Abstract
Introduction
Primary arthrofibrosis is of major concern after joint trauma or knee ligament surgery. The underlying mechanism in detail remains unclear. Highly differentiated fibroblastic cells, so-called myofibroblasts, express the actin isoform α-smooth muscle actin (ASMA) and have been found to play a major role in tissue contraction during wound healing and organ fibrosis. We therefore studied the expression of myofibroblasts in human primary knee arthrofibrosis tissue.
Materials and methods
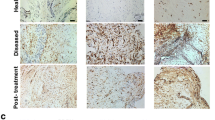
Tissue samples were taken from the infrapatellar fat pad and intercondylar region of nine patients who underwent revision surgery due to arthrofibrosis after anterior cruciate ligament (ACL) reconstruction (study group). Control tissue was taken from five patients who underwent primary ACL reconstruction (control group I) and from eight patients, who underwent second-look arthroscopy after primary ACL reconstruction (control group II). ASMA containing fibroblasts were immunostained with a monoclonal antibody. Histomorphometry was performed for total cell amount, ASMA containing fibroblasts, and vessel cross-sections.
Results
The arthrofibrosis group showed a tenfold higher amount of ASMA containing myofibroblasts (23.4% vs. 2.3%) than in control group I. There was a significantly higher total cell count and lower vessel density than in control group I. Control group II showed an upregulation of myofibroblasts almost five times that in control group I; nevertheless there was no evidence of scar formation or tissue fibrosis.
Conclusions
Myofibroblasts are responsible for scar tissue contraction during wound healing. In arthrofibrosis tissue fibroblast contraction may be involved in tissue fibrosis and contraction with consecutive loss of motion. We found that myofibroblasts are upregulated in arthrofibrosis tissue. ACL reconstruction itself caused an up regulation of myofibroblast content. Nevertheless these patients did not show any clinical or histological signs of arthrofibrosis. Thus it is reasonable to assume that the ratio of myofibroblasts and total cell amount in connective tissue are responsible for the onset of arthrofibrosis. Address the expression of this highly differentiated cell type may therefore present a target for future therapeutic interventions.






Similar content being viewed by others
References
Adler K, Craighead J, Vallyathan N, Evans J (1981) Actin-containing cell in human pulmonary fibrosis. Am J Pathol 102:427–437
Aglietti P, Buzzi R, De Felice R, Paolini G, Zaccherotti G (1995) Results of surgical treatment of arthrofibrosis after ACL reconstruction. Knee Surg Sports Traumatol Arthrosc 3:83–88
Atkinson T, Atkinson P, Mendenhall H, Haut R (1998) Patellar tendon and infrapatellar fat pad healing after harvest of an ACL graft. J Surg Res 79:25–30
Baur P, Parks D (1983) The myofibroblast anchoring strand: the fibronectin connection in wound healing and the possible loci of collagen fibril assembly. J Trauma 23:853–862
Border W, Noble N (1994) Transforming growth factor b in tissue fibrosis. N Engl J Med 331:1286–1292
Border WA, Ruoslahti E (1992) Transforming growth factor-b in disease: the dark side of tissue repair. J Clin Invest 90:1–7
Bosch U, Zeichen J, Lobenhoffer P, Albers I, van Griensven M (1999) [Arthrofibrosis—A chronic inflammatory process?]. Arthroskopie 12:117–120
Bosch U, Zeichen J, Lobenhoffer P, Skutek M, van Griensven M (1999) [Etiology of arthrofibrosis]. Arthroskopie 12:215–221
Bosch U, Zeichen J, Skutek M, Haeder L, van Griensven M (2001) Arthrofibrosis is the result of a T cell mediated immune response. Knee Surg Sports Traumatol Arthrosc 9:282–289
Cosgarea A, Dehaven K, Lovelock J (1994) The surgical treatment of arthrofibrosis of the knee. Am J Sports Med 22:184–191
Darby I, Skalli O, Gabbiani G (1990) Alpha-smooth muscle actin is transiently expressed by myofibroblasts during experimental wound healing. Lab Invest 63:21–29
Desmouliere A, Geinoz A, Gabbiani F, Gabbiani G (1993) Transforming growth factor-b1 induces a-smooth muscle actin expression in granulation tissue myofibroblasts and in quiescent and growing cultured fibroblasts. J Cell Biol 122:103–111
Eddy R, Petro J, Tomasek J (1988) Evidence of the nonmuscle nature of the “Myofibroblast” of granulation tissue and hypertrophic scar. Am J Pathol 130:252–260
Estes J, Vande Berg J, Adzick N, McGillivray T, Desmouliere A, Gabbiani G (1994) Phenotypic and functional features of myofibroblasts in sheep fetal wounds. Differentiation 56:173–181
Faryniarz DA, Chaponnier C, Gabbiani G, Yannas IV, Spector M (1996) Myofibroblasts in the healing lapine medial collateral ligament: possible mechanisms of contraction. J Orthop Res 14:228–237
Fukushima K, Badlani N, Usas A, Riano F, Fu F, Huard J (2001) The use of an antifibrosis agent to improve muscle recovery after laceration. Am J Sports Med 29:394–402
Gabbiani G, Majno G (1972) Dupuytren’s contracture: fibroblast contraction? An ultrastructural study. Am J Pathol 66:131–146
Gabbiani G, Hirschel BJ, Ryan GB, Statkov PR, Majno G (1972) Granulation tissue as a contractile organ. A study of structure and function. J Exp Med 135:719–734
Gögüs A, Lobenhoffer P (1993) [Arthroscopic therapy of arthrofibrosis of the knee joint]. Unfallchirurg 96:100–108
Harner C, Irrgang J, Paul J, Dearwater S, Fu F (1992) Loss of motion after anterior cruciate ligament reconstruction. Am J Sports Med 20:499–506
Harris A, Stopak D, Wild P (1981) Fibroblast traction as a mechanism for collagen morphogenesis. Nature 290:249–251
Lobenhoffer H, Bosch U, Gerich T (1996) Role of posterior capulotomy for the treatment of extension deficits of the knee. Knee Surg Sports Traumatol Arthrosc 4:237–241
Mariani P, Santori N, Rovere P, Della Rocca C, Adriani E (1997) Histological and structural study of the adhesive tissue in knee fibroarthrosis: a clinical-pathological correlation. Arthroscopy 13:313–318
Martin P (1997) Wound healing: aiming for perfect skin regeneration. Science 276:75–86
Murakami S, Muneta T, Ezura Y, Furuya K, Yamamoto H (1997) Quantitative analysis of synovial fibrosis in the infrapatellar fat pad before and after ACL reconstruction. Am J Sports Med 25:29–34
Nagle RB, Kneiser MR, Bulger RE, Benditt EP (1973) Induction of smooth muscle characteristics in renal interstitial fibroblasts during obstructive nephropathy. Lab Invest 29:422–427
Oda D, Gown A, Vande Berg J, Stern R (1988) The fibroblast-like nature of myofibroblasts. Exp Mol Pathol 49:316–329
Parisien J (1988) The role of arthroscopy in the treatment of postoperative fibroarthrosis of the knee joint. Clin Orthop 229:185–192
Paulos L, Rosenberg T, Drawbert, Manning J, Abbott P (1987) Infrapatellar contracture syndrome: an unrecognized cause of knee stiffness with patella entrapment and patella infera. Am J Sports Med 15:331–341
Paulos L, Wnorowski D, Greenwald A (1994) Infrapatellar contracture syndrome: diagnosis, treatment and longterm follow-up. Am J Sports Med 22:440–449
Ronnov-Jessen L, Petersen OW (1993) Induction of a-smooth muscle actin by transforming growth factor-b1 in quiescent human breast gland fibroblasts. Implications for myofibroblast generation in breast neoplasis. Lab Invest 68:696–707
Rudolph R, McLure W, Woodward (1979) Contractile fibroblasts in chronic alcoholic cirrhosis. Gastroenterology 76:704–709
Sappino A, Schürch W, Gabbiani G (1990) Biology of disease—differentiation repertoire of fibroblastic cells: expression of cytoskeletal proteins as markers of phenotypic modulations. Lab Invest 63:144–161
Schürch W, Seemeyer T, Gabbiani G (1998) The myofibroblast: a quarter century after its discovery. Am J Surg Pathol 22:141–147
Serini G, Gabbiani G (1999) Mechanisms of myofibroblast activity and phenotypic modulation. Exp Cell Res 250:273–283
Shelbourne K, Trumper R (1997) Preventing anterior knee pain after anterior cruciate ligament reconstruction. Am J Sports Med 25:41–47
Shelbourne KD, Wilckens JH, Mollabashy A, DeCarlo M (1991) Arthrofibrosis in acute anterior cruciate ligament reconstruction. The effect of timing of reconstruction and rehabilitation. Am J Sports Med 19:332–336
Shelbourne KD, Patel DV (1999) Treatment of limited motion after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 7:85–92
Skalli O, Pelte M, Peclet M, Gabbiani G, Gugliotta P, Bussolati, Ravazzola M, Orci L (1989) Alpha-smooth muscle actin, a differentiation marker of smooth muscle cells, is present in microfilamentous bundles of pericytes. J Histochem Cytochem 37:315–321
Skalli O, Ropraz P, Trzeciak, Benzonana G, Gillessen D, Gabbiani G (1986) A monoclonal antibody against alpha-smooth muscle actin: a new probe for smooth muscle differentiation. J Cell Biol 103:2787–2796
Skalli O, Schürch W, Seemeyer T, Lagace R, Montandon, Pittet B, Gabbiani G (1989) Myofibroblasts from diverse pathologic settings are heterogeneous in their content of actin isoforms and intermediate filament proteins. Lab Invest 60:275–285
Sporn MB, Roberts A (1992) Transforming growth factor-β: recent progress and new challenges. J Cell Biol 119:1017–1021
Stopak D, Harris A (1982) Connective tissue morphogenesis by fibroblast traction. I. Tissue culture observations. Dev Biol 90:383–398
Weiler A, Unterhauser FN, Bail HJ, Huning M, Haas NP (2002) Alpha-smooth muscle actin is expressed by fibroblastic cells of the ovine anterior cruciate ligament and its free tendon graft during remodeling. J Orthop Res 20:310–317
Zeichen J, van Griensven M, Albers I, Lobenhoffer P, Bosch U (1999) Immunohistochemical localization of collagen VI in arthrofibrosis. Arch Orthop Trauma Surg 119:315–318
Zhang K, Rekhter M, Gordon D, Phan S (1994) Myofibroblasts and their role in lung collagen gene expression during pulmonary fibrosis. A combined immunohistochemical and in situ hybridization study. Am J Pathol 145:114–125
Author information
Authors and Affiliations
Corresponding author
Additional information
Winner of the AGA-DonJoy Award 2003
Rights and permissions
About this article
Cite this article
Unterhauser, F.N., Bosch, U., Zeichen, J. et al. α-Smooth muscle actin containing contractile fibroblastic cells in human knee arthrofibrosis tissue. Arch Orthop Trauma Surg 124, 585–591 (2004). https://doi.org/10.1007/s00402-004-0742-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-004-0742-x