Abstract
Purpose
Our recent study has reported that whole mung bean showed better beneficial effects on high-fat diet (HFD)-induced obesity and gut microbiota disorders when compared with the decorticated mung bean at the same intervention dose level, suggesting that the mung bean seed coat (MBC) may play a crucial role in its health benefits. This study aims to investigate whether MBC has beneficial benefits on the prevention of HFD-induced obesity and the modulation of gut microbiota in mice when it was supplemented in HFD.
Methods
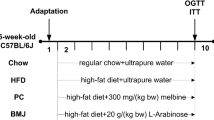
Herein, male C57BL/6 J mice were fed with normal control diet, HFD, and HFD supplemented with MBC (3–6%, w/w) for 12 weeks. The changes in physiological, histological, biochemical parameters, serum endotoxin, proinflammatory cytokines, and gut microbiota composition of mice were determined to assess the ability of MBC to alleviate HFD-induced obesity and modulate gut microbiota disorders in mice.
Results
MBC supplementation exhibited significant reductions in the HFD-induced adiposity, fat accumulation, serum lipid levels, lipopolysaccharide, and proinflammatory cytokines concentrations (P < 0.05), which was accompanied by improvements in hepatic steatosis and adipocyte size. Especially, the elevated fasting blood glucose and insulin resistance were also significantly improved by MBC supplementation (P < 0.05). Furthermore, high-throughput sequencing of the 16S rRNA gene revealed that MBC could normalize HFD-induced gut microbiota dysbiosis. MBC not only could promote the bloom of Akkermansia, but also restore several HFD-dependent taxa (Blautia, Ruminiclostridium_9, Bilophila, and unclassified_f_Ruminococcaceae) back to normal status, co-occurring with the decreases in obesity-related indices.
Conclusions
This study provides evidence that MBC may be mainly responsible for the beneficial effects of whole mung bean on preventing the HFD-induced changes, thus enlarging the application value of MBC.








Similar content being viewed by others
References
Rebello CJ, Greenway FL, Finley JW (2014) A review of the nutritional value of legumes and their effects on obesity and its related co-morbidities. Obes Rev 15(5):392–407. https://doi.org/10.1111/obr.12144
Tanaka M, Honda Y, Miwa S, Akahori R, Matsumoto K (2019) Comparison of the effects of roasted and boiled red kidney beans (Phaseolus vulgaris L.) on glucose/lipid metabolism and intestinal immunity in a high-fat diet-induced murine obesity model. J Food Sci 84(5):1180–1187. https://doi.org/10.1111/1750-3841.14583
Zhu Y-S, Shuai S, FitzGerald R (2018) Mung bean proteins and peptides: nutritional, functional and bioactive properties. Food Nutr Res. https://doi.org/10.29219/fnr.v62.1290(62 (0))
Hou D, Yousaf L, Xue Y, Hu J, Wu J, Hu X, Feng N, Shen Q (2019) Mung bean (Vigna radiata L.): bioactive polyphenols, polysaccharides, peptides, and health benefits. Nutrients 11(6):1238. https://doi.org/10.3390/nu11061238
Hou D, Zhao Q, Yousaf L, Xue Y, Shen Q (2020) Whole mung bean (Vigna radiata L.) supplementation prevents high-fat diet-induced obesity and disorders in a lipid profile and modulates gut microbiota in mice. Eur J Nutr. https://doi.org/10.1007/s00394-020-02196-2
Liyanage R, Kiramage C, Visvanathan R, Jayathilake C, Weththasinghe P, Bangamuwage R, Chaminda Jayawardana B, Vidanarachchi J (2018) Hypolipidemic and hypoglycemic potential of raw, boiled, and sprouted mung beans (Vigna radiata L. Wilczek) in rats. J Food Biochem 42(1):e12457. https://doi.org/10.1111/jfbc.12457
Luo J, Cai W, Wu T, Xu B (2016) Phytochemical distribution in hull and cotyledon of adzuki bean (Vigna angularis L.) and mung bean (Vigna radiate L.), and their contribution to antioxidant, anti-inflammatory and anti-diabetic activities. Food Chem 201:350–360. https://doi.org/10.1016/j.foodchem.2016.01.101
Inhae K, Seojin C, Joung HT, Munji C, Hae-Ri W, Won LB, Myoungsook L (2015) Effects of mung bean (Vigna radiata L.) ethanol extracts decrease proinflammatory cytokine-induced lipogenesis in the KK-Ay diabese mouse model. J Med Food 18(8):841–849. https://doi.org/10.1089/jmf.2014.3364
Jang Y-H, Kang M-J, Choe E-O, Shin M, Kim J-I (2014) Mung bean coat ameliorates hyperglycemia and the antioxidant status in type 2 diabetic db/db mice. Food Sci Biotechnol 23(1):247–252. https://doi.org/10.1007/s10068-014-0034-3
Kasprzak M, Rzedzicki Z (2010) Effect of pea seed coat admixture on physical properties and chemical composition of bread. Int Agrophys 24(2):149–156. https://doi.org/10.1016/j.indcrop.2009.09.012
Zhong L, Fang Z, Wahlqvist ML, Wu G, Hodgson JM, Johnson SK (2018) Seed coats of pulses as a food ingredient: characterization, processing, and applications. Trends Food Sci Tech 80:35–42. https://doi.org/10.1016/j.tifs.2018.07.021
Nicholson JK, Holmes E, Kinross J, Burcelin R, Gibson G, Jia W, Pettersson S (2012) Host-gut microbiota metabolic interactions. Science 336(6086):1262. https://doi.org/10.1126/science.1223813
Ley RE, Turnbaugh PJ, Klein S, Gordon JI (2006) Human gut microbes associated with obesity. Nature 444(7122):1022–1023. https://doi.org/10.1038/4441022a
Ridaura VK, Faith JJ, Rey FE, Cheng J, Duncan AE, Kau AL, Griffin NW, Lombard V, Henrissat B, Bain JR, Muehlbauer MJ, Ilkayeva O, Semenkovich CF, Funai K, Hayashi DK, Lyle BJ, Martini MC, Ursell LK, Clemente JC, Van Treuren W, Walters WA, Knight R, Newgard CB, Heath AC, Gordon JI (2013) Gut Microbiota from twins discordant for obesity modulate metabolism in mice. Science 341(6150):1241214. https://doi.org/10.1126/science.1241214
Rowland I, Gibson G, Heinken A, Scott K, Swann J, Thiele I, Tuohy K (2018) Gut microbiota functions: metabolism of nutrients and other food components. Eur J Nutr 57(1):1–24. https://doi.org/10.1007/s00394-017-1445-8
Bianchi F, Duque ALRF, Saad SMI, Sivieri K (2019) Gut microbiome approaches to treat obesity in humans. Appl Microbiol Biotechnol 103(3):1081–1094. https://doi.org/10.1007/s00253-018-9570-8
Gan R-Y, Deng Z-Q, Yan A-X, Shah NP, Lui W-Y, Chan C-L, Corke H (2016) Pigmented edible bean coats as natural sources of polyphenols with antioxidant and antibacterial effects. LWT-Food Sci Technol 73:168–177. https://doi.org/10.1016/j.lwt.2016.06.012
AACC (2000) Approved methods of the American association of cereal chemists. Methods 08–01(ash), 30–10(fat), 46–08 (protein), 32–05(total fiber) (10th ed.). St. Paul, Minnesota: American association of cereal chemists
Xu BJ, Chang SKC (2007) A comparative study on phenolic profiles and antioxidant activities of legumes as affected by extraction solvents. J Food Sci 72(2):S159–S166. https://doi.org/10.1111/j.1750-3841.2006.00260.x
Katsuki A, Sumida Y, Gabazza EC, Murashima S, Furuta M, Araki-Sasaki R, Hori Y, Yano Y, Adachi Y (2001) Homeostasis model assessment is a reliable indicator of insulin resistance during follow-up of patients with type 2 diabetes. Diabetes Care 24(2):362–365. https://doi.org/10.2337/diacare.24.2.362
Anhê FF, Roy D, Pilon G, Dudonné S, Matamoros S, Varin TV, Garofalo C, Moine Q, Desjardins Y, Levy E, Marette A (2015) A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice. Gut 64(6):872–883. https://doi.org/10.1136/gutjnl-2014-307142
Hou D, Zhao Q, Yousaf L, Chen B, Xue Y, Shen Q (2020) A comparison between whole mung bean and decorticated mung bean: beneficial effects on the regulation of serum glucose and lipid disorders and the gut microbiota in high-fat diet and streptozotocin-induced prediabetic mice. Food Funct 11(6):5525–5537. https://doi.org/10.1039/D0FO00379D
Zhang L, Shi M, Ji J, Hu X, Chen F (2019) Gut microbiota determines the prevention effects of Luffa cylindrica (L.) Roem supplementation against obesity and associated metabolic disorders induced by high-fat diet. FASEB J 33(9):10339–10352. https://doi.org/10.1096/fj.201900488R
Wolf G (1996) High-fat, high-cholesterol diet raises plasma HDL cholesterol: studies on the mechanism of this effect. Nutr Rev 54(1):34–35. https://doi.org/10.1111/j.1753-4884887.1996.tb03772.x
Yao Y, Chen F, Wang M, Wang J, Ren G (2008) Antidiabetic activity of mung bean extracts in diabetic KK-Ay mice. J Agr Food Chem 56(19):8869–8873. https://doi.org/10.1021/jf8009238
Kohno M, Motoyama T, Shigihara Y, Sakamoto M, Sugano H (2017) Improvement of glucose metabolism via mung bean protein consumption: a clinical trial of GLUCODIA TM isolated mung bean protein in Japan. Funct Foods Health Dis 7:115–134. https://doi.org/10.31989/ffhd.v7i2.320
Watanabe H, Inaba Y, Inoue H, Kimura K, Kaneko S, Asahara S-i, Kido Y, Matsumoto M, Kohno M, Tachibana N, Motoyama T (2016) Dietary mung bean protein reduces hepatic steatosis, fibrosis, and inflammation in male mice with diet-induced. Nonalcoholic Fatty Liver Dis J Nutr 147(1):52–60. https://doi.org/10.3945/jn.116.231662
Hashemi Z, Yang K, Yang H, Jin A, Ozga J, Chan CB (2014) Cooking enhances beneficial effects of pea seed coat consumption on glucose tolerance, incretin, and pancreatic hormones in high-fat-diet–fed rats. Appl Physiol Nutr Me 40(4):323–333. https://doi.org/10.1139/apnm-2014-0380
Chan CB, Gupta J, Kozicky L, Hashemi Z, Yang K (2014) Improved glucose tolerance in insulin-resistant rats after pea hull feeding is associated with changes in lipid metabolism-targeted transcriptome. Appl Physiol Nutr Me 39(10):1112–1119. https://doi.org/10.1139/apnm-2014-0054
Hashemi Z, Fouhse J, Im HS, Chan CB, Willing BP (2017) Dietary pea fiber supplementation improves glycemia and induces changes in the composition of gut microbiota, serum short chain fatty acid profile and expression of mucins in glucose intolerant rats. Nutrients 9(11):1236. https://doi.org/10.3390/nu9111236
Saltiel AR, Olefsky JM (2017) Inflammatory mechanisms linking obesity and metabolic disease. J Clin Invest 127(1):1–4. https://doi.org/10.1172/JCI92035
Cani PD, Bibiloni R, Knauf C, Waget A, Neyrinck AM, Delzenne NM, Burcelin R (2008) Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes 57(6):1470–1481. https://doi.org/10.2337/db07-1403
Moreira APB, Texeira TFS, Ferreira AB, do Carmo Gouveia Peluzio M, de Cássia Gonçalves Alfenas R (2012) Influence of a high-fat diet on gut microbiota, intestinal permeability and metabolic endotoxaemia. Brit J Nutr 108(5):801–809. https://doi.org/10.1017/S0007114512001213
Fuke N, Nagata N, Suganuma H, Ota T (2019) Regulation of gut microbiota and metabolic endotoxemia with dietary factors. Nutrients 11(10):2277. https://doi.org/10.3390/nu11102277
Wang J, Wang P, Li D, Hu X, Chen F (2020) Beneficial effects of ginger on prevention of obesity through modulation of gut microbiota in mice. Eur J Nutr 59(2):699–718. https://doi.org/10.1007/s00394-019-01938-1
Li T, Gao J, Du M, Mao X (2019) Bovine α-lactalbumin hydrolysates ameliorate obesity-associated endotoxemia and inflammation in high-fat diet-fed mice through modulation of gut microbiota. Food Funct 10(6):3368–3378. https://doi.org/10.1039/C8FO01967C
Suzuki T, Hara H (2011) Role of flavonoids in intestinal tight junction regulation. J Nutr Biochem 22(5):401–408. https://doi.org/10.1016/j.jnutbio.2010.08.001
Sae-tan S, Kumrungsee T, Yanaka N (2020) Mungbean seed coat water extract inhibits inflammation in LPS-induced acute liver injury mice and LPS-stimulated RAW 246.7 macrophages via the inhibition of TAK1/IκBα/NF-κB. J Food Sci Technol 57(7):2659–2668. https://doi.org/10.1007/s13197-020-04302-y
Zhu S, Li W, Li J, Jundoria A, Sama AE, Wang H (2012) It is not just folklore: the aqueous extract of mung bean coat is protective against sepsis. BMC Complem Altern M 2012:498467. https://doi.org/10.1155/2012/498467
Amabebe E, Robert FO, Agbalalah T, Orubu ESF (2020) Microbial dysbiosis-induced obesity: role of gut microbiota in homoeostasis of energy metabolism. Brit J Nutr 123(10):1127–1137. https://doi.org/10.1017/S0007114520000380
Laparra JM, Sanz Y (2010) Interactions of gut microbiota with functional food components and nutraceuticals. Pharmacolo Res 61(3):219–225. https://doi.org/10.1016/j.phrs.2009.11.001
Hou D, Zhao Q, Yousaf L, Khan J, Xue Y, Shen Q (2020) Consumption of mung bean (Vigna radiata L.) attenuates obesity, ameliorates lipid metabolic disorders and modifies the gut microbiota composition in mice fed a high-fat diet. J Funct Foods 64:103687. https://doi.org/10.1016/j.jff.2019.103687
Nakatani A, Li X, Miyamoto J, Igarashi M, Watanabe H, Sutou A, Watanabe K, Motoyama T, Tachibana N, Kohno M, Inoue H, Kimura I (2018) Dietary mung bean protein reduces high-fat diet-induced weight gain by modulating host bile acid metabolism in a gut microbiota-dependent manner. Biochem Bioph Res Co 501(4):955–961. https://doi.org/10.1016/j.bbrc.2018.05.090
Makki K, Deehan EC, Walter J, Bäckhed F (2018) The impact of dietary fiber on gut microbiota in host health and disease. Cell Host Microbe 23(6):705–715. https://doi.org/10.1016/j.chom.2018.05.012
Awika JM, Rose DJ, Simsek S (2018) Complementary effects of cereal and pulse polyphenols and dietary fiber on chronic inflammation and gut health. Food Funct 9(3):1389–1409. https://doi.org/10.1039/C7FO02011B
Kilua A, Chihiro H, Han K-H, Homma K, Fukuma N, Kamitani T, Suzuki T, Fukushima M (2020) Whole kidney bean (Phaseolus vulgaris) and bean hull reduce the total serum cholesterol, modulate the gut microbiota and affect the caecal fermentation in rats. Bioact Carbohyd Dietary Fibre 24:100232. https://doi.org/10.1016/j.bcdf.2020.100232
Myint H, Kishi H, Iwahashi Y, Saburi W, Koike S, Kobayashi Y (2018) Functional modulation of caecal fermentation and microbiota in rat by feeding bean husk as a dietary fibre supplement. Benef Mirbobes 9(6):963–974. https://doi.org/10.3920/bm2017.0174
Yang L, Zhao Y, Huang J, Zhang H, Lin Q, Han L, Liu J, Wang J, Liu H (2020) Insoluble dietary fiber from soy hulls regulates the gut microbiota in vitro and increases the abundance of bifidobacteriales and lactobacillales. J Food Sci Technol 57(1):152–162. https://doi.org/10.1007/s13197-019-04041-9
Yang L, Lin Q, Han L, Wang Z, Luo M, Kang W, Liu J, Wang J, Ma T, Liu H (2020) Soy hull dietary fiber alleviates inflammation in BALB/C mice by modulating the gut microbiota and suppressing the TLR-4/NF-κB signaling pathway. Food Funct 11(7):5965–5975. https://doi.org/10.1039/D0FO01102A
Canfora EE, Meex RCR, Venema K, Blaak EE (2019) Gut microbial metabolites in obesity, NAFLD and T2DM. Nat Rev Endocrinol 15(5):261–273. https://doi.org/10.1038/s41574-019-0156-z
Zhao L (2013) The gut microbiota and obesity: from correlation to causality. Nat Rev Microbiol 11:639. https://doi.org/10.1038/nrmicro3089
Daniel H, Gholami AM, Berry D, Desmarchelier C, Hahne H, Loh G, Mondot S, Lepage P, Rothballer M, Walker A, Böhm C, Wenning M, Wagner M, Blaut M, Schmitt-Kopplin P, Kuster B, Haller D, Clavel T (2014) High-fat diet alters gut microbiota physiology in mice. ISME J 8(2):295–308. https://doi.org/10.1038/ismej.2013.155
Clemente Jose C, Ursell Luke K, Parfrey Laura W, Knight R (2012) The impact of the gut microbiota on human health: an integrative view. Cell 148(6):1258–1270. https://doi.org/10.1016/j.cell.2012.01.035
Coelho OGL, Cândido FG, Alfenas RdCG (2019) Dietary fat and gut microbiota: mechanisms involved in obesity control. Crit Rev Food Sci 59(19):3045–3053. https://doi.org/10.1080/10408398.2018.1481821
De Filippo C, Cavalieri D, Di Paola M, Ramazzotti M, Poullet JB, Massart S, Collini S, Pieraccini G, Lionetti P (2010) Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. P Natl Sci 107(33):14691–14696. https://doi.org/10.1073/pnas.1005963107
Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI (2006) An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444(7122):1027–1031. https://doi.org/10.1038/nature05414
Araújo JR, Tomas J, Brenner C, Sansonetti PJ (2017) Impact of high-fat diet on the intestinal microbiota and small intestinal physiology before and after the onset of obesity. Biochimie 141:97–106. https://doi.org/10.1016/j.biochi.2017.05.019
Hildebrandt MA, Hoffmann C, Sherrill-Mix SA, Keilbaugh SA, Hamady M, Chen YY, Knight R, Ahima RS, Bushman F, Wu GD (2009) High-fat diet determines the composition of the murine gut microbiome independently of obesity. Gastroenterology 137(5):1716–1724.e1712. https://doi.org/10.1053/j.gastro.2009.08.042
Belzer C, de Vos WM (2012) Microbes inside—from diversity to function: the case of Akkermansia. ISME J 6(8):1449–1458. https://doi.org/10.1038/ismej.2012.6
Shin N-R, Lee J-C, Lee H-Y, Kim M-S, Whon TW, Lee M-S, Bae J-W (2014) An increase in the Akkermansia spp. population induced by metformin treatment improves glucose homeostasis in diet-induced obese mice. Gut 63(5):727–735. https://doi.org/10.1136/gutjnl-2012-303839
Everard A, Belzer C, Geurts L, Ouwerkerk JP, Druart C, Bindels LB, Guiot Y, Derrien M, Muccioli GG, Delzenne NM, de Vos WM, Cani PD (2013) Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. P Natl Sci 110(22):9066–9071. https://doi.org/10.1073/pnas.1219451110
Zhang Z, Wu XY, Cao SY, Cromie M, Shen YH, Feng YM, Yang H, Li L (2017) Chlorogenic acid ameliorates experimental colitis by promoting growth of Akkermansia in mice. Nutrients 9(7):13. https://doi.org/10.3390/nu9070677
Zhang Z, Wu XY, Cao SY, Wang L, Wang D, Yang H, Feng YM, Wang SL, Li L (2016) Caffeic acid ameliorates colitis in association with increased Akkermansia population in the gut microbiota of mice. Oncotarget 7(22):31790–31799
Fang Q, Hu J, Nie Q, Nie S (2019) Effects of polysaccharides on glycometabolism based on gut microbiota alteration. Trends Food Sci Tech 92:65–70. https://doi.org/10.1016/j.tifs.2019.08.015
Lai F, Wen Q, Li L, Wu H, Li X (2010) Antioxidant activities of water-soluble polysaccharide extracted from mung bean (Vigna radiata L.) hull with ultrasonic assisted treatment. Carbohyd Polym 81(2):323–329. https://doi.org/10.1016/j.carbpol.2010.02.011
Sharma V, Smolin J, Nayak J, Ayala JE, Scott DA, Peterson SN, Freeze HH (2018) Mannose alters gut microbiome, prevents diet-induced obesity, and improves host metabolism. Cell Rep 24(12):3087–3098. https://doi.org/10.1016/j.celrep.2018.08.064
Sánchez B, Delgado S, Blanco-Míguez A, Lourenço A, Gueimonde M, Margolles A (2017) Probiotics, gut microbiota, and their influence on host health and disease. Mol Nutr Food Res 61(1):1600240. https://doi.org/10.1002/mnfr.201600240
Wan MLY, Ling KH, El-Nezami H, Wang MF (2019) Influence of functional food components on gut health. Crit Rev Food Sci 59(12):1927–1936. https://doi.org/10.1080/10408398.2018.1433629
Acknowledgements
This work was financially supported by the National Key Research and Development Program of China (2017YFD0401202).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hou, D., Zhao, Q., Yousaf, L. et al. Beneficial effects of mung bean seed coat on the prevention of high-fat diet-induced obesity and the modulation of gut microbiota in mice. Eur J Nutr 60, 2029–2045 (2021). https://doi.org/10.1007/s00394-020-02395-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-020-02395-x