Abstract
Purpose
Triple negative breast cancers (TNBC) are associated with an adverse outcome, although these tumors are sensitive to chemotherapy. In part, this phenomenon could be caused by tumor immune escape. The current study investigates immunogenicity of TNBC cells in vitro and the presence of immunosuppressive factors in the tumor microenvironment (pAKT and B7H1 expression, infiltration with regulatory T cells, [Tregs]).
Methods
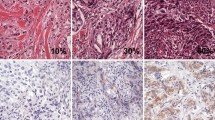
Natural killer (NK)-cell induced lysis was evaluated in estrogen receptor (ER) positive MCF 7 breast cancers, in MDA-MB231 and MDA-MB468 and in HCC-1937 (BRCA 1 mutated) and HCC-1806 TNBC cells. Expression of pAKT, B7H1 and infiltration with Tregs were determined by immunohistochemistry in human specimens of benign and malignant breast disease.
Results
NK-cell induced lysis was significantly increased (p < 0.05) in four TNBC cell lines compared to ER + MCF 7 cells. Fibroadenomas and mastectomy samples were not infiltrated with Tregs. Infiltration with Tregs was 0.92 ± 0.21 in ER/PR + breast cancers and significantly higher in TNBC without (2.30 ± 0.34) and also significantly higher with mutation of BRCA 1 (2.10 ± 0.34). Expression of pAKT was absent in benign controls and 1.23 ± 0.36 in ER/PR + breast cancers, 1.78 ± 0.40 in TNBC without and 2.40 ± 0.30 with mutated BRCA 1. No significant differences of B7H1 expression occurred among the breast cancer subgroups.
Conclusion
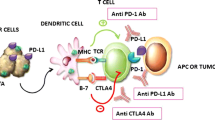
TNBC cell stimulate the NK-cell immune response significantly stronger than ER positive breast cancer cells. This could explain why infiltration with immunosuppressive Tregs is increased in human specimens of TNBC with and without mutated BRCA 1. Accordingly, immunomodulatory treatment strategies should be further explored in TNBC.


Similar content being viewed by others
References
Jemal A et al (2010) Cancer statistics. CA Cancer J Clin 60(5):277–300
Prat A, Perou CM (2010) Deconstructing the molecular portraits of breast cancer. Mol Oncol 5(1):5–23
Peppercorn J, Perou CM, Carey LA (2008) Molecular subtypes in breast cancer evaluation and management: divide and conquer. Cancer Invest 26(1):1–10
Alizadeh AA et al (2001) Towards a novel classification of human malignancies based on gene expression patterns. J Pathol 195(1):41–52
Barrett SV (2010) Breast cancer. J R Coll Physicians Edinb 40(4):335–338 quiz 339
Schneider BP et al (2008) Triple-negative breast cancer: risk factors to potential targets. Clin Cancer Res 14(24):8010–8018
Hennessy BT et al (2007) Pharmacodynamic markers of perifosine efficacy. Clin Cancer Res 13(24):7421–7431
Dent R et al (2007) Triple-negative breast cancer: clinical features and patterns of recurrence. Clin Cancer Res 13(15 Pt 1):4429–4434
Tanner B et al (2006) ErbB-3 predicts survival in ovarian cancer. J Clin Oncol 24(26):4317–4323
Rakha EA et al (2007) Prognostic markers in triple-negative breast cancer. Cancer 109(1):25–32
Haffty BG et al (2006) Locoregional relapse and distant metastasis in conservatively managed triple negative early-stage breast cancer. J Clin Oncol 24(36):5652–5657
Nahleh Z (2010) Neoadjuvant chemotherapy for “triple negative” breast cancer: a review of current practice and future outlook. Med Oncol 27(2):531–539
Olkhanud PB et al (2009) Breast cancer lung metastasis requires expression of chemokine receptor CCR4 and regulatory T cells. Cancer Res 69(14):5996–6004
Tischkowitz M et al (2007) Use of immunohistochemical markers can refine prognosis in triple negative breast cancer. BMC Cancer 7:134
Paradiso A, Formenti S (2011) Hereditary breast cancer: clinical features and risk reduction strategies. Ann Oncol 22(Suppl 1):i31–i36
Narod SA (2010) BRCA mutations in the management of breast cancer: the state of the art. Nat Rev Clin Oncol 7(12):702–707
Alfonso-De Matte MY et al (2002) Telomerase is regulated by c-Jun NH2-terminal kinase in ovarian surface epithelial cells. Cancer Res 62(16):4575–4578
Darnell RB, Posner JB (2003) Observing the invisible: successful tumor immunity in humans. Nat Immunol 4(3):201
Wolf AM et al (2003) Increase of regulatory T cells in the peripheral blood of cancer patients. Clin Cancer Res 9(2):606–612
Boshoff C, Weiss R (2002) AIDS-related malignancies. Nat Rev Cancer 2(5):373–382
Algarra I, Cabrera T, Garrido F (2000) The HLA crossroad in tumor immunology. Hum Immunol 61(1):65–73
Marincola FM et al (2000) Escape of human solid tumors from T-cell recognition: molecular mechanisms and functional significance. Adv Immunol 74:181–273
Seliger B et al (2002) HLA class I antigen abnormalities and immune escape by malignant cells. Semin Cancer Biol 12(1):3–13
Kim R et al (2006) Tumor-driven evolution of immunosuppressive networks during malignant progression. Cancer Res 66(11):5527–5536
Thomas DA, Massague J (2005) TGF-beta directly targets cytotoxic T cell functions during tumor evasion of immune surveillance. Cancer Cell 8(5):369–380
Kryczek I et al (2006) B7–H4 expression identifies a novel suppressive macrophage population in human ovarian carcinoma. J Exp Med 203(4):871–881
Kryczek I et al (2006) Cutting Edge: induction of B7–H4 on APCs through IL-10: novel suppressive mode for regulatory T Cells. J Immunol 177(1):40–44
Curiel TJ et al (2004) Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med 10(9):942–949
Ling X et al (2010) Mesenchymal Stem Cells Overexpressing IFN-beta Inhibit Breast Cancer Growth and Metastases through Stat3 Signaling in a Syngeneic Tumor Model. Cancer Microenviron 3(1):83–95
Tan W et al (2011) Tumour-infiltrating regulatory T cells stimulate mammary cancer metastasis through RANKL-RANK signalling. Nature 470(7335):548–553
Black MM, Speer FD (1956) In vitro and clinical effects of urethane plus triethylene melamine on human breast cancer. Surg Gynecol Obstet 102(4):420–426
Ladoire S et al (2011) Presence of Foxp3 expression in tumor cells predicts better survival in HER2-overexpressing breast cancer patients treated with neoadjuvant chemotherapy. Breast Cancer Res Treat 125(1):65–72
Black MM, Opler SR, Speer FD (1956) Structural representations of tumor-host relationships in gastric carcinoma. Surg Gynecol Obstet 102(5):599–603
Mahmoud SM et al (2011) An evaluation of the clinical significance of FOXP3 + infiltrating cells in human breast cancer. Breast Cancer Res Treat 127(1):99–108
Denkert C et al (2010) Tumor-associated lymphocytes as an independent predictor of response to neoadjuvant chemotherapy in breast cancer. J Clin Oncol 28(1):105–113
Ladoire S et al (2011) Presence of Foxp3 expression in tumor cells predicts better survival in HER2-overexpressing breast cancer patients treated with neoadjuvant chemotherapy. Breast Cancer Res Treat 125(1):65–72
Yi T et al (2011) Host APCs augment in vivo expansion of donor natural regulatory T cells via B7H1/B7.1 in allogeneic recipients. J Immunol 186(5):2739–2749
Wang S, Chen L (2011) Immunobiology of cancer therapies targeting CD137 and B7–H1/PD-1 cosignal pathways. Curr Top Microbiol Immunol 344:245–267
Parsa AT et al (2007) Loss of tumor suppressor PTEN function increases B7–H1 expression and immunoresistance in glioma. Nat Med 13(1):84–88
Hers I, Vincent EE, Tavare JM (2011) Akt signalling in health and disease. Cell Signal 23(10):1515–1527
Lopez-Knowles E et al (2010) PI3 K pathway activation in breast cancer is associated with the basal-like phenotype and cancer-specific mortality. Int J Cancer 126(5):1121–1131
Lehmann BD et al (2011) Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J Clin Invest 121(7):2750–2767
Righi E et al (2011) CXCL12/CXCR4 blockade induces multimodal antitumor effects that prolong survival in an immunocompetent mouse model of ovarian cancer. Cancer Res 71(16):5522–5534
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Engel, J.B., Honig, A., Kapp, M. et al. Mechanisms of tumor immune escape in triple-negative breast cancers (TNBC) with and without mutated BRCA 1. Arch Gynecol Obstet 289, 141–147 (2014). https://doi.org/10.1007/s00404-013-2922-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-013-2922-9