Abstract
Soil aggregation contributes to soil organic C (SOC) sequestration, but it is still unclear how the organic functional groups and microbial necromass operate to influence soil aggregation. This issue was explored after 33 years of manure (i.e. farmyard manure at agronomic and elevated rates) and chemical fertilizer application in a Calcic Cambisol on the Loess Plateau. The application of manure increased SOC and the mass proportion of macroaggregates by 1.1- to 1.8-fold and 1.5- to 2.3-fold, respectively, compared with the unfertilized control. The aliphaticity, aromaticity and hydrophobicity indices of SOC as characterized by 13C nuclear magnetic resonance spectroscopy were higher in the manure-amended soils than in the chemical fertilizer-treated soils. The application of manure increased the microbial necromass by 1.3- to 2.6-fold compared with the control. Fungal necromass was enriched in the macroaggregates, while bacterial necromass was enriched in the microaggregates. Compared with the control, manure application increased (P < 0.05) the relative contribution of fungal necromass to SOC, which was positively correlated with the chemical recalcitrance of the SOC (i.e. aliphaticity, aromaticity and hydrophobicity) (P < 0.05). Furthermore, the chemical recalcitrance and relative contribution of microbial necromass, particularly that of fungal necromass, in the bulk soil (P < 0.05) and soil aggregates (P < 0.01) were positively correlated with the mass proportion of macroaggregates. Therefore, the accumulation of recalcitrant C components and fungal necromass contributes to soil aggregation in the Calcic Cambisols of the Loess Plateau.





Similar content being viewed by others
References
Abiven S, Menasseri S, Chenu C (2009) The effects of organic inputs over time on soil aggregate stability-a literature analysis. Soil Biol Biochem 41:1–12
Amelung W, Brodowski S, Sandhage-Hofmann A, Bol R (2008) Combining biomarker with stable isotope analyses for assessing the transformation and turnover of soil organic matter. Adv Agron 100:155–250
Appuhn A, Joergensen RG (2006) Microbial colonisation of roots as a function of plant species. Soil Biol Biochem 38:1040–1051
Bach EM, Williams RJ, Hargreaves SK, Yang F, Hofmockel KS (2018) Greatest soil microbial diversity found in micro-habitats. Soil Biol Biochem 118:217–226
Banfield CC, Pausch J, Kuzyakov Y, Dippold MA (2018) Microbial processing of plant residues in the subsoil-The role of biopores. Soil Biol Biochem 125:309–308
Bao SD (2000) Soil and agricultural chemistry analysis. Chinese Agricultural Press, Beijing, China (in Chinese)
Bayer C, Martin-Neto L, Mielniczuk J, Dieckow J, Amado TJC (2006) C and N stocks and the role of molecular recalcitrance and organomineral interaction in stabilizing soil organic matter in a subtropical Acrisol managed under no-tillage. Geoderma 133:258–268
Chen RR, Senbayram M, Blagodatsky S, Myachina O, Dittert K, Lin XG, Blagodatskaya E, Kuzyakov Y (2014) Soil C and N availability determine the priming effect: microbial N mining and stoichiometric decomposition theories. Glob Chang Biol 20:2356–2367
Chen XB, Xia YH, Rui YC, Ning Z, Hu YJ, Tang HM, He HB, Li HX, Kuzyakov Y, Ge TD, Wu JS, Su YR (2020) Microbial carbon use efficiency, biomass turnover, and necromass accumulation in paddy soil depending on fertilization. Agric Ecosyst Environ 292:106816
Chen XB, Hu YJ, Xia YH, Zheng SM, Ma C, Rui YC, He HB, Huang DY, Zhang ZH, Ge TD, Wu JS, Guggenberger G, Kuzyakov Y, Su YR (2021) Contrasting pathways of carbon sequestration in paddy and upland soils. Glob Chang Biol 27:2478–2490
Chenu C, Hassink J, Bloem J (2001) Short-term changes in the spatial distribution of microorganisms in soil aggregates as affected by glucose addition. Biol Fertil Soils 34:349–356
Deng SH, Zheng XD, Chen XB, Zheng SM, He XY, Ge TD, Kuzyakov Y, Wu JS, Su YR, Hu YJ (2021) Divergent mineralization of hydrophilic and hydrophobic organic substrates and their priming effect in soils depending on their preferential utilization by bacteria and fungi. Biol Fertil Soils 57:65–76
Ding XL, Han XZ, Zhang XD (2013) Long-term impacts of manure, straw, and fertilizer on amino sugars in a silty clay loam soil under temperate conditions. Biol Fertil Soils 49:949–954
Ding XL, Liang C, Zhang B, Yuan YR, Han XZ (2015) Higher rates of manure application lead to greater accumulation of both fungal and bacterial residues in macroaggregates of a clay soil. Soil Biol Biochem 84:137–146
Ding XL, Han XZ (2014) Effects of long-term fertilization on contents and distribution of microbial residues within aggregate structures of a clay soil. Biol Fertil Soils 50:549–554
Dou S, Zhang JJ, Li K (2008) Effect of organic matter applications on 13C-NMR spectra of humic acids of soil. Eur J Soil Sci 59:532–539
Elliott ET (1986) Aggregate structure and carbon, nitrogen, and phosphorus in native and cultivated soils. Soil Sci Soc Am J 50:627–633
Fernandez CW, Koide RT (2014) Initial melanin and nitrogen concentrations control the decomposition of ectomycorrhizal fungal litter. Soil Biol Biochem 77:150–157
Fernández-Ugalde O, Virto I, Barré P, Gartzia-Bengoetxea N, Enrique A, Imaz MJ, Bescansa P (2011) Effect of carbonates on the hierarchical model of aggregation in calcareous semi-arid Mediterranean soils. Geoderma 164:203–214
Gandois L, Perrin AS, Probst A (2011) Impact of nitrogenous fertiliser-induced proton release on cultivated soils with contrasting carbonate contents: a column experiment. Geochim Cosmochim Acta 75:1185–1198
Gunina A, Kuzyakov Y (2015) Sugars in soil and sweets for microorganisms: review of origin, content, composition and fate. Soil Biol Biochem 90:87–100
Huang XL, Jia ZX, Guo JJ, Li TL, Sun DS, Meng HS, Yu GH, He XH, Ran W, Zhang SS, Hong JP, Shen QR (2019) Ten-year long-term organic fertilization enhances carbon sequestration and calcium-mediated stabilization of aggregate-associated organic carbon in a reclaimed Cambisol. Geoderma 355:113880
Huang XL, Jia ZX, Jiao XY, Wang JL, Huang XF (2022) Long-term manure applications to increase carbon sequestration and macroaggregate-stabilized carbon. Soil Biol Biochem 174:108827
IUSS Working Group WRB, 2015. World Reference Base for Soil Resources 2014, update 2015. International soil classification system for naming soils and creating legends for soil maps. World Soil Resources Reports No. 106. FAO, Rome
Jiang X, Wright AL, Wang X, Liang F (2011) Tillage-induced changes in fungal and bacterial biomass associated with soil aggregates: a long-term field study in a subtropical rice soil in China. Appl Soil Ecol l48:168–173
Joergensen RG (1996) The fumigation-extraction method to estimate soil microbial biomass: calibration of the kEC value. Soil Biol Biochem 28:25–31
Joergensen RG (2018) Amino sugars as specific indices for fungal and bacterial residues in soil. Biol Fertil Soils 54:559–568
Joergensen RG, Mueller T (1996) The fumigation-extraction method to estimate soil microbial biomass: calibration of the kEN value. Soil Biol Biochem 28:33–37
Jouany C (1991) Surface free energy components of clay-synthetic humic acid complexes from contact-angle measurements. Clay Clay Miner 39:43–49
Kästner M, Miltner A, Thiele-Bruhn S, Liang C (2021) Microbial necromass in soils-linking microbes to soil processes and carbon turnover. Front Environ Sci 9:756378
Lehmann J, Kleber M (2015) The contentious nature of soil organic matter. Nature 528:60–68
Li YC, Li YF, Chang SX, Liang X, Qin H, Chen JH, Xu QF (2017) Linking soil fungal community structure and function to soil organic carbon chemical composition in intensively managed subtropical bamboo forests. Soil Biol Biochem 107:19–31
Liang C, Balser TC (2010) Microbial production of recalcitrant organic matter in global soils: implications for productivity and climate policy. Nat Rev Microbiol 9:75
Liang C, Schimel JP, Jastrow JD (2017) The importance of anabolism in microbial control over soil carbon storage. Nat Microbiol 2:17105
Liang C, Amelung W, Lehmann J, Kastner M (2019) Quantitative assessment of microbial necromass contribution to soil organic matter. Glob Chang Biol 25:3578–3590
Liao HP, Lu XM, Rensing C, Friman VP, Geisen S, Chen Z, Yu Z, Wei Z, Zhou SG, Zhu YG (2018) Hyperthermophilic composting accelerates the removal of antibiotic resistance genes and mobile genetic elements in sewage sludge. Environ Sci Technol 52:266–276
Lützow M, Kögel-Knabner I, Ekschmitt K, Matzner E, Guggenberger G, Marschner B, Flessa H (2006) Stabilization of organic matter in temperate soils: mechanisms and their relevance under different soil conditions-a review. Eur J Soil Sci 57:426–445
Ma T, Zhu SS, Wang ZH, Chen DM, Dai GH, Feng BW, Su XY, Hu HF, Li KH, Han WX, Liang C, Bai YF, Feng XJ (2018) Divergent accumulation of microbial necromass and plant lignin components in grassland soils. Nat Commun 9:3480
McCarthy JF, Ilavsky J, Jastrow JD, Mayer LM, Perfect E, Zhuang J (2008) Protection of organic carbon in soil microaggregates occur via restructuring of aggregate porosity and filling pores with accumulating organic matter. Geochim Cosmochim Acta 72:4725–4744
Ostertag R, Marín-Spiotta E, Silver WL, Schulten J (2008) Litterfall and decomposition in relation to soil carbon pools along a secondary forest chronosequence in Puerto Rico. Ecosystems 11:701–714
Rillig MC, Mummey DL (2006) Mycorrhizas and soil structure. New Phytol 171:41–53
Rowley MC, Grand S, Verrecchia EP (2018) Calcium-mediated stabilization of soil organic matter. Biogeochemistry 137:27–49
Said-Pullicino D, Kaiser K, Guggenberger G, Gigliotti G (2007) Changes in the chemical composition of water-extractable organic matter during composting: distribution between stable and labile organic matter pools. Chemosphere 66:2166–2176
Schmidt MWI, Knicker H, Hatcher PG, Kögel-Knabner I (1997) Improvement of 13C and 15N CPMAS NMR spectra of bulk soils, particle size fractions and organic material by treatment with 10% hydrofluoric acid. Eur J Soil Sci 48:319–328
Schmidt MWI, Torn MS, Abiven S, Dittmar T, Guggenberger G, Janssens IA, Kleber M, Kögel-Knabner I, Lehmann J, Manning DAC, Nannipieri P, Rasse DP, Weiner S, Trumbore SE (2011) Persistence of soil organic matter as an ecosystem property. Nature 478:49–56
Schweigert M, Herrmann S, Miltner A, Fester T, Kästner M (2015) Fate of ectomycorrhizal fungal biomass in a soil bioreactor system and its contribution to soil organic matter formation. Soil Biol Biochem 88:120–127
Six J, Elliott ET, Paustian K (2000) Soil macroaggregate turnover and microaggregate formation: a mechanism for C sequestration under no-tillage agriculture. Soil Biol Biochem 32:2099–2103
Six J, Bossuyt H, Degryze S, Denef K (2004) A history of research on the link between (micro)aggregates, soil biota, and soil organic matter dynamics. Soil Tillage Res 79:7–31
Sowers T, Adhikari D, Wang J, Yang Y, Sparks DL (2018) Spatial associations and chemical composition of organic carbon sequestered in Fe, Ca, and organic carbon ternary systems. Environ Sci Technol 52:6936–6944
Spaccini R, Mbagwu JSC, Conte P, Piccolo A (2006) Changes of humic substances characteristics from forested to cultivated soils in Ethiopia. Geoderma 132:9–19
Strickland MS, Rousk J (2010) Considering fungal:bacterial dominance in soils-methods, controls, and ecosystem implications. Soil Biol Biochem 42:1385–1395
Sun FS, Polizzotto ML, Guan DX, Wu J, Shen QR, Ran W, Wang BR, Yu GH (2017) Exploring the interactions and binding sites between Cd and functional groups in soil using two-dimensional correlation spectroscopy and synchrotron radiation based spectromicroscopies. J Hazard Mater 326:18–25
Tisdall JM, Oades JM (1982) Organic matter and water-stable aggregates in soils. J Soil Sci 33:141–163
Totsche KU, Amelung W, Gerzabek MH, Guggenberger G, Klumpp E, Knief C, Lehndorff E, Mikutta R, Peth S, Prechtel A (2018) Microaggregates in soils. J Plant Nutr Soil Sci 181:104–136
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass C. Soil Biol Biochem 19:703–707
Xia YH, Chen XB, Zheng XD, Deng SH, Hu YJ, Zheng SM, He XY, Wu JS, Kuzyakov Y, Su YR (2020) Preferential uptake of hydrophilic and hydrophobic compounds by bacteria and fungi in upland and paddy soils. Soil Biol Biochem 148:107879
Ye GP, Lin YX, Kuzyakov Y, Liu DY, Luo JF, Lindsey S, Wang WJ, Fan JB, Ding WX (2019) Manure over crop residues increases soil organic matter but decreases microbial necromass relative contribution in upland Ultisols: results of a 27-year field experiment. Soil Biol Biochem 134:15–24
Yu GH, Tang Z, Xu YC, Shen QR (2011) Multiple fluorescence labeling and two dimensional FTIR-13C NMR heterospectral correlation spectroscopy to characterize extracellular polymeric substances in biofilms produced during composting. Environ Sci Technol 45:9224–9231
Yu HY, Ding WX, Luo JF, Geng RL, Cai ZC (2012) Long-term application of organic manure and mineral fertilizers on aggregation and aggregate-associated carbon in a sandy loam soil. Soil Tillage Res 124:170–177
Yu GH, Xiao J, Hu SJ, Polizzotto ML, Zhao FJ, Mcgrath SP, Li H, Ran W, Shen QR (2017) Mineral availability as a key regulator of soil carbon storage. Environ Sci Technol 51:4960–4969
Zhang XD, Amelung W (1996) Gas chromatographic determination of muramic acid, glucosamine, mannosamine, and galactosamine in soils. Soil Biol Biochem 28:1201–1206
Zhang X, Amelung W, Yuan Y, Zech W, Samson-Liebig S, Brown L, Zech W (1999) Land-use effects on amino sugars in particle-size fractions of an Argiudoll. Appl Soil Ecol 11:271–275
Zhou P, Pan GX, Spaccini R, Piccolo A (2010) Molecular changes in particulate organic matter (POM) in a typical Chinese paddy soil under different long-term fertilizer treatments. Eur J Soil Sci 61:231–242
Acknowledgements
We are grateful to the editor-in-chief (Paolo Nannipieri) and the anonymous reviewers for their constructive comments.
Funding
This work was supported by the State Key Laboratory of Sustainable Dryland Agriculture (in preparation), Shanxi Agricultural University (grant no. 202001-8 and 202105D121008), the National Natural Science Foundation of China (grant no. 42277345 and 41907083) and the Distinguished and Excellent Young Scholar Cultivation Project of Shanxi Agricultural University (2022YQPYGC06).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information

Fig. S1
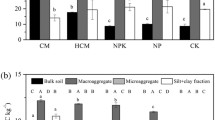
Annual mean crop yields as affected by different long-term (33 years) fertilization treatments from 1988 to 2020. The solid lines and circles, the lower and upper limits and bars outside the boxes represent median and mean values, 25th and 75th, 10th and 90th percentile yield values, respectively, while black squares above and below boxes represent, respectively, the < 10th and > 90th percentiles of data. Treatments CT, N, NP, M, NM, NPM, NDM and NPDM represent no fertilizer control, N fertilizer, N and P fertilizers, farmyard manure at an agronomic rate, N plus manure, NP plus manure, N plus double amounts of manure and NP plus double amounts of manure, respectively. Different letters indicate significant differences between treatments at P < 0.05 according to Duncan's multiple range test. (PNG 38 kb)

Fig. S2
Solid-state CPMAS 13C NMR spectroscopy of the farmyard manure. (PNG 15 kb)

Fig. S3
Effects of different long-term (33 years) fertilization treatments on the contents of glucosamine (a), muramic acid (b) and the ratio of glucosamine to muramic acid (c) in the bulk soil and soil aggregates. Treatments CT, N, NP, M, NM, NPM, NDM and NPDM represent no fertilizer control, N fertilizer, N and P fertilizers, farmyard manure at an agronomic rate, N plus manure, NP plus manure, N plus double amounts of manure and NP plus double amounts of manure, respectively. Error bars are standard errors of means (n = 3). Different capital and lowercase letters indicate significant differences between aggregate fractions within a fertilizer treatment and between fertilizer treatments within an aggregate fraction, respectively, at P < 0.05 according to Duncan's multiple range test. (PNG 81 kb)

Fig. S4
Effects of different long-term (33 years) fertilization treatments on the ratio of fungal necromass to bacterial necromass in the bulk soil and soil aggregates. Treatments CT, N, NP, M, NM, NPM, NDM and NPDM represent no fertilizer control, N fertilizer, N and P fertilizers, farmyard manure at an agronomic rate, N plus manure, NP plus manure, N plus double amounts of manure and NP plus double amounts of manure, respectively. Error bars are standard errors of means (n = 3). Different capital and lowercase letters indicate significant differences between aggregate fractions within a fertilizer treatment and between fertilizer treatments within an aggregate fraction, respectively, at P < 0.05 according to Duncan's multiple range test. (PNG 36 kb)

Fig. S5
Effects of different long-term fertilization treatments on the relative contributions (%) of total microbial necromass (a), bacterial necromass (b) and fungal necromass (c) to soil organic C (SOC) in the bulk soil and soil aggregates. Treatments CT, N, NP, M, NM, NPM, NDM and NPDM represent no fertilizer control, N fertilizer, N and P fertilizers, farmyard manure at an agronomic rate, N plus manure, NP plus manure, N plus double amounts of manure and NP plus double amounts of manure, respectively. Error bars are standard errors of means (n = 3). Different capital and lowercase letters indicate significant differences between aggregate fractions within a fertilizer treatment and between fertilizer treatments within an aggregate fraction, respectively, at P < 0.05 according to Duncan's multiple range test. (PNG 87 kb)
Table S1
Experimental design and application rates of chemical and organic fertilizers in different fertilizer treatments in the summer cropping and winter fallow agroecosystem. (DOCX 17 kb)
Table S2
Changes in soil C/N ratios after 10% HF treatment of soil samples and the calculated R factors. (DOCX 16 kb)
Table S3
The relative proportions (%) of different organic functional groups of the farmyard manure. (DOCX 15 kb)
Table S4
Effects of different long-term (33 years) fertilization treatments on the aggregate-associated soil organic C (SOC) content and amount. Values are means ± standard errors (n = 3). Different capital and lowercase letters indicate significant differences between soil aggregate fractions within a fertilizer treatment and between fertilizer treatments within an aggregate fraction, respectively, at P < 0.05 according to Duncan's multiple range test. (DOCX 18 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, X., Jia, Z., Wang, J. et al. Linking soil aggregation to organic matter chemistry in a Calcic Cambisol: evidence from a 33-year field experiment. Biol Fertil Soils 59, 73–85 (2023). https://doi.org/10.1007/s00374-022-01684-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-022-01684-3