Abstract
Purpose
In recent years, many studies have made considerable progress in the development of stem cell-based therapies for Hirschsprung’s disease (HD). However, the question of whether enteric neural crest-derived cells (ENCCs) that are transplanted into the aganglionic gut can migrate, proliferate, and differentiate in a normal manner remains unanswered. Thus, we designed this study to compare the behavior of ENCCs transplanted into the aganglionic gut of endothelin receptor B knockout (Ednrb-KO) mice versus wild-type (WT) mice.
Methods
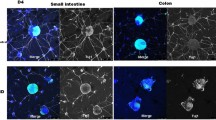
ENCCs were isolated from the fetal guts of Sox10 transgenic mice, in which ENCCs were labeled with an enhanced green fluorescent protein, Venus, on an embryonic day 18.5 (E18.5). Neurospheres were generated and transplanted into the aganglionic region of either Ednrb-KO mice gut, or WT mice gut that had not yet been colonized, on E12.5. Time-lapse imaging of the transplanted ENCCs was performed after 24, 48, and 72 h of culture. Neuronal differentiation was evaluated using whole-mount immunohistochemistry.
Results
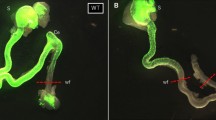
Sox10-positive ENCCs were seen to successfully migrate into the myenteric region of the aganglionic gut following transplantation in both the Ednrb-KO and WT mice. The ratio of Tuj1-positive/Sox10-positive cells was significantly increased after 72 h of culture compared to 24 h in the Ednrb-KO mice, which suggests that the transplanted ENCCs differentiated over time. In addition, at the 72 h timepoint, neuronal differentiation of transplanted ENCC in the aganglionic gut of Ednrb-KO mice was significantly increased compared to that of WT mice.
Conclusions
The results of our study demonstrated that transplanted ENCCs migrated into the myenteric region of the aganglionic recipient gut in mice. The increased neuronal differentiation of transplanted ENCC in Endrb-KO mice gut suggests that the microenvironment of this region affects ENCC behavior following transplantation. Further research to explore the characteristics of this microenvironment will improve the potential of developing cell therapy to treat HD patients.



Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Okamoto E, Satani M, Kuwata K (1982) Histologic and embryologic studies on the innervation of the pelvic viscera in patients with Hirschsprung’s disease. Surg Gynecol Obstet 155:823–828
Young HM, Bergner AJ, Anderson RB et al (2004) Dynamics of neural crest-derived cell migration in the embryonic mouse gut. Dev Biol 270:455–473
Alhawaj AF (2021) Stem cell-based therapy for hirschsprung disease, do we have the guts to treat? Gene Ther. https://doi.org/10.1038/s41434-021-00268-4
Shibata S, Yasuda A, Renault-Mihara F et al (2010) Sox10-Venus mice: a new tool for real-time labeling of neural crest lineage cells and oligodendrocytes. Mol Brain 3:31
Mueller JL, Goldstein AM (2022) The science of Hirschsprung disease: what we know and where we are headed. Semin Pediatr Surg. https://doi.org/10.1016/j.sempedsurg.2022.151157
Nandor N, Goldstein AM (2006) Endothelin-3 regulates neural crest cell proliferation and differentiation in the hindgut enteric nervous system. Dev Biol 293:203–217
Bondurand N, Dufour, et al (2018) News from the endothelin-3/EDNRB signaling pathway: Role during enteric nervous system development and involvement in neural crest-associated disorders. Dev Biol 444(Suppl 1):S156–S169
Druckenbrod NR, Epstein ML (2009) Age-dependent changes in the gut environment restrict the invasion of the hindgut by enteric neural progenitors. Development 136:3195–3203
Breau MA, Pietri T, Eder O et al (2006) Lack of beta1 integrins in enteric neural crest cells leads to a Hirschsprung-like phenotype. Development 133:1725–1734
Nakazawa-Tanaka N, Miyahara K, Fujiwara N et al (2020) Decreased expression of β1 integrin in enteric neural crest cells of the endothelin receptor B null mouse model. Surg Int 36:43–48
Parikh DH, Tam PK, Lloyd DA et al (1992) Quantitative and qualitative analysis of the extracellular matrix protein, laminin, in Hirschsprung’s disease. J Pediatr Surg 27:991–996
Nakazawa-Tanaka N, Fujiwara N, Miyahara K et al (2018) The effect of laminin-1 on enteric neural crest-derived cell migration in the Hirschsprung’s disease mouse model. Pediatr Surg Int 34:143–147
Acknowledgements
We thank Ms Mirei Takahashi for her support. This work was supported by JSPS KAKENHI Grant Number 21K08650.
Author information
Authors and Affiliations
Contributions
Nana Nakazawa-Tanaka performed laboratory experiments, summarized the results and wrote the manuscript. Naho Fujiwara and Katsumi Miyahara contributed some parts of experiments. Chihiro Akazawa, Masahiko Urao and Atsuyuki Yamataka supervised the interpretation of result.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nakazawa-Tanaka, N., Fujiwara, N., Miyahara, K. et al. Increased enteric neural crest cell differentiation after transplantation into aganglionic mouse gut. Pediatr Surg Int 39, 29 (2023). https://doi.org/10.1007/s00383-022-05324-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s00383-022-05324-7