Abstract
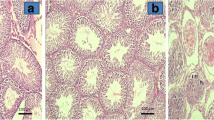
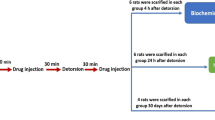
Fertility in patients treated for unilateral testicular torsion has been shown to be significantly reduced in all the reported series to date, implying that the present-day treatment requires further refinement in the form of adjunct pharmacotherapeutic intervention (Lomodex and MgSO4) in addition to scrotal exploration. Prepubertal Holtzman strain rats (35 days old) were used for our study. Two sets were formed with six groups of rats in each set. Rats were treated as follows: group 1, sham-operated group; group 2, torsion (4 h); group 3, torsion + detorsion (1 h); group 4, torsion + ATP–MgCl2 + detorsion; group 5, torsion + Lomodex–MgSO4 + detorsion; group 6, torsion + normal saline + detorsion. Whereas the first set of animals was sacrificed immediately at the end of experiment, animals in set 2 were sacrificed 8 weeks after the end of the experiment to look for the development of antisperm antibodies. Parameters studied were thiobarbituric acid reductase (TBAR) assay, histology of testicular tissue, and sperm agglutination test. Student’s t-test was used for significance. With detorsion (149.95±30.68) there was a significant rise in the TBAR values (P<0.05) compared with torsion (57.39±14.47). Treatment with both Lomodex–MgSO4 (40.74±6.39) and ATP–MgCl2 (48.30±18.35) yielded TBAR levels comparable to those in the sham group (31.35±11.96). Similar injury was also seen on the contralateral testis, with detorsion (114.28±10.68) much more detrimental than torsion (40.59±15.02) and rescue seen following treatment with Lomodex–MgSO4 (27.55±8.64) as well as ATP–MgCl2 (38.61±12.23). Regarding th histology, with detorsion there was evidence of severe distortion of tubules, with almost all the tubules showing maturation arrest and a few tubules completely devoid of any germinal cells. Treatment with Lomodex–MgSO4 as well as ATP–MgCl2 showed preservation of tubular morphology. Our study failed to document the presence of agglutinating antibodies (antisperm antibodies) in any of the groups. Unilateral testicular torsion has bilateral effects and is a form of ischemia–reperfusion injury. Treatment of torsion by detorsion alone does not prevent testicular damage. The results of the present study show that administration of Lomodex + MgSO4 prior to detorsion results in prolonged testicular salvage with a potential of subsequent improvement in semen quality and fertility and reduction in long-term morbidity. The presence of agglutinating antibodies could not be detected in the present study.


Similar content being viewed by others
References
Harrison RG, De Marval MJM (1981) Mechanism of damage to the contralateral testis in rats with an ischaemic testis. Lancet 2:723–725
Krarup T (1978) The testes after torsion. Br J Urol 50:43–46
Hagen P, Buchholz MM, Eigenmann J, Bandhauer K (1992) Testicular dysplasia causing disturbances of spermiogenesis in patients with unilateral torsion of the testis. Urol Int 49:154
Schutte B, Becker H, Vydra G (1986) Exocrine and endocrine testicular function following unilateral testicular torsion—a retrospective clinical study of 36 patients. Urologe A 25:142–146
Ryan PC, Whelan CA, Gaffney EF, et al. (1988) The effect of unilateral testicular torsion on spermatogenesis and fertility. Br J Urol 62:359–366
Anderson L, Brasso K, Kay L, et al. (1995) Late sequelae after testicular torsion. Ugeskr Laeger 157:170–172
Abes M, Sarihan H (2001) The effect of ATP–MgCl2 on the prevention of reperfusion injury after unilateral testicular torsion. Eur J Paediatr Surg 11:255–258
Gennaro A, Tsirigotis M, et al. (1985) Remington’s pharmaceutical sciences, 17th edn. Mack Publishing, pp 820–821
Akgur FM, Kilinc K (1993) Reperfusion injury after detorsion of unilateral torsion. Urol Res 21:395–399
Akgur FM, Klinic K, et al. (1994) The effect of allopurinol pretreatment before detorsing testicular torsion. J Urol 151:1715
Greenstein A, Smith-Harrison LI (1992) The effect of polyethylene glycol superoxide dismutase administration on the histological damage following spermatic cord torsion. J Urol 148:639–641
Filho DW, Torres MA, et al. (2004) Spermatic cord torsion, reactive oxygen and nitrogen species and ischemia–reperfusion injury. Mol Aspects Med 25):199–210
Oh SJ, Kwak C, et al. (2004) Histologic and molecular changes in the ipsilateral and contralateral epididymides of the rat in response to unilateral testicular torsion followed by detorsion. Fertil Steril 81(suppl):882–887
Ambrosio G, Flaherty JT, et al. (1991) Oxygen radicals generated at reflow induce peroxidation of membrane lipids in reperfused hearts. J Clin Invest 87:2056
Goldberg M, Serafin D, et al. (1990) Quantification of neutrophil adhesion to skeletal muscle venules following ischemia–reperfusion. J Reconstr Microsurg 6:267
Werns SW, Lucchesi BR (1990) Free radicals and ischemic tissue injury. Trends Pharmacol Sci 11:161
Saba M, Morales CR, Lamirande ED, Gagnon C (1997) Morphological and biochemical changes following acute unilateral testicular torsion in prepubertal rats. J Urol 157:1149–1154
Romeo C, Antonuccio P, et al. (2004) Raxofelast, a hydrophilic vitamin-E like antioxidant, reduces testicular ischemia–reperfusion injury. Urol Res 32:367–371
Ros CTD, Teloken C, et al. (1998) Does intratesticular testosterone administration modify the evolution of transitory testicular ischemia in prepubertal rats? J Urol 159:1752–1754
Palmer JS, Cromie WJ, et al. (1997) A platelet activating factor antagonist attenuates the effects of testicular ischemia. J Urol 158:1186–1190
Abas iyanik A, Dagdonderen L (2004) Beneficial effects of melatonin compared with allopurinol in experimental testicular torsion. J Pediatr Surg 39:1238–1241
Termeer CC, Weiss JM, Schopf E, et al. (1998) The low molecular weight Dextran 40 inhibits the adhesion of T lymphocytes to endothelial cells. Clin Exp Immunol 114:422–426
Pribush A, Mankuta D, Meiselman HJ, et al. (2000) The effect of low molecular weight dextran on erythrocyte aggregation in normal and preeclamptic pregnancy. Clin Hemorheol Microcirc 22:143–152
Laumonier T, Mohacsi PJ, et al.(2004) Endothelial cell protection by dextran sulfate: a novel strategy to prevent acute vascular rejection in xenotransplantation. Am J Transplant 4:181–187
Daffa SR, Milaat WA (2002) Role of magnesium sulfate in treatment of severe persistent pulmonary hypertension of the newborn. Saudi Med J 23:1266–1269
Feng DF, Zhu Za, Lu YC (2004) Effects of magnesium sulfate on traumatic brain edema in rats. Chin J Traumatol 7:148–152
Chung SY, Lin JY, Lin MC, et al. (2004) Synergistic efficacy of magnesium sulfate and FK506 on cerebral ischemia-induced infarct volume in gerbil. Med Sci Monit 10:BR105–BR108
Rantone TH, Gronlund JU, et al. (2002) Comparison of the effects of antenatal magnesium sulfate and ritodrine exposure on circulatory adaptation in preterm infants. Clin Physiol Funct Imaging 22:13–17
Acknowledgements
The authors thank Dr. C.P. Puri, director of NIRRH, for permission to carry out the study and for his encouragement. The authors gratefully acknowledge the technical assistance of Mr. M.S. Kadam, Mr. Ravi Kadam, Mrs. Jacintha Pereira, and the animal house staff at NIRRH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Adivarekar, P.K., Bhagwat, S.S., Raghavan, V. et al. Effect of Lomodex–MgSO4 in the prevention of reperfusion injury following unilateral testicular torsion: an experimental study in rats. Ped Surgery Int 21, 184–190 (2005). https://doi.org/10.1007/s00383-004-1317-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-004-1317-1