Abstract
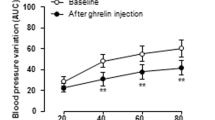
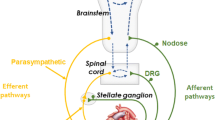
Vagal nerve stimulation has been postulated to confer an antifibrillatory effect. We studied whether ghrelin administration would exert an antiarrhythmic effect via modulation of autonomic nerve activity in rats after acute myocardial ischemia (MI). Male Sprague-Dawley rats were exposed to 30 min of ischemia following ligation of the left coronary artery. Animals were then randomized to receive either ghrelin (n = 26) or saline (n = 26) during the period of coronary ligation. Power spectral analysis of heart-rate variability revealed that the administration of ghrelin increased the high-frequency (HF) power and decreased the low-frequency (LF)/HF ratio. Ventricular tachyarrhythmias were less frequent in rats after MI who received ghrelin in comparison with rats that received saline. Immunoblotting and immunohistochemistry revealed that rats given saline alone during MI exhibited a marked reduction in phosphorylated connexin-43 within the left ventricle, whereas those that received ghrelin displayed only minor reductions in comparison with sham-operated rats. These effects of ghrelin were diminished by the coadministration of atropine or the blockade of vagal afferents. These data demonstrate that the beneficial effect of ghrelin might be mediated by modulation of cardiac autonomic nerve activity.






Similar content being viewed by others
References
Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K (1999) Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 402:656–660
Nakazato M, Murakami N, Date Y, Kojima M, Matsuo H, Kangawa K, Matsukura S (2001) A role for ghrelin in the central regulation of feeding. Nature 409:194–198
Kishimoto I, Tokudome T, Schwenke DO, Soeki T, Hosoda H, Nagaya N, Kangawa K (2009) Therapeutic potential of ghrelin in cardiac diseases. Expert Rev Endocrinol Metab 4:283–289
Soeki T, Kishimoto I, Schwenke DO, Tokudome T, Horio T, Yoshida M, Hosoda H, Kangawa K (2008) Ghrelin suppresses cardiac sympathetic activity and prevents early left ventricular remodeling in rats with myocardial infarction. Am J Physiol Heart Circ Physiol 294:H426–H432
Schwenke DO, Tokudome T, Kishimoto I, Horio T, Shirai M, Cragg PA, Kangawa K (2008) Early ghrelin treatment after myocardial infarction prevents an increase in cardiac sympathetic tone and reduces mortality. Endocrinology 149:5172–5176
Sternick EB, Piorkowski C, Hindricks G, Dagres N, Sommer P (2011) Electrical storm originating from a left ventricular epicardial scar in a patient with completely normal endocardial voltage. Heart Vessels 26:663–666
Abulaiti A, Hu D, Zhu D, Zhang R (2011) Influence of fastigial nucleus stimulation on heart rate variability of surgically induced myocardial infarction rats: fastigial nucleus stimulation and autonomous nerve activity. Heart Vessels 26:654–662
Ando M, Katare RG, Kakinuma Y, Zhang D, Yamasaki F, Muramoto K, Sato T (2005) Efferent vagal nerve stimulation protects heart against ischemia-induced arrhythmias by preserving connexin43 protein. Circulation 112:164–170
Jiang H, Hu X, Lu Z, Wen H, Zhao D, Tang Q, Yang B (2008) Effects of sympathetic nerve stimulation on ischemia-induced ventricular arrhythmias by modulating connexin43 in rats. Arch Med Res 39:647–654
Roell W, Lewalter T, Sasse P, Tallini YN, Choi BR, Breitbach M, Doran R, Becher UM, Hwang SM, Bostani T, von Maltzahn J, Hofmann A, Reining S, Eiberger B, Gabris B, Pfeifer A, Welz A, Willecke K, Salama G, Schrickel JW, Kotlikoff MI, Fleischmann BK (2007) Engraftment of connexin 43-expressing cells prevents post-infarct arrhythmia. Nature 450:819–824
Date Y, Murakami N, Toshinai K, Matsukura S, Niijima A, Matsuo H, Kangawa K, Nakazato M (2002) The role of the gastric afferent vagal nerve in ghrelin-induced feeding and growth hormone secretion in rats. Gastroenterology 123:1120–1128
Matsumura K, Tsuchihashi T, Fujii K, Abe I, Iida M (2002) Central ghrelin modulates sympathetic activity in conscious rabbits. Hypertension 40:694–699
Lin Y, Matsumura K, Fukuhara M, Kagiyama S, Fujii K, Iida M (2004) Ghrelin acts at the nucleus of the solitary tract to decrease arterial pressure in rats. Hypertension 43:977–982
Vanoli E, De Ferrari GM, Stramba-Badiale M, Hull SS Jr, Foreman RD, Schwartz PJ (1991) Vagal stimulation and prevention of sudden death in conscious dogs with a healed myocardial infarction. Circ Res 68:1471–1481
Li M, Zheng C, Sato T, Kawada T, Sugimachi M, Sunagawa K (2004) Vagal nerve stimulation markedly improves long-term survival after chronic heart failure in rats. Circulation 109:120–124
Beardslee MA, Lerner DL, Tadros PN, Laing JG, Beyer EC, Yamada KA, Kléber AG, Schuessler RB, Saffitz JE (2000) Dephosphorylation and intracellular redistribution of ventricular connexin43 during electrical uncoupling induced by ischemia. Circ Res 87:656–662
Lerner DL, Yamada KA, Schuessler RB, Saffitz JE (2000) Accelerated onset and increased incidence of ventricular arrhythmias induced by ischemia in Cx43-deficient mice. Circulation 101:547–552
Gutstein DE, Morley GE, Tamaddon H, Vaidya D, Schneider MD, Chen J, Chien KR, Stuhlmann H, Fishman GI (2001) Conduction slowing and sudden arrhythmic death in mice with cardiac-restricted inactivation of connexin43. Circ Res 88:333–339
van Koppen CJ, Kaiser B (2003) Regulation of muscarinic acetylcholine receptor signaling. Pharmacol Ther 98:197–220
Liu H, McPherson BC, Zhu X, Da Costa ML, Jeevanandam V, Yao Z (2001) Role of nitric oxide and protein kinase C in ACh-induced cardioprotection. Am J Physiol Heart Circ Physiol 281:H191–H197
Shimada M, Terada T (2002) Roles of cAMP in regulation of both MAP kinase and p34(cdc2) kinase activity during meiotic progression, especially beyond the MI stage. Mol Reprod Dev 62:124–131
Hervé JC, Plaisance I, Loncarek J, Duthe F, Sarrouilhe D (2004) Is the junctional uncoupling elicited in rat ventricular myocytes by some dephosphorylation treatments due to changes in the phosphorylation status of Cx43? Eur Biophys J 33:201–210
Lampe PD, Lau AF (2004) The effects of connexin phosphorylation on gap junctional communication. Int J Biochem Cell Biol 36:1171–1186
Nagaya N, Kangawa K (2003) Ghrelin improves left ventricular dysfunction and cardiac cachexia in heart failure. Curr Opin Pharmacol 3:146–151
Baldanzi G, Filigheddu N, Cutrupi S, Catapano F, Bonissoni S, Fubini A, Malan D, Baj G, Granata R, Broglio F, Papotti M, Surico N, Bussolino F, Isgaard J, Deghenghi R, Sinigaglia F, Prat M, Muccioli G, Ghigo E, Graziani A (2002) Ghrelin and des-acyl ghrelin inhibit cell death in cardiomyocytes and endothelial cells through ERK1/2 and PI 3-kinase/AKT. J Cell Biol 159:1029–1037
Iglesias MJ, Pineiro R, Blanco M, Gallego R, Dieguez C, Gualillo O, Gonzalez-Juanatey JR, Lago F (2004) Growth hormone releasing peptide (ghrelin) is synthesized and secreted by cardiomyocytes. Cardiovasc Res 62:481–488
Chang L, Ren Y, Liu X, Li WG, Yang J, Geng B, Weintraub NL, Tang C (2004) Protective effects of ghrelin on ischemia/reperfusion injury in the isolated rat heart. J Cardiovasc Pharmacol 43:165–170
Acknowledgments
The authors thank Kazue Ishikawa for her assistance and contributions to the study. This study was supported by research grants from the Japan Society for the Promotion of Science (JSPS) and a Grant-in-Aid for Scientific Research (C) [JSPS (C)-21590899]. All authors have reported that they have no relationships to disclose relevant to the content of this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Soeki, T., Niki, T., Uematsu, E. et al. Ghrelin protects the heart against ischemia-induced arrhythmias by preserving connexin-43 protein. Heart Vessels 28, 795–801 (2013). https://doi.org/10.1007/s00380-013-0333-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-013-0333-2