Abstract
Purpose
The roles of commensal bacteria after intestinal ischemia and reperfusion (IIR) are unclear. In current study, we aim to investigate the effects and underlying mechanisms of commensal bacteria in injury and epithelial restitution after IIR.
Methods
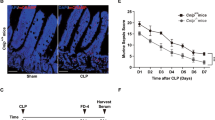
Commensal gut bacteria were deleted by broad-spectrum antibiotics in mice. IIR was induced by clamping superior mesenteric artery. Intestinal injury, permeability, epithelial proliferation, and proinflammatory activity of mesenteric lymph were investigated.
Results
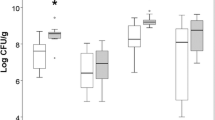
Commensals deletion improved mice survival in the early phase, but failed to improve the overall survival at 96 h after IIR. Commensals deletion reduced proliferation of intestinal epithelial cells (IEC) and augmented proinflammatory activity of mesenteric lymph after IIR. Lipopolysaccharides (LPS) supplement promoted IEC proliferation and improved survival in mice with commensals deletion after IIR. LPS induced production of prostaglandin E2 (PGE2) in mucosa via toll-like receptor 4-NFκB-cyclooxygenase 2 pathway. PGE2 enhanced IEC proliferation in vivo, which was preceded by activation of Akt and extracellular signal-regulated kinase (ERK) 1/2. Blocking of EGFR, PI3K/Akt activity abolished LPS-induced IEC proliferation.
Conclusions
Commensal bacteria are essential for epithelial restitution after IIR, which enhance IEC proliferation via induction of PGE2.






Similar content being viewed by others
References
Granger DNHL, Kvietys P (2015) The gastrointestinal circulation: physiology and pathophysiology. Compr Physiol 5:1541–1583. https://doi.org/10.1002/cphy.c150007
Deitch E, Xu D, Kaise V (2006) Role of the gut in the development of injury- and shock induced SIRS and MODS: the gut-lymph hypothesis, a review. Front Biosci 1:520–528. https://doi.org/10.2741/1816
Deitch E (2012) Gut-origin sepsis: evolution of a concept. Surgeon 10:350–356. https://doi.org/10.1016/j.surge.2012.03.003
Yi J, Slaughter A, Kotter C, Moore E, Hauser C, Itagaki K, Wohlauer M, Frank D, Silliman C, Banerjee A, Peltz E (2015) A "clean case" of systemic injury: mesenteric lymph after hemorrhagic shock elicites a sterile inflammatory response. Shock 44:336–340. https://doi.org/10.1097/SHK.0000000000000431
Kagnoff M (2014) The intestinal epithelium is an integral component of a communications network. J Clin Invest 124:2841–2843. https://doi.org/10.1172/JCI75225
Yoshiya K, Lapchak P, Thai T, Kannan L, Rani P, Dalle Lucca J, Tsokos G (2011) Depletion of gut commensal bacteria attenuates intestinal ischemia/reperfusion injury. Am J Physiol Gastrointest Liver Physiol 301:G1020–1030. https://doi.org/10.1152/ajpgi.00239.2011
Souza D, Vieira A, Soares A, Pinho V, Nicoli J, Vieira L, Teixeira M (2004) The essential role of the intestinal microbiota in facilitating acute inflammatory responses. J Immunol 173:4137–4146. https://doi.org/10.4049/jimmunol.173.6.4137
Sorkine P, Szold O, Halpern P, Gutman M, Greemland M, Rudick V, Goldman G (1997) Gut decontamination reduces bowel ischemia-induced lung injury in rats. Chest 112:491–495. https://doi.org/10.1378/chest.112.2.491
Chen L, Chang W, Chen P, Liu W, Hsu C (2008) TLR ligand decreases mesenteric ischemia and reperfusion injury-induced gut damage through TNF-alpha signaling. Shock 30:563–570. https://doi.org/10.1097/SHK.0b013e31816a3458
van der Hoven B, Nabuurs M, van Leengoed L, Groeneveld A, Thijs L (2001) Gut luminal endotoxin reduces ischemia-reperfusion injury of the small gut in germ-free pigs. Shock 16:28–32
Tatum PM Jr, Lorenz RG, Dimmitt RA (2010) Toll-like receptor 4 is protective against neonatal murine ischemia–reperfusion intestinal injury. J Pediatr Surg 45:1246–1255. https://doi.org/10.1016/j.jpedsurg.2010.02.093
Gatt M, Reddy B, MacFie J (2007) Review article: bacterial translocation in the critically ill-evidence and methods of prevention. Aliment Pharmacol Ther 25:741–757. https://doi.org/10.1111/j.1365-2036.2006.03174.x
Rakoff-Nahoum SPJ, Eslami-Varzaneh F, Edberg S, Medzhitov R (2004) Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell 118:229–241. https://doi.org/10.1016/j.cell.2004.07.002
Zhang H, Besner G, Feng J (2016) Antibody blockade of mucosal addressing cell adhesion molecule-1 attenuates proinflammatory activity of mesenteric lymph after hemorrhagic shock and resuscitation. Surgery 159:1449–1460. https://doi.org/10.1016/j.surg.2015.12.013
James I, Chen C, Huang G, Zhang H, Velten M, Besner G (2010) HB-EGF protects the lungs after intestinal ischemia/reperfusion injury. J Surg Res 163:86–95. https://doi.org/10.1016/j.jss.2010.03.062
El-Assal O, Besner G (2005) HB-EGF enhances restitution after intestinal ischemia/reperfusion via PI3K/Akt and MEK/ERK1/2 activation. Gastroenterology 129:609–625. https://doi.org/10.1016/j.gastro.2005.05.054
Round J, Mazmanian S (2009) The gut microbiota shapes intestinal immune responses during health and disease. Nat Rev Immunol 9:313–323. https://doi.org/10.1038/nri2515
Nezu Y, Tagawa M, Sakaue Y, Hara Y, Tsuchida S, Ogawa R (2002) Kinetics of endotoxin concentration and tumor necrosis factor-alpha, interleukin-1beta, and interleukin-6 activities in the systemic and portal circulation during small intestinal ischemia and reperfusion in dogs. Am J Vet Res 63:1682–1686
Cicalese L, Billiar T, Rao A, Bauer A (1997) Interaction between ischemia/reperfusion-induced leukocyte emigration and translocating bacterial enterotoxins on enteric muscle function. Transplant Proc 29:1815. https://doi.org/10.1016/s0041-1345(97)00080-8
Bauer P, Russell J, Granger D (1999) Role of endotoxin in intestinal reperfusion-induced expression of E-selectin. Am J Physiol 276:G479–484. https://doi.org/10.1152/ajpgi.1999.276.2.G479
Hardaway R (1998) Traumatic and septic shock alias post-trauma critical illness. Br J Surg 88:1473–1479. https://doi.org/10.1046/j.1365-2168.1998.00911.x
Deitch E (2010) Gut lymph and lymphatics: a source of factors leading to organ injury and dysfunction. Ann N Y Acad Sci 1207:E103–111. https://doi.org/10.1111/j.1749-6632.2010.05713.x
Tang Y, Swartz-Basile D, Swietlicki E, Yi L, Rubin D, Levin M (2004) Bax is required for resection-induced changes in apoptosis, proliferation, and members of the extrinsic cell death pathways. Gastroenterology 126:220–230. https://doi.org/10.1053/j.gastro.2003.10.077
Kubinak J, Round J (2012) Toll-like receptors promote mutually beneficial commensal-host interactions. PLoS Pathog 8:e1002785. https://doi.org/10.1371/journal.ppat.1002785
Im E, Riegler F, Pothoulakis C, Rhee S (2012) Elevated lipopolysaccharide in the colon evokes intestinal inflammation, aggravated in immune modulator-impaired mice. Am J Physiol Gastrointest Liver Physiol 303:G490–497. https://doi.org/10.1152/ajpgi.00120.2012
Shao J, Sheng G, Mifflin R, Powell D, Sheng H (2006) Roles of myofibroblasts in prostaglandin E2-stimulated intestinal epithelial proliferation and angiogenesis. Cancer Res 66:846–855. https://doi.org/10.1158/0008-5472.CAN-05-2606
Iwanaga K, Okada M, Murata T, Hori M, Ozaki H (2012) Prostaglandin E2 promotes wound-induced migration of intestinal subepithelial myofibroblasts via EP2, EP3, and EP4 prostanoid receptor activation. J Pharmacol Exp Ther 340:604–611. https://doi.org/10.1124/jpet.111.189845
Funding
This study was supported by grants from the National Natural Science Foundation of China (nos. 81200266 and 81270441).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, Hy., Wang, F., Chen, X. et al. Dual roles of commensal bacteria after intestinal ischemia and reperfusion. Pediatr Surg Int 36, 81–91 (2020). https://doi.org/10.1007/s00383-019-04555-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-019-04555-5