Abstract
Background
The human enteric nervous system (ENS) descends from migrating neural crest cells (NCC) and is structured into different plexuses embedded in the gastrointestinal tract wall. The development of this entity strongly depends on the supply of an appropriate support with trophic factors during organogenesis. The lack of important factors, such as glial cell line-derived neurotrophic factor, leads to severe disturbances in the ENS and, thus, to motility disorders in children. The isolation of neuronal precursor cells as well as their transplantation after expansion in vitro is therefore a hopeful new approach concerning all forms of dysganglionosis in children.
Methods
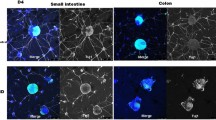
We therefore established a way to isolate and expand precursor cells from the developing and postnatal human ENS. Bowel samples were obtained from human fetuses and children (from the 9th week of gestation to 5 years postnatal). Myenteric plexus was isolated by enzymatical digestion and cultivated until spheroid aggregates, the so-called neurospheres, developed. These neurospheres could be differentiated and also be transplanted after dissociation into aganglionic bowel in vitro.
Results
Enteric neurospheres could be grown from different gestational ages, including postmortem material. Undifferentiated proliferating precursor cells were kept in culture for up to 72 days and could be differentiated in neurons and glial cells in vitro.
Conclusion
The first results using isolated enteric neurospheres in aganglionic bowel are quite promising and are a basis to develop an appropriate cell therapy for all kinds of dysganglionosis, especially for cases where a surgical approach is not sufficient or not even possible.




Similar content being viewed by others
References
Bixby S, Kruger GM, Mosher JT, Joseph NM, Morrison SJ (2002) Cell-intrinsic differences between stem cells from different regions of the peripheral nervous system regulate the generation of neural diversity. Neuron 35:643–656
Bondurand N, Natarajan D, Thapar N, Atkins C, Pachnis V (2003) Neuron and glia generating progenitors of the mammalian enteric nervous system isolated from foetal and postnatal gut cultures. Development 130:6387–6400
Brizzolara A, Favre A, Schäfer K-H, Michelazzi A, Sanguineti M, Martucciello G, Jasonni V (2002) Mouse-isolated plexus differentiates neural crest precursors into enteric neuroblasts. Eur J Pediatr Surg 12:391–396
Burns AJ, Pasricha PJ, Young HM (2004) Enteric neural crest-derived cells and neural stem cells: biology and therapeutic potential. Neurogastroenterol Motil 16:3–7
Chalazonitis A (2004) Neurotrophin-3 in the development of the enteric nervous system. Prog Brain Res 146:243–263
Fu M, Tam PKH, Sham MH, Lui VCH (2004) Embryonic development of the ganglion plexuses and the concentric layer structure of human gut: a topographical study. Anat Embryol 208:33–41
Gritti A, Cova L, Parati EA, Galli R, Vescovi AL (1995) Basic fibroblast growth factor supports the proliferation of epidermal growth factor-generated neuronal precursor cells of the adult mouse CNS. Neurosci Lett 185:151–154
Kruger GM, Mosher JT, Bixby S, Joseph N, Iwashita T, Morrison SJ (2002) Neural stem cells persist in the adult gut but undergo changes in self-renewal, neuronal subtype potential, and factor responsiveness. Neuron 35:657–669
Lendahl U, Zimmermann LB, McKay RDG (1990) CNS stem cells express a new class of intermediate filament proteins. Cell 60:585–595
Lo L, Anderson DJ (1995) Postmigratory neural crest cells expressing c-RET display restricted developmental and proliferative capacities. Neuron 15:527–539
Newgreen D, Young HM (2002) Enteric nervous system: development and developmental disturbances—part 2. Pediatr Dev Pathol 5:329–349
Palmer TD, Schwartz PH, Taupin P, Kaspar B, Stein SA, Gage FH (2001) Progenitor cells from human brain after death. Nature 411:42–43
Rakic P (1972) Mode of cell migration to the superficial layers of fetal monkey neocortex. J Comp Neurol 145:61–83
Reynolds BA, Weiss S (1992) Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 255:1707–1710
Schäfer K-H, Saffrey MJ, Burnstock G, Mestres-Ventura P (1997) A new method for the isolation of myenteric plexus from the newborn rat gastrointestinal tract. Brain Res Protoc 1:109–113
Schäfer K-H (2000) Reaggregation of rat dissociated myenteric plexus in an extracellular matrix gel as a model for the transplantation of enteric neurones. In: Krammer HJ, Singer MV (eds) Neurogastroenterology from the basics to the clinics. Proceedings of the Falk Symposium 112. Kluwer, Dordrecht, 60 pp
Schäfer K-H, Hagl CI, Rauch U (2003) Differentiation of neurospheres from the enteric nervous system. Pediatr Surg Int 19:340–344
Schäfer K-H, Mestres P (1997) Human newborn and adult myenteric plexus display different growth patterns in tissue culture. Cell Mol Biol 43(8):1171–1180
Tew EM, Anderson PN, Saffrey MJ, Burnstock G (1994) Transplantation of the postnatal rat myenteric plexus into the adult rat corpus striatum: an electron microscopic study. Exp Neurol 129:120–129
Tohyama T, Lee VM, Rorke LB, Marvin M, McKay RD, Trojanowski JQ (1992) Nestin expression in embryonic human neuroepithelium and in human neuroepithelial tumor cells. Lab Invest 66:303–313
Vescovi AL, Reynolds BA, Fraser DD, Weiss S (1993) bFGF regulates the proliferative fate of unipotent (neuronal) and bipotent (neuronal/astroglial) EGF-generated CNS progenitor cells. Neuron 11:951–966
Young HM, Anderson RB, Anderson CR (2004) Guidance cues involved in the development of the peripheral autonomic nervous system. Auton Neurosci 112:1–14
Acknowledgements
We thank Elvira Wink and Silke Maas-Omlor for their excellent technical assistance. This study was supported by grant DFG SCHA 878/1-1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rauch, U., Hänsgen, A., Hagl, C. et al. Isolation and cultivation of neuronal precursor cells from the developing human enteric nervous system as a tool for cell therapy in dysganglionosis. Int J Colorectal Dis 21, 554–559 (2006). https://doi.org/10.1007/s00384-005-0051-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-005-0051-z