Abstract
Objective
To determine the effect of dopamine agonists in a surgically induced endometriosis model on rats.
Study design
In this prospective randomized experimental study, surgical induction of endometriosis was performed by autotransplantation technique on 52 adult female Wistar-Albino rats. Endometriosis formation was confirmed by a second-look laparotomy (n:48) 1 month later. Four study groups were randomly generated according to their treatment regimens: group 1 (leuprolide acetate, n = 12), group 2 (bromocriptine, n = 12), group 3 (cabergoline, n = 12) and group 4 (control, n = 12). Endometriotic implants were excised for histopathological examination after treatment at the setting of laparotomy. The mean surface areas and histopathological glandular tissue (GT) and stromal tissue (ST) scores of endometriotic implants were studied and compared among groups.
Results
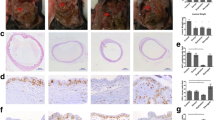
After 30 days of treatment, the mean surface area of the endometriotic implants of leuprolide acetate, bromocriptine and cabergoline groups was significantly decreased. The regression of endometriotic foci size in comparison to control was highest in group 1, followed by group 2, then group 3. In the histopathological evaluation both the ST and GT scores of group 1, 2 and 3 were significantly decreased in comparison to controls without a statistically significant difference between the groups.
Conclusion
Dopamine agonists are as effective as GnRH agonists in the regression of experimental endometriotic implants in rats. Further trials are needed to elucidate the pathways affected by dopamine agonists.




Similar content being viewed by others
References
Giudice LC, Kao LC (2004) Endometriosis. Lancet 364(9447):1789–1799
Bulun SE (2009) Endometriosis. N Engl J Med 360(3):268–279
Soares SR, Martínez-Varea A, Hidalgo-Mora JJ, Pellicer A (2012) Pharmacologic therapies in endometriosis: a systematic review. Fertil Steril 98(3):529–555
Rock JA, Zacur HA, Dlugi AM, Jones HW Jr, TeLinde RW (1982) Pregnancy success following surgical correction of imperforate hymen and complete transverse vaginal septum. Obstet Gynecol 59(4):448–451
Lucidi RS, Witz CA, Chrisco M, Binkley PA, Shain SA, Schenken RS (2005) A novel in vitro model of the early endometriotic lesion demonstrates that attachment of endometrial cells to mesothelial cells is dependent on the source of endometrial cells. Fertil Steril 84(1):16–21
Schenken RS (2012) Pathogenesis, clinical features, and diagnosis of endometriosis. http://pmtwww.uptodate.com/contents/pathogenesis-clinical-features-and-diagnosis-of-endometriosis. Accessed 23 Dec 2012
Laschke MW, Menger MD (2012) Anti-angiogenic treatment strategies for the therapy of endometriosis. Hum Reprod Update 18(6):682–702
Zhang JJ, Xu ZM, Dai HY, Ji XQ, Duan YY, Zhang CM, Qin DY (2010) Application of the nuclear factor-κB inhibitor pyrrolidine dithiocarbamate for the treatment of endometriosis: an in vitro study. Fertil Steril 94(7):2942–2944
Hull ML, Charnock-Jones DS, Chan CL, Bruner-Tran KL, Osteen KG, Tom BD, Fan TP, Smith SK (2003) Antiangiogenic agents are effective inhibitors of endometriosis. J Clin Endocrinol Metab 88(6):2889–2899
Basu S, Nagy JA, Pal S, Vasile E, Eckelhoefer IA, Bliss VS, Manseau EJ, Dasgupta PS, Dvorak HF, Mukhopadhyay D (2001) The neurotransmitter dopamine inhibits angiogenesis induced by vascular permeability factor/vascular endothelial growth factor. Nat Med 7(5):569–574
Alvarez C, Martí-Bonmatí L, Novella-Maestre E, Sanz R, Gómez R, Fernández-Sánchez M, Simón C, Pellicer A (2007) Dopamine agonist cabergoline reduces hemoconcentration and ascites in hyperstimulated women undergoing assisted reproduction. J Clin Endocrinol Metab 92(8):2931–2937
Novella-Maestre E, Carda C, Noguera I, Ruiz-Saurí A, García-Velasco JA, Simón C, Pellicer A (2009) Dopamine agonist administration causes a reduction in endometrial implants through modulation of angiogenesis in experimentally induced endometriosis. Hum Reprod 24(5):1025–1035
Vernon MW, Wilson EA (1985) Studies on the surgical induction of endometriosis in the rat. Fertil Steril 44(5):684–694
Altintas D, Kokcu A, Kandemir B, Tosun M, Cetinkaya MB (2010) Comparison of the effects of raloxifene and anastrozole on experimental endometriosis. Eur J Obstet Gynecol Reprod Biol 150(1):84–87
Witz CA (2002) Pathogenesis of endometriosis. Gynecol Obstet Invest 53(Suppl 1):52–62 (Review)
Sampson JA (1927) Peritoneal endometriosis due to menstrual dissemination of endometrial tissue into the peritoneal cavity. Am J Obstet Gynecol 14:422–469
Guo SW (2003) The best-fit model for endometriosis. Fertil Steril 80(1):232
Redwine DB (2002) Was Sampson wrong? Fertil Steril 78(4):686–693
Carmeliet P, Jain RK (2011) Molecular mechanisms and clinical applications of angiogenesis. Nature 473(7347):298–307
Oosterlynck DJ, Meuleman C, Sobis H, Vandeputte M, Koninckx PR (1993) Angiogenic activity of peritoneal fluid from women with endometriosis. Fertil Steril 59(4):778–782
Healy DL, Rogers PA, Hii L, Wingfield M (1998) Angiogenesis: a new theory for endometriosis. Hum Reprod Update 4(5):736–740
Vernon MW, Wilson EA (1985) Studies on the surgical induction of endometriosis in the rat. Fertil Steril 44(5):684–694
Laschke MW, Giebels C, Nickels RM, Scheuer C, Menger MD (2011) Endothelial progenitor cells contribute to the vascularization of endometriotic lesions. Am J Pathol 178(1):442–450
Shifren JL, Tseng JF, Zaloudek CJ, Ryan IP, Meng YG, Ferrara N, Jaffe RB, Taylor RN (1996) Ovarian steroid regulation of vascular endothelial growth factor in the human endometrium: implications for angiogenesis during the menstrual cycle and in the pathogenesis of endometriosis. J Clin Endocrinol Metab 81(8):3112–3118
Donnez J, Smoes P, Gillerot S, Casanas-Roux F, Nisolle M (1998) Vascular endothelial growth factor (VEGA) in endometriosis. Hum Reprod 13(6):1686–1690
Ceyhan ST, Onguru O, Baser I, Gunhan O (2008) Expression of cyclooxygenase-2 and vascular endothelial growth factor in ovarian endometriotic cysts and their relationship with angiogenesis. Fertil Steril 90(4):988–993
Ceyhan ST, Onguru O, Fidan U, Ide T, Yaman H, Kilic S, Baser I (2011) Comparison of aromatase inhibitor (letrozole) and immunomodulators (infliximab and etanercept) on the regression of endometriotic implants in a rat model. Eur J Obstet Gynecol Reprod Biol 154(1):100–104
Jain RK, Duda DG, Clark JW, Loeffler JS (2006) Lessons from phase III clinical trials on anti-VEGA therapy for cancer. Nat Clin Pract Oncol 3(1):24–40
Becker CM, Sampson DA, Rupnick MA, Rohan RM, Efstathiou JA, Short SM, Taylor GA, Folkman J, D’Amato RJ (2005) Endostatin inhibits the growth of endometriotic lesions but does not affect fertility. Fertil Steril 84(Suppl 2):1144–1155
Via LE, Gore-Langton RE, Pluda JM (2000) Clinical trials referral resource. Current clinical trials administering the antiangiogenesis agent SU5416. Oncology (Williston Park) 14(9):1312, 1315–6, 1321–3
Gillam MP, Molitch ME, Lombardi G, Colao A (2006) Advances in the treatment of prolactinomas. Endocr Rev 27(5):485–534
Basu S, Nagy JA, Pal S, Vasile E, Eckelhoefer IA, Bliss VS, Manseau EJ, Dasgupta PS, Dvorak HF, Mukhopadhyay D (2001) The neurotransmitter dopamine inhibits angiogenesis induced by vascular permeability factor/vascular endothelial growth factor. Nat Med 7(5):569–574
Novella-Maestre E, Carda C, Ruiz-Sauri A, Garcia-Velasco JA, Simon C, Pellicer A (2010) Identification and quantification of dopamine receptor 2 in human eutopic and ectopic endometrium: a novel molecular target for endometriosis therapy. Biol Reprod 83(5):866–873
Schade R, Andersohn F, Suissa S, Haverkamp W, Garbe E (2007) Dopamine agonists and the risk of cardiac-valve regurgitation. N Engl J Med 356(1):29–38
Delgado-Rosas F, Gómez R, Ferrero H, Gaytan F, Garcia-Velasco J, Simón C, Pellicer A (2011) The effects of ergot and non-ergot-derived dopamine agonists in an experimental mouse model of endometriosis. Reproduction 142(5):745–755
Vance ML, Evans WS, Thorner MO (1984) Drugs five years later. Bromocriptine. Ann Intern Med 100(1):78–91
Góth MI, Hubina E, Raptis S, Nagy GM, Tóth BE (2003) Physiological and pathological angiogenesis in the endocrine system. Microsc Res Tech 60(1):98–106
Gomez R, Gonzalez-Izquierdo M, Simon C, Remohi J, Pellicer A (2003) Tyroxine hydroxylase (TH) downregulation in hyperstimulated ovaries reveals he dopamine agonist bromocriptine (Br 2) as an effective and specific method to block increased vascular permeability (VP) in OHSS (Abstract O-113). Fertil Steril 80(Suppl 3):43–44
Gomez R, Gonzalez-Izquierdo M, Zimmermann RC, Novella-Maestre E, Alonso-Muriel I, Sanchez-Criado J, Remohi J, Simon C, Pellicer A (2006) Low-dose dopamine agonist administration blocks vascular endothelial growth factor (VEGA)-mediated vascular hyperpermeability without altering VEGA receptor 2-dependent luteal angiogenesis in a rat ovarian hyperstimulation model. Endocrinology 147(11):5400–5411
Brown J, Pan A, Hart RJ (2010) Gonadotrophin-releasing hormone analogues for pain associated with endometriosis. Cochrane Database Syst Rev (12):CD008475
Acknowledgments
Study was supported by GMMA Research Center with the project number AR-2012/14. No funding was received for the trial. The authors acknowledge the assistance and support of Veterinary Dr Sahin SAGALTICI and Dr Osman Tansu TOMBUS during the study.
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ercan, C.M., Kayaalp, O., Cengiz, M. et al. Comparison of efficacy of bromocriptine and cabergoline to GnRH agonist in a rat endometriosis model. Arch Gynecol Obstet 291, 1103–1111 (2015). https://doi.org/10.1007/s00404-014-3524-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-014-3524-x