Abstract
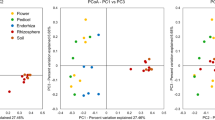
This study is the first approach to evaluate the diversity of bacterial β-glucosidase-encoding gene sequences, aiming to identify the main environmental factors structuring bacterial β-glucosidase genetic diversity in semiarid soils. Two agricultural management systems, soils under spontaneous cover vegetation vs. noncovered herbicide-treated soils, were tested. The weed biomass generated in the former was estimated around 2,600 kg ha−1 year−1, whereas leaves and root exudates from olive trees were the only input of C biomass in the latter. Dendrograms generated from polymerase chain reaction–denaturing gradient gel electrophoresis profiles of bacterial β-glucosidase-encoding genes revealed two clusters determined by soil treatment and sharing <20 % similarity. The sequences of a total of 59 DNA fragments, representing 39 operational taxonomic units, were successfully determined. The Proteobacteria phylum clearly dominated all the soil samples, but representatives of Chloroflexi, Deinococci, Actinobacteria, Thermotogae, and Firmicutes class were also detected. Management strategies favoring the presence of spontaneous vegetation determined a higher genetic diversity of β-glucosidase-encoding genes of soil bacteria. However, since there is little information of β-glucosidase gene sequences available in databases, it is difficult to establish particular relationships between bacterial networks for C degradation and land use. Results from canonical correspondence analysis indicated that bacterial metabolic networks for oligomeric C substrates utilization were affected by the physicochemical properties of the soil; the uppermost 10 cm of covered soil clustered together and were positively correlated with some chemical properties related to soil fertility, whereas less influence of soil texture was observed for the deeper layers of bare soils.



Similar content being viewed by others
References
Allen DE, Singh BP, Dalal RC (2011) Soil health indicators under climate change: a review of current knowledge. In: Singh BP, Cowie AL, Chan KY (eds) Soil biology. Soil health and climate change. Springer, Berlin, pp 25–45
Austin AT, Yahdjian L, Stark JM, Belnap J, Porporato A, Norton U, Ravetta DA, Schaeffer SM (2004) Water pulses and biogeochemical cycles in arid and semiarid ecosystems. Oecologia 141:221–235
Benitez E, Nogales R, Elvira C, Masciandaro G, Ceccanti B (1999) Enzyme activities as indicators of the stabilization of sewage sludges composting with Eisenia foetida. Bioresource Technol 67:297–303
Busto MD, Ortega N, Perez-Mateos M (1995) Induction of β-glucosidase in fungal and soil bacterial cultures. Soil Biol Biochem 27:949–954
Cañizares R, Benitez E, Ogunseitan OA (2011) Molecular analyses of [beta]-glucosidase diversity and function in soil. Eur J Soil Biol 47:1–8
Cañizares R, Moreno B, Benitez E (2012a) Biochemical characterization with detection and expression of bacterial β-glucosidase encoding genes of a Mediterranean soil under different long-term management practices. Biol Fertil Soils 48:651–663
Cañizares R, Moreno B, Benitez E (2012b) Bacterial β-glucosidase function and metabolic activity depend on soil management in semiarid rainfed agriculture. Ecol Evol 2:727–731
Cardelli R, Marchini F, Saviozzi A (2012) Soil organic matter characteristics, biochemical activity and antioxidant capacity in Mediterranean land use systems. Soil Till Res 120:8–14
Castro J, Fernández-Ondoño E, Rodríguez C, Lallena AM, Sierra M, Aguilar J (2008) Effects of different olive-grove management systems on the organic carbon and nitrogen content of the soil in Jaén (Spain). Soil Till Res 98:56–67
Degens BP, Schippers LA, Sparling GP, Vojvodic-Vokovic M (2000) Decreases in organic C reserves in soils can reduce the catabolic diversity of soil microbial communities. Soil Biol Biochem 32:189–196
Eivazi F, Tabatabai MA (1988) Glucosidases and galactosidases in soils. Soil Biol Biochem 20:601–606
Excoffier L, Laval G, Schneider S (2005) Arlequin ver. 3.0: an integrated software package for population genetics data analysis. Evol Bioinformatics Online 1:47–50
FAO (1998) World reference base for soil resources. World Soil Resources Reports 84, FAO-ISRIC-ISSS, Rome
Fernandez I, Mahieu N, Cadisch G (2003) Carbon isotopic fractionation during decomposition of plant materials of different quality. Global Biogeochem Cy 17:1075
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontologia Electronica 4:1–9
Klose SK, Tabatabai MT (2002) Response of glycosidases in soils to chloroform fumigation. Biol Fertil Soils 35:262–269
Knight TR, Dick RP (2004) Differentiating microbial and stabilized β-glucosidase activity relative to soil quality. Soil Biol Biochem 36:2089–2096
Li YN, Porter AW, Mumford A, Zhao XH, Young LY (2012) Bacterial community structure and bamA gene diversity in anaerobic degradation of toluene and benzoate under denitrifying conditions. J Appl Microbiol 112:269–27
Mao Y, Yannarell AC, Mackie RI (2011) Changes in N-transforming archaea and bacteria in soil during the establishment of bioenergy crops. PLoS One 6:e24750. doi:10.1371/journal.pone.0024750
Matsumoto K, Kawamura K, Uchida M, Shibata Y (2007) Radiocarbon content and stable carbon isotopic ratios of individual fatty acids in subsurface soil: implication for selective microbial degradation and modification of soil organic matter. Geochem J 41:483–492
Metzger MJ, Rounsevell MDA, Acosta-Michlik L, Leemans R, Schröter D (2006) The vulnerability of ecosystem services to land use change. Agr Ecosyst Environ 114:69–85
Moscatelli MC, Lagomarsino A, Garzillo AMV, Pignataro A, Grego S (2012) β-Glucosidase kinetic parameters as indicators of soil quality under conventional and organic cropping systems applying two analytical approaches. Ecol Ind 13:322–327
Muyzer G, De Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microb 59:695–700
Naafs DFW, Van Bergen PF, Boogert SJ, De Leeuw JW (2004) Solvent-extractable lipids in an acid andic forest soil; variations with depth and season. Soil Biol Biochem 36:297–308
Nannipieri P (2006) Role of stabilized enzymes in microbial ecology and enzyme extraction from soil with potential applications in soil proteomics. In: Nannipieri P, Smalla K (eds) Nucleic acids and proteins in soil, vol 8. Springer, Berlin, pp 217–255
Nei M, Li WH (1979) Mathematical model for studying genetic variation in terms of restriction endonucleases. P Natl Acad Sci Usa 76:5269–5273
Newman T, de Bruijn FJ, Green P, Keegstra K, Kende H, McIntosh L, Ohlrogge J, Raikhel N, Somerville S, Thomashow M, Retzel E, Somerville C (1994) Genes galore: a summary of methods for accessing results from large-scale partial sequencing of anonymous Arabidopsis cDNA clones. Plant Physiol 106:1241–1255
Qian H, Hu B, Cao D, Chen W, Xu X, Lu Y (2007) Bio-safety assessment of validamycin formulation on bacterial and fungal biomass in soil monitored by real-time PCR. B Environ Cont Toxicol 78:239–244
Raup DM, Crick RE (1979) Measurement of faunal similarity in paleontology. J Paleontol 53:1213–1227
Resat H, Bailey V, McCue LA, Konopka A (2012) Modeling microbial dynamics in heterogeneous environments: growth on soil carbon sources. Microb Ecol 63:883–897
Rowan AK, Snape JR, Fearnside D, Barer MR, Curtis TP, Head IM (2003) Composition and diversity of ammonia-oxidising bacterial communities in wastewater treatment reactors of different design treating identical wastewater. FEMS Microbiol Ecol 43:195–206
Rubino M, Lubritto C, D’Onofrio A, Terrasi F, Kramer C, Gleixner G, Cotrufo M (2009) Isotopic evidences for microbiologically mediated and direct C input to soil compounds from three different leaf litters during their decomposition. Environ Chem Lett 7:85–95
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB, Parks DH, Robinson CJ, Sahl JW, Stres B, Thallinger GG, Van Horn DJ, Weber CF (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbio 75:7537–7541
Schwieger F, Tebbe CC (1998) A new approach to utilize PCR-single-strand-conformation polymorphism for 16S rRNA gene-based microbial community analysis. Appl Environ Microbiol 64:4870–4876
Shackle VJ, Freeman C, Reynolds B (2000) Carbon supply and the regulation of enzyme activity in constructed wetlands. Soil Biol Biochem 32:1935–1940
Stevenson FJ, Cole MA (1999) Cycles of soil: carbon, nitrogen, phosphorus, sulfur, micronutrients. Wiley, New York, 427 pp
Stott DE, Andrews SS, Liebig MA, Wienhold BJ, Karlen DL (2010) Evaluation of β-glucosidase activity as a soil quality indicator for the soil management assessment framework. Soil Sci Soc Am J 74:107–119
Štursová M, Baldrian P (2011) Effects of soil properties and management on the activity of soil organic matter transforming enzymes and the quantification of soil-bound and free activity. Plant Soil 338:99–110
Ter Braak CJF, Smilauer P (2002) CANOCO reference manual and CanoDraw for Windows user’s guide: software for canonical community ordination (version 4.5). Microcomputer Power, Ithaca, Available at http://www.canoco.com
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Trasar-Cepeda C, Gil-Sotres F, Leirós MC (2007) Thermodynamic parameters of enzymes in grassland soils from Galicia, NW Spain. Soil Biol Biochem 39:311–319
Vivas A, Moreno B, Garcia-Rodriguez S, Benitez E (2009) Assessing the impact of composting and vermicomposting on bacterial community size and structure, and microbial functional diversity of an olive-mill waste. Biores Technol 100:1319–1326
Zimmerman AR, Ahn MY (2011) Organo-mineral–enzyme interaction and soil enzyme activity. In: Shukla G, Varma A (eds) Soil enzymology. Springer, Berlin, pp 271–292
Acknowledgments
This work was supported by ERDF-cofinanced grant CGL2009-07907 from the Spanish Ministry of Science of Innovation. R. Cañizares is supported by the JAE-CSIC predoctoral program. We also thank J. Castro for providing some chemical and physical data for the soils used in this study and the anonymous reviewers comments that greatly improved the manuscript. The chromatographic analyses were made at the Scientific Instrumentation Service, Estación Experimental del Zaidín, CSIC, Granada, Spain.
Author information
Authors and Affiliations
Corresponding author
Additional information
Beatriz Moreno and Rosa Cañizares contributed equally to this work.
Rights and permissions
About this article
Cite this article
Moreno, B., Cañizares, R., Nuñez, R. et al. Genetic diversity of bacterial β-glucosidase-encoding genes as a function of soil management. Biol Fertil Soils 49, 735–745 (2013). https://doi.org/10.1007/s00374-012-0765-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-012-0765-3