Abstract
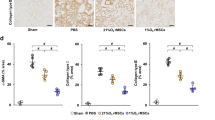
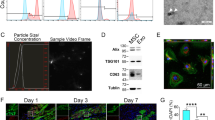
Microvascular obstruction (MVO) after primary percutaneous coronary intervention (pPCI) is identified as an independent risk factor for poor prognosis in patients with acute myocardial infarction (AMI). The inflammatory response induced by ischemia and reperfusion (I/R) injury is considered one of the main mechanisms of MVO. Mesenchymal stem cells (MSCs) are a unique stromal cell type that confers an immunomodulatory effect in cardiac disease. The present study aimed to investigate whether immediate intravenous delivery of MSCs could be used as a potential therapeutic method to attenuate MVO formation. A cardiac catheterization-induced porcine model of myocardial I/R injury was established, and allograft MSCs were immediately delivered intravenously. Cardiac magnetic resonance (CMR) imaging was performed on days 2 and 7 after the operation to determine the infarct area, MVO, and cardiac function. The pigs with allograft MSCs showed decreased MVO and infarct size, as well as an improved left ventricular ejection fraction (LVEF). Histological analysis revealed decreased myocyte area, fibrosis, and inflammatory cell infiltration in the peri-infarct zone of pigs with allograft MSCs. Moreover, the concentrations of interleukin-1β (IL-1β), interleukin-6 (IL-6) and C-reactive protein (CRP) in the serum were reduced in the allograft MSC group compared to the control group. Flow cytometry indicated decreased natural killer (NK) cells in the peripheral blood and ischemic heart tissue in the pigs with allograft MSCs. In summary, allograft MSCs delivered intravenously and immediately after myocardial I/R injury can attenuate MVO formation in a porcine model through a decline in the number of NK cells in the myocardium.





Similar content being viewed by others
References
Afzal MR, Samanta A, Shah ZI, Jeevanantham V, Abdel-Latif A, Zuba-Surma EK, Dawn B (2015) Adult bone marrow cell therapy for ischemic heart disease: evidence and insights from randomized controlled trials. Circ Res 117:558–575. https://doi.org/10.1161/CIRCRESAHA.114.304792
Ansboro S, Roelofs AJ, De Bari C (2017) Mesenchymal stem cells for the management of rheumatoid arthritis: immune modulation, repair or both? Curr Opin Rheumatol 29:201–207. https://doi.org/10.1097/BOR.0000000000000370
Barbash IM, Chouraqui P, Baron J, Feinberg MS, Etzion S, Tessone A, Miller L, Guetta E, Zipori D, Kedes LH, Kloner RA, Leor J (2003) Systemic delivery of bone marrow-derived mesenchymal stem cells to the infarcted myocardium: feasibility, cell migration, and body distribution. Circulation 108:863–868. https://doi.org/10.1161/01.CIR.0000084828.50310.6A
Bekkers SC, Yazdani SK, Virmani R, Waltenberger J (2010) Microvascular obstruction: underlying pathophysiology and clinical diagnosis. J Am Coll Cardiol 55:1649–1660. https://doi.org/10.1016/j.jacc.2009.12.037
Carrick D, Haig C, Ahmed N, McEntegart M, Petrie MC, Eteiba H, Hood S, Watkins S, Lindsay MM, Davie A, Mahrous A, Mordi I, Rauhalammi S, Sattar N, Welsh P, Radjenovic A, Ford I, Oldroyd KG, Berry C (2016) Myocardial hemorrhage after acute reperfused ST-segment-elevation myocardial infarction: relation to microvascular obstruction and prognostic significance. Circ Cardiovasc Imaging 9:e004148. https://doi.org/10.1161/CIRCIMAGING.115.004148
Chullikana A, Majumdar AS, Gottipamula S, Krishnamurthy S, Kumar AS, Prakash VS, Gupta PK (2015) Randomized, double-blind, phase I/II study of intravenous allogeneic mesenchymal stromal cells in acute myocardial infarction. Cytotherapy 17:250–261. https://doi.org/10.1016/j.jcyt.2014.10.009
De Maria GL, Alkhalil M, Wolfrum M, Fahrni G, Borlotti A, Gaughran L, Dawkins S, Langrish JP, Lucking AJ, Choudhury RP, Porto I, Crea F, Dall'Armellina E, Channon KM, Kharbanda RK, Banning AP (2019) Index of microcirculatory resistance as a tool to characterize microvascular obstruction and to predict infarct size regression in patients with STEMI undergoing primary PCI. JACC Cardiovasc Imaging 12:837–848. https://doi.org/10.1016/j.jcmg.2018.02.018
Deftereos S, Giannopoulos G, Angelidis C, Alexopoulos N, Filippatos G, Papoutsidakis N, Sianos G, Goudevenos J, Alexopoulos D, Pyrgakis V, Cleman MW, Manolis AS, Tousoulis D, Lekakis J (2015) Anti-inflammatory treatment with colchicine in acute myocardial infarction: a pilot study. Circulation 132:1395–1403. https://doi.org/10.1161/CIRCULATIONAHA.115.017611
Di Nicola M, Carlo-Stella C, Magni M, Milanesi M, Longoni PD, Matteucci P, Grisanti S, Gianni AM (2002) Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 99:3838–3843. https://doi.org/10.1182/blood.v99.10.3838
Gyongyosi M, Giurgea GA, Syeda B, Charwat S, Marzluf B, Mascherbauer J, Jakab A, Zimba A, Sarkozy M, Pavo N, Sochor H, Graf S, Lang I, Maurer G, Bergler-Klein J, investigators M (2016) Long-term outcome of combined (percutaneous intramyocardial and intracoronary) application of autologous bone marrow mononuclear cells post myocardial infarction: the 5-year MYSTAR study. PLoS ONE 11:e0164908. https://doi.org/10.1371/journal.pone.0164908
Halkos ME, Zhao ZQ, Kerendi F, Wang NP, Jiang R, Schmarkey LS, Martin BJ, Quyyumi AA, Few WL, Kin H, Guyton RA, Vinten-Johansen J (2008) Intravenous infusion of mesenchymal stem cells enhances regional perfusion and improves ventricular function in a porcine model of myocardial infarction. Basic Res Cardiol 103:525–536. https://doi.org/10.1007/s00395-008-0741-0
Hamirani YS, Wong A, Kramer CM, Salerno M (2014) Effect of microvascular obstruction and intramyocardial hemorrhage by CMR on LV remodeling and outcomes after myocardial infarction: a systematic review and meta-analysis. JACC Cardiovasc Imaging 7:940–952. https://doi.org/10.1016/j.jcmg.2014.06.012
Hare JM, Traverse JH, Henry TD, Dib N, Strumpf RK, Schulman SP, Gerstenblith G, DeMaria AN, Denktas AE, Gammon RS, Hermiller JB Jr, Reisman MA, Schaer GL, Sherman W (2009) A randomized, double-blind, placebo-controlled, dose-escalation study of intravenous adult human mesenchymal stem cells (prochymal) after acute myocardial infarction. J Am Coll Cardiol 54:2277–2286. https://doi.org/10.1016/j.jacc.2009.06.055
Hashmi S, Ahmed M, Murad MH, Litzow MR, Adams RH, Ball LM, Prasad VK, Kebriaei P, Ringden O (2016) Survival after mesenchymal stromal cell therapy in steroid-refractory acute graft-versus-host disease: systematic review and meta-analysis. Lancet Haematol 3:e45–52. https://doi.org/10.1016/S2352-3026(15)00224-0
Heldman AW, DiFede DL, Fishman JE, Zambrano JP, Trachtenberg BH, Karantalis V, Mushtaq M, Williams AR, Suncion VY, McNiece IK, Ghersin E, Soto V, Lopera G, Miki R, Willens H, Hendel R, Mitrani R, Pattany P, Feigenbaum G, Oskouei B, Byrnes J, Lowery MH, Sierra J, Pujol MV, Delgado C, Gonzalez PJ, Rodriguez JE, Bagno LL, Rouy D, Altman P, Foo CW, da Silva J, Anderson E, Schwarz R, Mendizabal A, Hare JM (2014) Transendocardial mesenchymal stem cells and mononuclear bone marrow cells for ischemic cardiomyopathy: the TAC-HFT randomized trial. JAMA 311:62–73. https://doi.org/10.1001/jama.2013.282909
Heusch G (2016) The coronary circulation as a target of cardioprotection. Circ Res 118:1643–1658. https://doi.org/10.1161/CIRCRESAHA.116.308640
Heusch G (2019) Coronary microvascular obstruction: the new frontier in cardioprotection. Basic Res Cardiol 114:45. https://doi.org/10.1007/s00395-019-0756-8
Heusch G, Skyschally A, Kleinbongard P (2018) Coronary microembolization and microvascular dysfunction. Int J Cardiol 258:17–23. https://doi.org/10.1016/j.ijcard.2018.02.010
Ibanez B, Aletras AH, Arai AE, Arheden H, Bax J, Berry C, Bucciarelli-Ducci C, Croisille P, Dall'Armellina E, Dharmakumar R, Eitel I, Fernandez-Jimenez R, Friedrich MG, Garcia-Dorado D, Hausenloy DJ, Kim RJ, Kozerke S, Kramer CM, Salerno M, Sanchez-Gonzalez J, Sanz J, Fuster V (2019) Cardiac MRI endpoints in myocardial infarction experimental and clinical trials: JACC scientific expert panel. J Am Coll Cardiol 74:238–256. https://doi.org/10.1016/j.jacc.2019.05.024
Kaibuchi N, Iwata T, Onizuka S, Yano K, Yamato M, Okano T, Ando T (2017) Cytological character of mini pig mesenchymal stromal cells from various tissues and the attempt of cell sheet formation. Regener Ther 6:83–89. https://doi.org/10.1016/j.reth.2017.02.001
Knorr M, Munzel T, Wenzel P (2014) Interplay of NK cells and monocytes in vascular inflammation and myocardial infarction. Front Physiol 5:295. https://doi.org/10.3389/fphys.2014.00295
Leuschner F, Dutta P, Gorbatov R, Novobrantseva TI, Donahoe JS, Courties G, Lee KM, Kim JI, Markmann JF, Marinelli B, Panizzi P, Lee WW, Iwamoto Y, Milstein S, Epstein-Barash H, Cantley W, Wong J, Cortez-Retamozo V, Newton A, Love K, Libby P, Pittet MJ, Swirski FK, Koteliansky V, Langer R, Weissleder R, Anderson DG, Nahrendorf M (2011) Therapeutic siRNA silencing in inflammatory monocytes in mice. Nat Biotechnol 29:1005–1010. https://doi.org/10.1038/nbt.1989
Liu W, Xie Y, Gao T, Huang F, Wang L, Ding L, Wang W, Liu S, Dai J, Wang B (2018) Reflection and observation: cell-based screening failing to detect HBV in HUMSCs derived from HBV-infected mothers underscores the importance of more stringent donor eligibility to reduce risk of transmission of infectious diseases for stem cell-based medical products. Stem cell Res Ther 9:177. https://doi.org/10.1186/s13287-018-0920-3
Luger D, Lipinski MJ, Westman PC, Glover DK, Dimastromatteo J, Frias JC, Albelda MT, Sikora S, Kharazi A, Vertelov G, Waksman R, Epstein SE (2017) Intravenously delivered mesenchymal stem cells: systemic anti-inflammatory effects improve left ventricular dysfunction in acute myocardial infarction and ischemic cardiomyopathy. Circ Res 120:1598–1613. https://doi.org/10.1161/CIRCRESAHA.117.310599
Majmudar MD, Keliher EJ, Heidt T, Leuschner F, Truelove J, Sena BF, Gorbatov R, Iwamoto Y, Dutta P, Wojtkiewicz G, Courties G, Sebas M, Borodovsky A, Fitzgerald K, Nolte MW, Dickneite G, Chen JW, Anderson DG, Swirski FK, Weissleder R, Nahrendorf M (2013) Monocyte-directed RNAi targeting CCR2 improves infarct healing in atherosclerosis-prone mice. Circulation 127:2038–2046. https://doi.org/10.1161/CIRCULATIONAHA.112.000116
Mayr A, Klug G, Schocke M, Trieb T, Mair J, Pedarnig K, Pachinger O, Jaschke W, Metzler B (2012) Late microvascular obstruction after acute myocardial infarction: relation with cardiac and inflammatory markers. Int J Cardiol 157:391–396. https://doi.org/10.1016/j.ijcard.2010.12.090
Mu D, Zhang XL, Xie J, Yuan HH, Wang K, Huang W, Li GN, Lu JR, Mao LJ, Wang L, Cheng L, Mai XL, Yang J, Tian CS, Kang LN, Gu R, Zhu B, Xu B (2016) Intracoronary transplantation of mesenchymal stem cells with overexpressed integrin-linked kinase improves cardiac function in porcine myocardial infarction. Sci Rep 6:19155. https://doi.org/10.1038/srep19155
Niccoli G, Montone RA, Ibanez B, Thiele H, Crea F, Heusch G, Bulluck H, Hausenloy DJ, Berry C, Stiermaier T, Camici PG, Eitel I (2019) Optimized treatment of ST-elevation myocardial infarction. Circ Res 125:245–258. https://doi.org/10.1161/CIRCRESAHA.119.315344
Prabhu SD, Frangogiannis NG (2016) The biological basis for cardiac repair after myocardial infarction: from inflammation to fibrosis. Circ Res 119:91–112. https://doi.org/10.1161/CIRCRESAHA.116.303577
Shammaa R, El-Kadiry AE, Abusarah J, Rafei M (2020) Mesenchymal Stem Cells Beyond Regenerative Medicine. Front Cell Dev Biol 8:72. https://doi.org/10.3389/fcell.2020.00072
Skyschally A, Haude M, Dorge H, Thielmann M, Duschin A, van de Sand A, Konietzka I, Buchert A, Aker S, Massoudy P, Schulz R, Erbel R, Heusch G (2004) Glucocorticoid treatment prevents progressive myocardial dysfunction resulting from experimental coronary microembolization. Circulation 109:2337–2342. https://doi.org/10.1161/01.CIR.0000127961.66744.F4
Symons R, Pontone G, Schwitter J, Francone M, Iglesias JF, Barison A, Zalewski J, de Luca L, Degrauwe S, Claus P, Guglielmo M, Nessler J, Carbone I, Ferro G, Durak M, Magistrelli P, Lo Presti A, Aquaro GD, Eeckhout E, Roguelov C, Andreini D, Vogt P, Guaricci AI, Mushtaq S, Lorenzoni V, Muller O, Desmet W, Agati L, Janssens S, Bogaert J, Masci PG (2018) Long-term incremental prognostic value of cardiovascular magnetic resonance after st-segment elevation myocardial infarction: a study of the collaborative registry on CMR in STEMI. JACC Cardiovasc Imag 11:813–825. https://doi.org/10.1016/j.jcmg.2017.05.023
Tardif JC, Kouz S, Waters DD, Bertrand OF, Diaz R, Maggioni AP, Pinto FJ, Ibrahim R, Gamra H, Kiwan GS, Berry C, Lopez-Sendon J, Ostadal P, Koenig W, Angoulvant D, Gregoire JC, Lavoie MA, Dube MP, Rhainds D, Provencher M, Blondeau L, Orfanos A, L'Allier PL, Guertin MC, Roubille F (2019) Efficacy and safety of low-dose colchicine after myocardial infarction. N Engl J Med 381:2497–2505. https://doi.org/10.1056/NEJMoa1912388
Traverse JH, Swingen CM, Henry TD, Fox J, Wang YL, Chavez IJ, Lips DL, Lesser JR, Pedersen WR, Burke NM, Pai A, Lindberg JL, Garberich RF (2019) NHLBI-sponsored randomized trial of postconditioning during primary percutaneous coronary intervention for ST-elevation myocardial infarction. Circ Res 124:769–778. https://doi.org/10.1161/CIRCRESAHA.118.314060
van den Akker F, de Jager SC, Sluijter JP (2013) Mesenchymal stem cell therapy for cardiac inflammation: immunomodulatory properties and the influence of toll-like receptors. Mediat Inflamm 2013:181020. https://doi.org/10.1155/2013/181020
van den Akker F, Deddens JC, Doevendans PA, Sluijter JP (2013) Cardiac stem cell therapy to modulate inflammation upon myocardial infarction. Biochem Biophys Acta 1830:2449–2458. https://doi.org/10.1016/j.bbagen.2012.08.026
Wang D, Li J, Zhang Y, Zhang M, Chen J, Li X, Hu X, Jiang S, Shi S, Sun L (2014) Umbilical cord mesenchymal stem cell transplantation in active and refractory systemic lupus erythematosus: a multicenter clinical study. Arthritis Res Ther 16:R79. https://doi.org/10.1186/ar4520
Zhao J, Li X, Hu J, Chen F, Qiao S, Sun X, Gao L, Xie J, Xu B (2019) Mesenchymal stromal cell-derived exosomes attenuate myocardial ischaemia-reperfusion injury through miR-182-regulated macrophage polarization. Cardiovasc Res 115:1205–1216. https://doi.org/10.1093/cvr/cvz040
Zheng G, Huang R, Qiu G, Ge M, Wang J, Shu Q, Xu J (2018) Mesenchymal stromal cell-derived extracellular vesicles: regenerative and immunomodulatory effects and potential applications in sepsis. Cell Tissue Res 374:1–15. https://doi.org/10.1007/s00441-018-2871-5
Acknowledgements
This work was supported by grants from the Natural Science Foundation of China (81870358), the Funds for Jiangsu Provincial Key Medical Discipline (ZDXKB2016013), the Key Projects of Science and Technology of Jiangsu Province (BE2016607 and BE2019602), the Funds for Jiangsu Provincial Medical Youth Talent (QNRC2016033), and the Programs of the Science Foundation in Nanjing (ZKX17011 and YKK17095).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The author(s) declare that they have no conflicts of interest.
Ethical approval
The manuscript does not contain clinical studies or patient data.
Electronic supplementary material
Below is the link to the electronic supplementary material.


Rights and permissions
About this article
Cite this article
Wang, J., Chen, Z., Dai, Q. et al. Intravenously delivered mesenchymal stem cells prevent microvascular obstruction formation after myocardial ischemia/reperfusion injury. Basic Res Cardiol 115, 40 (2020). https://doi.org/10.1007/s00395-020-0800-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-020-0800-8