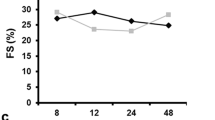
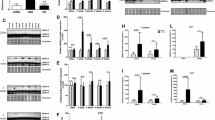
Abstract
Ventricular remodeling following myocardial infarction (MI) includes myocardial hypertrophy, a process requiring increased protein synthesis and sarcomere assembly. The anti-hypertrophic effect of MuRF1/MafBx, both muscle-specific E3-ubiquitin ligases, has been demonstrated in animal experiments and in cultured cardiomyocytes. We assessed MuRF1/MAFbx expression in myocardium remote of recently (<2 weeks) infarcted regions (MI), compared with patients undergoing coronary artery bypass surgery, with normal systolic function and without previous infarction (control or Con). Left ventricular myocardial biopsies were obtained from the contralateral normal zone in MI (n = 14) patients and from the Con (n = 12) group. MuRF-1/MAFbx expression was assessed using RT-PCR and Western blot (WB). In addition, the myocardial expression of TNF-α was measured (RT-PCR) and troponin I, β-myosin and phosphorylated Akt/Akt (pAkt/Akt) were quantified (WB). MuRF1 and MAFbx expression (mRNA and protein level) were significantly reduced in biopsies from MI patients. TNF-α was significantly higher in MI and exhibited a negative correlation with MuRF1 and MAFbx. The expression of troponin I and cardiomyocyte size were increased in MI in comparison to Con, whereas β-myosin expression was not altered. When compared with Con, pAkt/Akt was elevated. The results of the present study suggest that the atrogenes MuRF1/MAFbx are involved in regulating the hypertrophic response, characteristic of the early post-infarction remodeling phase. Reduced expression of MuRF1 and MAFbx in the myocardium might permit hypertrophy, which is supported by the elevation of troponin I. A regulatory role of TNF-α needs to be confirmed in further experiments.






Similar content being viewed by others
References
Abbate A, Biondi-Zoccai GG, Bussani R, Dobrina A, Camilot D, Feroce F, Rossiello R, Baldi F, Silvestri F, Biasucci LM, Baldi A (2003) Increased myocardial apoptosis in patients with unfavorable left ventricular remodeling and early symptomatic post-infarction heart failure. J Am Coll Cardiol 41:753–760
Adams V, Linke A, Gielen S, Erbs S, Hambrecht R, Schuler G (2008) Modulation of Murf-1 and MAFbx expression in the myocardium by physical exercise training. Eur J Cardiovasc Prev Rehabil 15:293–299
Adams V, Linke A, Wisloff U, Doring C, Erbs S, Krankel N, Witt CC, Labeit S, Muller-Werdan U, Schuler G, Hambrecht R (2007) Myocardial expression of Murf-1 and MAFbx after induction of chronic heart failure: effect on myocardial contractility. Cardiovasc Res 73:120–129
Anversa P, Olivetti G, Capasso JM (1991) Cellular basis of ventricular remodeling after myocardial infarction. Am J Cardiol 68:7D–16D
Anversa P, Olivetti G, Li P, Herman MV, Capasso JM (1993) Myocardial infarction, cardiac anatomy and ventricular loading. Cardioscience 4:55–62
Arya R, Kedar V, Hwang JR, McDonough H, Li HH, Taylor J, Patterson C (2004) Muscle ring finger protein-1 inhibits PKC{epsilon} activation and prevents cardiomyocyte hypertrophy. J Cell Biol 167:1147–1159
Centner T, Yano J, Kimura E, McElhinny AS, Pelin K, Witt CC, Bang ML, Trombitas K, Granzier H, Gregorio CC, Sorimachi H, Labeit S (2001) Identification of muscle specific ring finger proteins as potential regulators of the titin kinase domain. J Mol Biol 306:717–726
Condorelli G, Morisco C, Latronico MV, Claudio PP, Dent P, Tsichlis P, Frati G, Drusco A, Croce CM, Napoli C (2002) TNF-alpha signal transduction in rat neonatal cardiac myocytes: definition of pathways generating from the TNF-alpha receptor. FASEB J 16:1732–1737
Depre C, Wang Q, Yan L, Hedhli N, Peter P, Chen L, Hong C, Hittinger L, Ghaleh B, Sadoshima J, Vatner DE, Vatner SF, Madura K (2006) Activation of the cardiac proteasome during pressure overload promotes ventricular hypertrophy. Circulation 114:1821–1828
Dimmeler S, Assmus B, Hermann C, Haendeler J, Zeiher A (1998) Fluid shear stress stimulates phosphorylation of Akt in human endothelial cells. Circ Res 83:334–341
Fang CX, Dong F, Thomas DP, Ma H, He L, Ren J (2008) Hypertrophic cardiomyopathy in high-fat diet-induced obesity: role of suppression of forkhead transcription factor and atrophy gene transcription. Am J Physiol Heart Circ Physiol 295:H1206–H1215
Fielitz J, Kim MS, Shelton JM, Latif S, Spencer JA, Glass DJ, Richardson JA, Bassel-Duby R, Olson EN (2007) Myosin accumulation and striated muscle myopathy result from the loss of muscle RING finger 1 and 3. J Clin Invest 117:2486–2495
Fielitz J, van Rooij E, Spencer JA, Shelton JM, Latif S, van der Nagel R, Bezprozvannaya S, de Windt L, Richardson JA, Bassel-Duby R, Olson EN (2007) Loss of muscle-specific RING-finger 3 predisposes the heart to cardiac rupture after myocardial infarction. Proc Natl Acad Sci USA 104:4377–4382
Glass DJ (2005) Skeletal muscle hypertrophy and atrophy signaling pathways. Int J Biochem Cell Biol 37:1974–1984
Hiraoka E, Kawashima S, Takahashi T, Rikitake Y, Kitamura T, Ogawa W, Yokoyama M (2001) TNF-alpha induces protein synthesis through PI3-kinase-Akt/PKB pathway in cardiac myocytes. Am J Physiol Heart Circ Physiol 280:H1861–H1868
Houser SR, Margulies KB (2003) Is depressed myocyte contractility centrally involved in heart failure? Circ Res 92:350–358
Irwin MW, Mak S, Mann DL, Qu R, Penninger JM, Yan A, Dawood F, Wen W, Shou Z, Liu P (1999) Tissue expression and immunolocalization of tumor necrosis factor-α in postinfarction dysfunctional myocardium. Circulation 99:1492–1498
Kedar V, McDonough H, Arya R, Li HH, Rockman HA, Patterson C (2004) Muscle-specific RING finger 1 is a bona fide ubiquitin ligase that degrades cardiac troponin I. Proc Natl Acad Sci USA 101:18135–18140
Kubota T, McTiernan CF, Frye CS, Slawson SE, Lemster BH, Koretsky AP, Demetris AJ, Feldman AM (1997) Dilated cardiomyopathy in transgenic mice with cardiac-specific overexpression of tumor necrosis factor-alpha. Circ Res 81:627–635
Lauten A, Majos E, Mühlich A, Wahlers T, Weider S, Fischer JH, Figulla HR, Bloch W (2009) Ischemia-reperfusion injury activates early extracellular matrix processing and expression of endostatin in the heart with differential effects of temperature. Basic Res Cardiol 104:559–569
Li HH, Kedar V, Zhang C, McDonough H, Arya R, Wang DZ, Patterson C (2004) Atrogin-1/muscle atrophy F-box inhibits calcineurin-dependent cardiac hypertrophy by participating in an SCF ubiquitin ligase complex. J Clin Invest 114:1058–1071
Li HH, Willis MS, Lockyer P, Miller N, McDonough H, Glass DJ, Patterson C (2007) Atrogin-1 inhibits Akt-dependent cardiac hypertrophy in mice via ubiquitin-dependent coactivation of forkhead proteins. J Clin Invest 117:3211–3223
Mann DL (2002) Inflammatory mediators and the failing heart: past, present, and the foreseeable future. Circ Res 91:988–998
Mann DL, Bristow MR (2005) Mechanisms and models in heart failure: the biomechanical model and beyond. Circulation 111:2837–2849
Patterson C, Ike C, PWt Willis, Stouffer GA, Willis MS (2007) The bitter end: the ubiquitin–proteasome system and cardiac dysfunction. Circulation 115:1456–1463
Sack MN, Smith RM, Opie LH (2000) Tumor necrosis factor in myocardial hypertrophy and ischaemia: an anti-apoptotic perspective. Cardiovasc Res 45:688–695
Sack M (2002) Tumor necrosis factor-alpha in cardiovascular biology and the potential role for anti-tumor necrosis factor-alpha therapy in heart disease. Pharmacol Ther 94:23–35
Satoh M, Nakamura M, Akatsu T, Shimoda Y, Segawa I, Hiramori K (2006) C-reactive protein co-expresses with tumor necrosis factor-alpha in the myocardium in human dilated cardiomyopathy. Eur J Heart Fail 7:748–754
Schulz R, Heusch G (2009) Tumor necrosis factor-alpha and its receptors 1 and 2: Yin and Yang in myocardial infarction? Circulation 119:1355–1357
Schulze PC, Fang J, Kassik KA, Gannon J, Cupesi M, MacGillivray C, Lee RT, Rosenthal N (2005) Transgenic overexpression of locally acting insulin-like growth factor-1 inhibits ubiquitin-mediated muscle atrophy in chronic left-ventricular dysfunction. Circ Res 97:418–426
Skurk C, Izumiya Y, Maatz H, Razeghi P, Shiojima I, Sandri M, Sato K, Zeng L, Schiekofer S, Pimentel D, Lecker S, Taegtmeyer H, Goldberg AL, Walsh K (2005) The FOXO3a transcription factor regulates cardiac myocyte size downstream of AKT signaling. J Biol Chem 280:20814–20823
Skyschally A, Gres P, Hoffmann S, Haude M, Erbel R, Schulz R, Heusch G (2007) Bidirectional role of tumor necrosis factor-alpha in coronary microembolization: progressive contractile dysfunction versus delayed protection against infarction. Circ Res 100:140–146
Vanderheyden M, Mullens W, Delrue L, Goethals M, de Bruyne B, Wijns W, Geelen P, Verstreken S, Wellens F, Bartunek J (2008) Myocardial gene expression in heart failure patients treated with cardiac resynchronization therapy responders versus nonresponders. J Am Coll Cardiol 51:129–136
Wallace CK, Stetson SJ, Kucuker SA, Becker KA, Farmer JA, McRee SC, Koerner MM, Noon GP, Torre-Amione G (2005) Simvastatin decreases myocardial tumor necrosis factor alpha content in heart transplant recipients. J Heart Lung Transplant 24:46–51
Wang X, Robbins J (2006) Heart failure and protein quality control. Circ Res 99:1315–1328
Willis MS, Rojas M, Li L, Selzman CH, Tang RH, Stansfield WE, Rodriguez JE, Glass D, Patterson C (2009) Muscle ring finger-1 (MuRF1) mediates cardiac atrophy in vivo. Am J Physiol Heart Circ Physiol 296:H997–H1006
Acknowledgment
We would like to thank Nicole Urban and Tina Fischer for excellent technical assistance; and Siegfried Labeit for providing the MuRF1 specific antibody. This work was supported by Fonds voor Wetenschappelijk onderzoek (FWO)-Flanders (VC, clinical postdoctoral fellow) and by the Deutsche Forschungsgemeinschaft (LI946/3-1).
Conflict of interest statement
None declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Conraads , V.M., Vrints, C.J., Rodrigus, I.E. et al. Depressed expression of MuRF1 and MAFbx in areas remote of recent myocardial infarction: a mechanism contributing to myocardial remodeling? . Basic Res Cardiol 105, 219–226 (2010). https://doi.org/10.1007/s00395-009-0068-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00395-009-0068-5