Abstract
Background and aims
B1a lymphocytes (CD5+) are the major contributors of natural antibodies (Ab) implicated in the initial protection against several infections. The aim of this study was to assess the expression of these cells in the peripheral blood of ulcerative colitis (UC) patients who underwent restorative proctocolectomy (RPC) and others who were not operated on.
Materials and methods
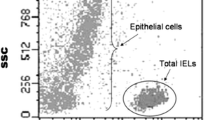
The blood concentration of CD5+ B cells was analysed by three-colour flow cytometry. Blood was collected from 38 UC patients, 20 of whom had undergone RPC and compared with the results in 18 healthy controls and in 12 familial adenomatous polyposis (FAP) patients who had undergone RPC. We were interested in evaluating if there was any correlation between B1a blood cell concentration and ESR and CRP levels, clinical, endoscopic and histological activity, perinuclear anti-neutrophil cytoplasmic antibody (pANCA) and extra-intestinal symptoms.
Results
B1a cell blood concentration was reduced in non-operated UC patients (20.7 ± 4.6/μl) with respect to that in healthy controls (71.1 ± 18.0/μl, p < 0.05). It was also lower in UC patients with RPC (24.9 ± 1.0/μl) compared to RPC for FAP (48.2 ± 6.2, p < 0.05). B1a cell rate correlated inversely in UC patients with ESR (R = −0.41, p < 0.05) and CRP levels (R = −0.47, p = 0.01).
Conclusion
B1a cell concentration was reduced in the blood of patients with UC even after the diseased organ was surgically removed by proctocolectomy. As these cells play an important role in natural immunity against luminal stimuli, consistently lower levels that are found in UC patients could be responsible for the impaired immunologic response to gut antigens in this disease.



Similar content being viewed by others
References
Radford-Smith G (1997) Ulcerative colitis: an immunological disease? Baillières Clin Gastroenterol 11:35–52
Kraehenbuhl JP, Corbett M (2004) Keeping the gut microflora at bay. Science 303:1624–1625
Baumgarth N, Tung JW, Herzenberg LA (2005) Inherent specificities in natural antibodies: a key to immune defense against pathogen invasion. Springer Semin Immunopathol 26:347–362
Macpherson A, Khoo UY, Forgacs I, Philpott-Howard J, Bjarnason I (1996) Mucosal antibodies in inflammatory bowel disease are directed against intestinal bacteria. Gut 38:365–375
Macpherson AJ, Gatto D, Sainsbury E, Harriman GR, Hengartner H, Zinkernagel RM (2000) A primitive T cell-independent mechanism of intestinal mucosal IgA responses to commensal bacteria. Science 288:2222–2226
Renaudineau Y, Pers JO, Bendaoud B, Jamin C, Youinou P (2004) Dysfunctional B cells in systemic lupus erythematosus. Autoimmun Rev 3:516–523
Seo M, Okada M, Yao T, Ueki M, Arima S, Okumura M (1992) An index of disease activity in patients with ulcerative colitis. Am J Gastroenterol 87:971–976
Baron JH, Connel AM, Lennard-Jones JE (1964) Variation between observers in describing mucosal appearances in proctocolitis. Br Med J 1:89–92
Floren CH, Benoni C, Willen R (1987) Histologic and colonoscopic assessment of disease extension in ulcerative colitis. Scand J Gastroenterol 22:458–462
Sandborn WJ, Tremaine WJ, Batts KP, Pemberton JH, Phillips SF (1994) Pouchitis after ileal pouch–anal anastomosis: a pouchitis disease activity index. Mayo Clinic Proc 69:409–415
Dougados M, Van der Linden S, Juhin R (1991) The European Spondyloarthropathy Study Group preliminary criteria for the classification of spondyloarthropathy. Arthritis Rheum 34:1218–1227
Senju M, Hulstaert F, Lowder J, Jewell DP (1991) Flow cytometric analysis of peripheral blood lymphocytes in ulcerative colitis and Crohn’s disease. Gut 32:779–783
Herzenberg LA (2000) B1 cells: the lineage question revisited. Immunol Rev 175:9–22
Hayakawa K, Hardy RR (2000) Development and function of B1 cells. Curr Opin Immunol 12:346–353
Su IH, Tarakhovsky A (2000) B-cells: orthodox or conformist? Curr Opin Immunol 12:191–194
Youinou P, Jamin C, Lydyard PM (1999) CD5 expression in human B-cell populations. Immunol Today 20:312–316
Bikah G, Carey J, Ciallella JR, Tarakhovsky A, Bondada S (1996) CD-5 mediated negative regulation of antigen receptor-induced growth signals in B-1 cells. Science 274:1906–1909
Sen G, Bikah G, Venkataraman C, Bondada S (1999) Negative regulation of antigen receptor-mediated signalling by constitutive association of CD5 with SHP-1 protein tyrosine phosphatase in B-1 cells. Eur J Immunol 29:3319–3328
Masmoudi H, Mota-Santos T, Huetz F, Coutinho a, Cazenave PA (1990) All T15 Id-positive antibodies (but not the majority of VHT15+ antibodies) are produced by peritoneal CD5+ B lymphocytes. Int Immunol 2:515–520
Hayakawa K, Hardy RR, Honda M, Herzenberg LA, Steinberg AD, Herzenberg LA (1984) Ly-1 B cells: functionally distinct lymphocytes that secrete IgM antibodies. Proc Natl Acad Sci U S A 81:2494–2498
Casali P, Burastero SE, Nakamura M, Inghirami G, Notkins AL (1987) Human lymphocytes making rheumatoid factor and antibody to ssDNA belong to Leu-1+ B-cell subset. Science 236:77–81
Brennan F, Plater-Zyberk C, Maini RN, Feldmann M (1989) Coordinate expansion of “fetal type” lymphocytes (TCR γδ+ T and CD5+ B) in rheumatoid arthritis and primary Sjogren’s syndrome. Clin Exp Immunol 77:175–178
Ebo D, DeClerck LS, Bridts CH, Stevens WJ (1994) Expression of CD5 and CD23 on B cells of patients with rheumatoid arthritis, systemic lupus erythematosus and Sjogren’s syndrome. Relationship with disease activity and treatment. In Vivo 8:577–580
Seidi OA, Semra YK, Sharief MK (2002) Expression of CD5 on B lymphocytes correlates with disease activity in patients with multiple sclerosis. J Neuroimmunol 133:205–210
Youinou P, Renaudineau Y (2004) The antiphospholipid syndrome as a model for B cell-induced autoimmune diseases. Thromb Res 114:363–369
Neil GA, Summers RW, Cheyne BA, Capenter C, Huang WL, Kansas GS, Waldshcmidt TJ (1992) CD5+ B cells are decreased in peripheral blood of patients with Crohn’s disease. Dig Dis Sci 37:1390–1395
Rizzello F, Gionchetti P, Venturi A, Amadini C, Romagnoli R, Campieri M (2002) Review article: monitoring activity in ulcerative colitis. Aliment Pharmacol Ther 16(Suppl 4):3–6
Nielsen OH, Vainer B, Madsen SM, Seidelin JB, Heegard NH (2000) Established and emerging biological activity markers of inflammatory bowel disease. Am J Gastroenterol 95:359–367
Sachar DB, Smith H, Chan S, Cohen LB, Lichtiger S, Messer J (1986) Erythrocytic sedimentation rate as a measure of clinical activity in inflammatory bowel disease. J Clin Gastroenterol 8:647–650
Dearing WH, McGuckin WH, Elveback LR (1969) Serum alpha1-acid glycoprotein in chronic ulcerative colitis. Gastroenterology 56:295–303
Fagan EA, Dyck RF, Maton PN, Hodgson HJ, Chadvick VS, Petrie A, Pepys MB (1982) Serum levels of C-reactive protein in Crohn’s disease and ulcerative colitis. Eur J Clin Invest 12:351–360
Vermeire S, Van Assche G, Rutgeerts P (2006) Laboratory markers in IBD: useful, magic, or unnecessary toys? Gut 55:426–431
Solem CA, Loftus EV, Tremaine WJ, Harmsen WS, Zinsmeister AR, Sandborn WJ (2005) Correlation of C-reactive protein with clinical, endoscopic, histologic and radiographic activity in inflammatory bowel disease. Inflamm Bowel Dis 11:707–712
Fiocchi C (1998) Inflammatory bowel disease: etiology and pathogenesis. Gastroenterology 115:182–205
Ruthlein J, Ibe M, Burghardt W, Mossner J, Auer IO (1992) Immunoglobulin G (IgG), IgG1 and IgG2 determination from endoscopy biopsy specimen in control, Crohn’s disease and ulcerative colitis subjects. Gut 33:507–512
Acknowledgment
The authors are grateful to Linda Inverso Moretti for her assistance in editing the English version of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Polese, L., De Franchis, G., Scarpa, M. et al. B1a lymphocytes in ulcerative colitis. Int J Colorectal Dis 22, 1005–1011 (2007). https://doi.org/10.1007/s00384-007-0298-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-007-0298-7