman SL, Schwartz S (2016) A systematic review of perioperative versus prophylactic antibiotics for cochlear implantation. Ann Otol Rhinol Laryngol 125(11):893–899
Antonelli PJ, Ojano-Dirain CP (2013) Microbial flora of cochlear implants by gene pyrosequencing. Otol Neurotol 34(7):e65-71
Filipo R, D’elia C, Covelli Eet al (2009) Haematoma after cochlear implantation: management of a minor complication. Acta Otolaryngol 130(1):108–113
Tarkan Ö, Tuncer Ü, Özdemir S et al (2013) Surgical and medical management for complications in 475 consecutive pediatric cochlear implantations. Int J Pediatr Otorhinolaryngol 77(4):473–479
Alzoubi F, Odat H, Nuseir A, Al Omari A, Al-Zuraiqi B (2015) Effect of otitis media with effusion on cochlear implant surgery: technical difficulties, post-operative complications and outcome. J Laryngol Otol 129(8):762–766
Saito T, Manabe Y, Shibamori Y (2001) Long-term follow-up results of electrogustometry and subjective taste disorder after middle ear surgery. Laryngoscope 111(11 Pt 1):2064–2070
Battmer R-D, O’Donoghue GM, Lenarz T (2007) A multicenter study of device failure in European cochlear implant centers. Ear Hear 28(2 Suppl):95S-99S
Wei BPC, Shepherd RK, Robins-Browne RM, Clark GM, O’leary SJ (2010) Pneumococcal meningitis post-cochlear implantation: preventative measures. Otolaryngol Head Neck Surg 143(S3):S9-S14
Rubin LG, Papsin B (2010) Policy statement–cochlear implants in children: surgical site infections and prevention and treatment of acute otitis media and meningitis. Pediatrics 126(2):381–391
Acknowledgements
The authors thank Ms E. Dekkers for assistance with the data collection.
Funding
Cochlear Europe Ltd financially supported this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Theunisse, H.J., Pennings, R.J.E., Kunst, H.P.M. et al. Risk factors for complications in cochlear implant surgery. Eur Arch Otorhinolaryngol 275, 895–903 (2018). https://doi.org/10.1007/s00405-018-4901-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-018-4901-z