Abstract
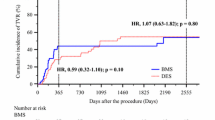
The impact of drug-coated balloon (DCB) on hemodialysis (HD) patients with coronary lesions remains unclear. This study aimed to compare outcomes after DCB treatment between HD and non-HD patients with de novo coronary lesions. A total of 235 consecutive patients who electively underwent DCB treatment for de novo coronary lesions were included (HD group: n = 100; non-HD group: n = 135). Angiographic follow-up was performed 6 months after the procedure. Patients were clinically followed up for 2 years. The incidence rates of target lesion revascularization (TLR) and major adverse cardiac events (MACE) were investigated. Diabetes and a history of coronary bypass grafting were more frequent in the HD group than in the non-HD group (69.0% vs. 50.7%, p = 0.007, and 24.0% vs 9.1%, p = 0.013, respectively). The reference diameter and pre-procedural diameter stenosis were greater in the HD group than in the non-HD group (2.49 mm vs. 2.24 mm, p = 0.007, and 65.9% vs. 59.6%, p = 0.015, respectively). Calcification was observed in 65.5% of all lesions, and rotational atherectomy was performed in 30.2% patients. The average diameter of the DCB was 2.51 mm (2.57 mm, HD group vs. 2.47 mm, non-HD group, p = 0.14). Although post-procedural diameter stenosis was similar between the groups, late lumen loss on follow-up angiography was larger in HD patients than in non-HD patients (0.27 mm vs. − 0.03 mm, p = 0.0009). The 2-year rates of freedom from TLR and MACE were lower in HD patients than in non-HD patients [79.3% vs. 91.7%, hazard ratio (HR) 2.76, 95% confidence interval (CI) 1.23–6.77, p = 0.014; and 61.6% vs. 89.4%, HR 4.60, 95% CI 2.30–10.2, p < 0.001, respectively]. In conclusion, the rates of TLR and MACE after DCB treatment were higher in HD patients than in non-HD patients.


Similar content being viewed by others
References
Herzog CA, Ma JZ, Collins AJ (1998) Poor long-term survival after acute myocardial infarction among patients on long-term dialysis. N Engl J Med 339:799–805
Parfrey PS, Foley RN (1999) The clinical epidemiology of cardiac disease in chronic renal failure. J Am Soc Nephrol 10:1606–1615
Cheung AK, Sarnak MJ, Yan G, Dwyer JT, Heyka RJ, Rocco MV, Teehan BP, Levey AS (2000) Atherosclerotic cardiovascular disease risks in chronic hemodialysis patients. Kidney Int 58:353–362
Herzog CA, Ma JZ, Collins AJ (2002) Comparative survival of dialysis patients in the United States after coronary angioplasty, coronary artery stenting, and coronary artery bypass surgery and impact of diabetes. Circulation 106:2207–22011
Otsuka Y, Ishiwata S, Inada T, Kanno H, Kyo E, Hayashi Y, Fujita H, Michishita I (2011) Comparison of haemodialysis patients and non-haemodialysis patients with respect to clinical characteristics and 3-year clinical outcomes after sirolimus-eluting stent implantation: insights from the Japan multi-centre post-marketing surveillance registry. Eur Heart J 32:829–837
Moses JW, Leon MB, Popma JJ, Fitzgerald PJ, Holmes DR, O’Shaughnessy C, Caputo RP, Kereiakes DJ, Williams DO, Teirstein PS, Jaeger JL, Kuntz RE, Investigators SIRIUS (2003) Sirolimus-eluting stents versus standard stents in patients with stenosis in a native coronary artery. N Engl J Med 349:1315–1323
Stone GW, Moses JW, Ellis SG, Schofer J, Dawkins KD, Morice MC, Colombo A, Schampaert E, Grube E, Kirtane AJ, Cutlip DE, Fahy M, Pocock SJ, Mehran R, Leon MB (2007) Safety and efficacy of sirolimus- and paclitaxel-eluting coronary stents. N Engl J Med 356:998–1008
Stone GW, Moses JW, Ellis SG, Schofer J, Dawkins KD, Morice MC, Colombo A, Schampaert E, Grube E, Kirtane AJ, Cutlip DE, Fahy M, Pocock SJ, Mehran R, Leon MB (2008) Comparison of an everolimus-eluting stent and a paclitaxel-eluting stent in patients with coronary artery disease: a randomized trial. JAMA 299:1903–1913
Stone GW, Rizvi A, Sudhir K, Newman W, Applegate RJ, Cannon LA, Maddux JT, Cutlip DE, Simonton CA, Sood P, Kereiakes DJ, Investigators SPIRITIV (2011) Randomized comparison of everolimus- and paclitaxel-eluting stents. 2-year follow-up from the SPIRIT (clinical evaluation of the XIENCE V everolimus eluting coronary stent system) IV trial. J Am Coll Cardiol 58:19–25
Bangalore S, Kumar S, Fusaro M, Amoroso N, Attubato MJ, Feit F, Bhatt DL, Slater J (2012) Short- and long-term outcomes with drug-eluting and bare-metal coronary stents: a mixed-treatment comparison analysis of 117 762 patient-years of follow-up from randomized trials. Circulation 125:2873–2891
Nakazawa G, Tanabe K, Aoki J, Yamamoto H, Higashikuni Y, Onuma Y, Yachi S, Nakajima H, Hara K (2007) Impact of renal insufficiency on clinical and angiographic outcomes following percutaneous coronary intervention with sirolimus-eluting stents. Catheter Cardiovasc Interv 69:808–814
Ichimoto E, Kobayashi Y, Iijima Y, Kuroda N, Kohno Y, Komuro I (2010) Long-term clinical outcomes after sirolimus-eluting stent implantation in dialysis patients. Int Heart J 51:92–97
Ikari Y, Kyono H, Isshiki T, Ishizuka S, Nasu K, Sano K, Okada H, Sugano T, Uehara Y (2015) Usefulness of everolimus-eluting coronary stent implantation in patients on maintenance hemodialysis. Am J Cardiol 116:872–876
Unverdorben M, Kleber FX, Heuer H, Figulla HR, Vallbracht C, Leschke M, Cremers B, Hardt S, Buerke M, Ackermann H, Boxberger M, Degenhardt R, Scheller B (2010) Treatment of small coronary arteries with a paclitaxel-coated balloon catheter. Clin Res Cardiol 99:165–174
Wöhrle J, Zadura M, Möbius-Winkler S, Leschke M, Opitz C, Ahmed W, Barragan P, Simon JP, Cassel G, Scheller B (2012) SeQuentPlease World Wide Registry: clinical results of SeQuent please paclitaxel-coated balloon angioplasty in a large-scale, prospective registry study. J Am Coll Cardiol 60:1733–1738
Zeymer U, Waliszewski M, Spiecker M, Gastmann O, Faurie B, Ferrari M, Alidoosti M, Palmieri C, Heang TN, Ong PJ, Dietz U (2014) Prospective “real world” registry for the use of the “PCB only” strategy in small vessel de novo lesions. Heart 100:311–316
Unverdorben M, Kleber FX, Heuer H, Figulla HR, Vallbracht C, Leschke M, Cremers B, Hardt S, Buerke M, Ackermann H, Boxberger M, Degenhardt R, Scheller B (2013) Treatment of small coronary arteries with a paclitaxel-coated balloon catheter in the PEPCAD I study: are lesions clinically stable from 12 to 36 months? EuroIntervention 9:620–628
Mintz GS, Popma JJ, Pichard AD, Kent KM, Satler LF, Chuang YC, Ditrano CJ, Leon MB (1995) Patterns of calcification in coronary artery disease. A statistical analysis of intravascular ultrasound and coronary angiography in 1155 lesions. Circulation 91:1959–1965
Ryan TJ, Faxon DP, Gunnar RM, Kennedy JW, King SB 3rd, Loop FD, Peterson KL, Reeves TJ, Williams DO, Winters WL Jr (1988) Guidelines for percutaneous transluminal coronary angioplasty. A report of the American College of Cardiology/American Heart Association Task Force on Assessment of Diagnostic and Therapeutic Cardiovascular Procedures (subcommittee on percutaneous transluminal coronary angioplasty). Circulation 78:486–502
Kleber FX, Rittger H, Bonaventura K, Zeymer U, Wöhrle J, Jeger R, Levenson B, Möbius-Winkler S, Bruch L, Fischer D, Hengstenberg C, Pörner T, Mathey D, Scheller B (2013) Drug-coated balloons for treatment of coronary artery disease: updated recommendations from a consensus group. Clin Res Cardiol 102:785–797
Ishii H, Toriyama T, Aoyama T, Takahashi H, Tanaka M, Yoshikawa D, Hayashi M, Yasuda Y, Maruyama S, Matsuo S, Matsubara T, Murohara T (2012) Percutaneous coronary intervention with bare metal stent vs. drug-eluting stent in hemodialysis patients. Circ J 76:1609–1615
Venetsanos D, Lawesson SS, Panayi G, Tödt T, Berglund U, Swahn E, Alfredsson J (2018) Long-term efficacy of drug coated balloons compared with new generation drug-eluting stents for the treatment of de novo coronary artery lesions. Catheter Cardiovasc Interv 92:E317–E326
Kaw D, Malhotra D (2006) Platelet dysfunction and end-stage renal disease. Semin Dial 19:317–322
Rios DR, Carvalho Md, Lwaleed BA, Simões e Silva AC, Borges KB, Dusse LM, (2010) Hemostatic changes in patients with end stage renal disease undergoing hemodialysis. Clin Chim Acta 411:135–139
Mintz GS, Weissman NJ (2006) Intravascular ultrasound in the drug-eluting stent era. J Am Coll Cardiol 48:421–429
Kang SJ, Mintz GS, Park DW, Lee SW, Kim YH, Whan Lee C, Han KH, Kim JJ, Park SW, Park SJ (2011) Mechanisms of in-stent restenosis after drug-eluting stent implantation: intravascular ultrasound analysis. Circ Cardiovasc Interv 4:9–14
Uren NG, Schwarzacher SP, Metz JA, Lee DP, Honda Y, Yeung AC, Fitzgerald PJ, Yock PG, Registry Investigators POST (2002) Predictors and outcomes of stent thrombosis: an intravascular ultrasound registry. Eur Heart J 23:124–132
Ito R, Ueno K, Yoshida T, Takahashi H, Tatsumi T, Hashimoto Y, Kojima Y, Kitamura T, Morita N (2018) Outcomes after drug-coated balloon treatment for patients with calcified coronary lesions. J Interv Cardiol 31:436–441
Rissanen TT, Uskela S, Siljander A, Kärkkäinen JM, Mäntylä P, Mustonen J, Eränen J (2017) Percutaneous coronary intervention of complex calcified lesions with drug-coated balloon after rotational atherectomy. J Interv Cardiol 30:139–146
Ueno K, Morita N, Kojima Y, Takahashi H, Kawasaki M, Ito R, Kondo H, Minatoguchi S, Yoshida T, Hashimoto Y, Tatsumi T, Kitamura T (2019) Safety and long-term efficacy of drug-coated balloon angioplasty following rotational atherectomy for severely calcified coronary lesions compared with new generation drug-eluting stents. J Interv Cardiol 2019:9094178
Jeger RV, Eccleshall S, Wan Ahmad WA, Ge J, Poerner TC, Shin ES, Alfonso F, Latib A, Ong PJ, Rissanen TT, Saucedo J, Scheller B, Kleber FX, International DCB Consensus Group (2020) Drug-coated balloons for coronary artery disease: third report of the International DCB Consensus Group. JACC Cardiovasc Interv 13:1391–1402
Takahashi R, Ito Y, Takahashi H, Ishii H, Kasuga H, Mizuno M, Suzuki Y, Yuzawa Y, Maruyama S, Murohara T, Imai E, Matsuo S (2012) Combined values of serum albumin, C-reactive protein and body mass index at dialysis initiation accurately predicts long-term mortality. Am J Nephrol 36:136–143
Takahashi H, Ito Y, Ishii H, Aoyama T, Kamoi D, Kasuga H, Yasuda K, Maruyama S, Matsuo S, Murohara T, Yuzawa Y (2014) Geriatric nutritional risk index accurately predicts cardiovascular mortality in incident hemodialysis patients. J Cardiol 64:32–36
Author information
Authors and Affiliations
Contributions
RI and TN conceptualization. SO and TS methodology. HI and MK analysis and investigation. RI writing—original draft preparation. TM supervision.
Corresponding author
Ethics declarations
Conflict of interest
H.I. received lecture fees from Astellas Pharma, AstraZeneca, Bayer Pharmaceutical Co.Ltd., Chugai Pharma Inc., Daiichi-Sankyo Co., Ltd., and MSD K. K. T.M. received lecture fees from Bayer Pharmaceutical Co., Ltd., Daiichi-Sankyo Co., Ltd., Dainippon Sumitomo Pharma Co., Ltd., Kowa Co., Ltd., MSD K. K., Mitsubishi Tanabe Pharma Co., Nippon Boehringer Ingelheim Co., Ltd., Novartis Pharma K. K., Pfizer Japan Inc., Sanofi-Aventis K. K., and Takeda Pharmaceutical Co., Ltd. For the remaining authors, no conflicts are declared.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ito, R., Ishii, H., Oshima, S. et al. Outcomes after drug-coated balloon interventions for de novo coronary lesions in the patients on chronic hemodialysis. Heart Vessels 36, 1646–1652 (2021). https://doi.org/10.1007/s00380-021-01858-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-021-01858-3