Abstract
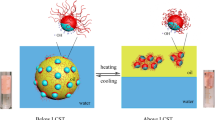
This work presents a unique desymmetrization method to produce a high quantity of Janus nanoparticles (JNPs) using dual-phase oil (melted wax)/water system. The four-stage process includes fixing, primary modification, releasing, and secondary modification. Unlike other works, dispersed hydrophilic nanoparticles were forced to be placed at oil/water interface using an upward water stream formed by buoyancy effects due to the applied heat to the bottom of the container. This eliminated some significant deficiencies of common desymmetrization processes which apply oil (melted wax)/water Pickering emulsion systems, e.g., the coalescence of melted /solidified wax droplets and high cost. The best effect of heat-driven buoyancy flow was ensured using saturation theory. In order to induce asymmetrical surface properties to the applied nanoparticles, (3-aminopropyl)triethoxysilane and hexadecyltrimethoxysilane were used in primary and secondary modification stages, respectively. Produced JNPs were characterized using FTIR, thermogravimetric analysis (TGA), and energy-dispersive X-ray spectroscopy (EDX) tests in order to confirm the attachment of modifier molecules on the surface of the applied nanoparticles. Furthermore, TGA results were used to calculate three-phase contact angle (β) as an important parameter dictating the asymmetric properties of Janus nanoparticles. Also, a dichloromethane/water mixture was used to demonstrate the differences of Janus nanoparticles with similar uniformly modified nanoparticles. Furthermore, using polystyrene/poly(methyl methacrylate) blend, it was shown that produced Janus nanoparticles tend to be placed at the interface while their corresponding uniformly modified nanoparticles stay in polystyrene or poly(methyl methacrylate) phase.













Similar content being viewed by others
References
Rotello V (2012) Nanoparticles: building blocks for nanotechnology. Springer, US
Kango S, Kalia S, Celli A, Njuguna J, Habibi Y, Kumar R (2013) Surface modification of inorganic nanoparticles for development of organic–inorganic nanocomposites—a review. Prog Polym Sci 38(8):1232–1261. doi:10.1016/j.progpolymsci.2013.02.003
Mahdavi M, Ahmad M, Haron M, Namvar F, Nadi B, Rahman M, Amin J (2013) Synthesis, surface modification and characterisation of biocompatible magnetic iron oxide nanoparticles for biomedical applications. Molecules 18(7):7533. doi:10.3390/molecules18077533
Caruso F (2001) Nanoengineering of particle surfaces. Adv Mater 13(1):11–22. doi:10.1002/1521-4095(200101)13:1<11::AID-ADMA11>3.0.CO;2-N
Hong L, Cacciuto A, Luijten E, Granick S (2006) Clusters of charged Janus spheres. Nano Lett 6(11):2510–2514. doi:10.1021/nl061857i
Iacovella CR, Horsch MA, Zhang Z, Glotzer SC (2005) Phase diagrams of self-assembled mono-tethered nanospheres from molecular simulation and comparison to surfactants. Langmuir 21(21):9488–9494. doi:10.1021/la051035l
Lv W, Lee KJ, Li J, Park T-H, Hwang S, Hart AJ, Zhang F, Lahann J (2012) Anisotropic Janus catalysts for spatially controlled chemical reactions. Small 8(20):3116–3122. doi:10.1002/smll.201200192
Dendukuri D, Doyle PS (2009) The synthesis and assembly of polymeric microparticles using microfluidics. Adv Mater 21(41):4071–4086. doi:10.1002/adma.200803386
Shah RK, Kim J-W, Weitz DA (2009) Janus supraparticles by induced phase separation of nanoparticles in droplets. Adv Mater 21(19):1949–1953. doi:10.1002/adma.200803115
Perro A, Meunier F, Schmitt V, Ravaine S (2009) Production of large quantities of “Janus” nanoparticles using wax-in-water emulsions. Colloids Surf 332(1):57–62. doi:10.1016/j.colsurfa.2008.08.027
Pickering SU (1907) CXCVI.—emulsions. J Chem Soc Trans 91(0):2001–2021. doi:10.1039/CT9079102001
Walther A, Müller AHE (2013) Janus particles: synthesis, self-assembly, physical properties, and applications. Chem Rev 113(7):5194–5261. doi:10.1021/cr300089t
Pieranski P (1980) Two-dimensional interfacial colloidal crystals. Phys Rev Lett 45(7):569–572. doi:10.1103/PhysRevLett.45.569
Jiang S, Granick S (2008) Controlling the geometry (Janus balance) of amphiphilic colloidal particles. Langmuir 24(6):2438–2445. doi:10.1021/la703274a
Giermanska-Kahn J, Laine V, Arditty S, Schmitt V, Leal-Calderon F (2005) Particle-stabilized emulsions comprised of solid droplets. Langmuir 21(10):4316–4323. doi:10.1021/la0501177
Hong L, Jiang S, Granick S (2006) Simple method to produce Janus colloidal particles in large quantity. Langmuir 22(23):9495–9499. doi:10.1021/la062716z
Hemmer E, Quintanilla M, Légaré F, Vetrone F (2015) Temperature-induced energy transfer in dye-conjugated Upconverting nanoparticles: a new candidate for Nanothermometry. Chem Mater 27(1):235–244. doi:10.1021/cm503799f
Nasrin R, Alim MA, Chamkha AJ (2012) Buoyancy-driven heat transfer of water–Al2O3 nanofluid in a closed chamber: effects of solid volume fraction, Prandtl number and aspect ratio. Int J Heat Mass Transf 55(25–26):7355–7365. doi:10.1016/j.ijheatmasstransfer.2012.08.011
Adam NK (1952) The physics and chemistry of surfaces. Oxford University Press, Oxford
Maghsoud Z, Navid Famili MH, Madaeni SS (2010) Phase diagram calculations of water/tetrahydrofurane/poly (vinyl chloride) ternary system based on a compressible regular solution model. Iran Polym J 19(8):581–588
Barzin J, Sadatnia B (2007) Theoretical phase diagram calculation and membrane morphology evaluation for water/solvent/polyethersulfone systems. Polymer 48(6):1620–1631. doi:10.1016/j.polymer.2007.01.049
Amirshaqaqi N, Salami-Kalajahi M, Mahdavian M (2014) Investigation of corrosion behavior of aluminum flakes coated by polymeric nanolayer: effect of polymer type. Corros Sci 87:392–396. doi:10.1016/j.corsci.2014.06.045
Lambert JB (1987) Introduction to organic spectroscopy. Macmillan, London
Xu L, Wang L, Shen Y, Ding Y, Cai Z (2015) Preparation of hexadecyltrimethoxysilane-modified silica nanocomposite hydrosol and superhydrophobic cotton coating. Fibers and Polymers 16(5):1082–1091. doi:10.1007/s12221-015-1082-x
Samadaei F, Salami-Kalajahi M, Roghani-Mamaqani H, Banaei M (2015) A structural study on ethylenediamine- and poly (amidoamine)-functionalized graphene oxide: simultaneous reduction, functionalization, and formation of 3D structure. RSC Adv 5(88):71835–71843. doi:10.1039/C5RA12086A
Amirshaqaqi N, Salami-Kalajahi M, Mahdavian M (2014) Corrosion behavior of aluminum/silica/polystyrene nanostructured hybrid flakes. Iran Polym J 23(9):699–706. doi:10.1007/s13726-014-0264-5
Panahian P, Salami-Kalajahi M, Salami Hosseini M (2014) Synthesis of dual Thermosensitive and pH-sensitive hollow Nanospheres based on poly (acrylic acid-b-2-hydroxyethyl methacrylate) via an atom transfer reversible addition–fragmentation radical process. Ind Eng Chem Res 53(19):8079–8086. doi:10.1021/ie500892b
Maity D, Chandrasekharan P, Feng S-S, Jun D (2010) Synthesis and studies of APTES functionalized magnetite nanoparticles. In: Nanoscience and Nanotechnology (ICONN), 2010 International Conference on, . IEEE, pp 94–97
Paopattra T, Sarintorn L, Kawee S (2012) Preparation of organosilane treated microcrystalline (SiMCC) and SiMCC/polypropylene composites. Journal of Metals, Materials and Minerals 22:13–19
Zhuravlev LT (2000) The surface chemistry of amorphous silica. Zhuravlev model. Colloids Surf 173(1–3):1–38. doi:10.1016/S0927-7757(00)00556-2
Shioji S, Kawaguchi M, Hayashi Y, Tokami K, Yamamoto H (2001) Rehydroxylation of dehydrated silica surfaces by water vapor adsorption. Adv Powder Technol 12(3):331–342. doi:10.1163/156855201750537884
Acres RG, Ellis AV, Alvino J, Lenahan CE, Khodakov DA, Metha GF, Andersson GG (2012) Molecular structure of 3-Aminopropyltriethoxysilane layers formed on silanol-terminated silicon surfaces. J Phys Chem C 116(10):6289–6297. doi:10.1021/jp212056s
Liu Y, Li Y, Li X-M, He T (2013) Kinetics of (3-aminopropyl)triethoxylsilane (APTES) silanization of superparamagnetic iron oxide nanoparticles. Langmuir 29(49):15275–15282. doi:10.1021/la403269u
Bhuiyan MHU, Saidur R, Amalina MA, Mostafizur RM, Islam A (2015) Effect of nanoparticles concentration and their sizes on surface tension of nanofluids. Procedia Engineering 105:431–437. doi:10.1016/j.proeng.2015.05.030
Sharifzadeh E, Salami-Kalajahi M, Hosseini MS, Aghjeh MKR (2016) A temperature-controlled method to produce Janus nanoparticles using high internal interface systems: experimental and theoretical approaches. Colloids Surf 506:56–62. doi:10.1016/j.colsurfa.2016.06.006
Vansant EF, Van Der Voort P, Vrancken KC (1995) Characterization and chemical modification of the silica surface. Elsevier Science, New York
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was not funded by any institution.
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(DOCX 638 kb)
Rights and permissions
About this article
Cite this article
Sharifzadeh, E., Salami-Kalajahi, M., Hosseini, M.S. et al. Synthesis of silica Janus nanoparticles by buoyancy effect-induced desymmetrization process and their placement at the PS/PMMA interface. Colloid Polym Sci 295, 25–36 (2017). https://doi.org/10.1007/s00396-016-3977-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-016-3977-5