Abstract
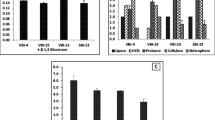
Plant growth-promoting bacteria (PGPB) were reported to influence the growth, yield, and nutrient uptake by an array of mechanisms. We selected seven different plant growth-promoting traits and antagonistic ability to screen 207 bacteria isolated from composts. Fifty-four percent of PGPB were from farm waste compost (FWC), 56% from rice straw compost (RSC), 64% from Gliricidia vermicompost (GVC), and 41% from macrofauna associated with FWC. Twelve isolates based on different plant growth-promoting traits and seed vigor index were evaluated at glasshouse for plant growth-promoting activity on pearl millet. Seven isolates significantly increased shoot length and ten isolates showed significant increase in leaf area, root length density, and plant weight. Maximum increase in plant weight was by Serratia marcescens EB 67 (56%), Pseudomonas sp. CDB 35 (52%), and Bacillus circulans EB 35 (42%). Plant growth-promoting activity of composts and bacteria (EB 35, EB 67, and CDB 35) was studied together. All the three composts showed significant increase in growth of pearl millet, which was 77% by RSC, 55% by GVC, and 30% by FWC. Application of composts with bacteria improved plant growth up to 88% by RSC with EB 67, 83% with GVC and EB 67. These results show the synergistic effect of selected bacteria applied with composts on growth of pearl millet.

Similar content being viewed by others
References
Alka V, Kamlesh K, Pathak DV, Suneja S, Neeru N (2001) In vitro production of plant growth regulators (PGRs) by Azotobacter chroococcum. Indian J Microbiol 41:305–307
Arancon NQ, Edwards CA, Bierman P, Metzger JD, Lee S, Welch C (2004) Effects of vermicomposts on growth and marketable fruits of field-grown tomatoes, peppers and strawberries. Pedobiolgia 47:731–735
Atiyeh RM, Subler S, Edwards CA, Bachman G, Metzger JD, Shuster W (2000) Effects of vermicomposts and composts on plant growth in horticultural media and soil. Pedobiolgia 44:579–590
Bacilio M, Vazquez P, Bashan Y (2003) Alleviation of noxious effects of cattle ranch composts on wheat seed germination by inoculation with Azospirillum spp. Biol Fertil Soils 38:261–266
Beri V, Sidhu BS, Gupta AP, Tiwari RC, Pareek RP, Rupela OP, Khera R, Ahluwalia JS (2003) Organic resources of a part of Indo-Gangetic plain and their utilization. PAU
Bernhard S, Neilands JB (1987) Universal chemical assay for detection and determination of siderophores. Anal Biochem 169:46–56
Bhatnagar SK, Khairwal IS, Pareek S (2002) Pearl millet nucleus and breeder seed production. Technical bulletin. Project co-ordinator (Pearl millet). All India co-ordinated pearl millet improvement project (AICPMIP), Jodhpur, India
Bric JM, Bostock RM, Silverstone SE (1991) Rapid in situ assay for indole acetic acid production by bacteria immobilized on a nitrocellulose membrane. Appl Environ Microbiol 57:535–538
Correa JD, Barrios ML, Galdona RP (2004) Screening for plant growth-promoting rhizobacteria in Chamaecytisus proliferus (tagasaste), a forage tree-shrub legume endemic to the Canary Islands. Plant Soil 266:75–84
Dey R, Pal KK, Bhatt DM, Chauhan SM (2004) Growth promotion and yield enhancement of peanut (Arachis hypogaea L.) by application of plant growth promoting rhizobacteria. Microbiol Res 159:371–394
Dworkin M, Foster J (1958) Experiments with some microorganisms which utilize ethane and hydrogen. J Bacteriol 75:592–601
Goldstein AH (1986) Bacterial solubilization of mineral phosphates: historical perspectives and future prospects. Am J Altern Agric 1:51–65
Gyaneshwar P, Naresh Kumar G, Parekh LJ (1998) Effect of buffering on the P-solubilizing ability of microorganisms. World J Microbiol Biotechnol 14:669–673
Haggag WM (2002) Sustainable agriculture management of plant diseases. J Biol Sci 2(4):280–284. http://www.ansinet.org/fulltext/jbs/jbs24280–284.pdf (online journal)
Han J, Sun L, Dong X, Cai Z, Yang H, Wang Y, Song W (2005) Characterization of a novel plant growth-promoting bacteria strain Delftia tsuruhatensis HR4 both as a diazotroph and a potential biocontrol agent against various pathogens. Syst Appl Microbiol 28:66–76
Hoitnik HAJ, Boehm MJ (1999) Biocontrol within the context of soil microbial communities: a substrate-dependent phenomenon. Annu Rev Phytopathol 37:427–446
Hontzeas N, Jerome Zoidakis J, Glick BR, Abu-Omar MM (2004) Expression and characterization of 1-aminocyclopropane-1-carboxylate deaminase from the rhizobacterium Pseudomonas putida UW4: a key enzyme in bacterial plant growth promotion. Biochim Biophys Acta 1703:11–19
Jacobson BC, Pasternak JJ, Glick BR (1994) Partial purification characterization of 1-aminocyclopropane-1-carboxylate deaminase from the plant growth promoting rhizobacterium Pseudomonas putida GR 12-2. Can J Microbiol 40:1019–1025
Josey DP, Beynon JL, Johnston AWB, Beringer JE (1979) Strain indentification in Rhizobium using intrinsic antibiotic resistance. J Appl Bacteriol 46:343–350
Kalbe C, Marten P, Sastry GRK (1996) Strains of genus Serratia as beneficial rhizobacteria of oilseed rape with antifungal properties. Microbiol Res 151:433–439
Khalid A, Arshad M, Zahir ZA (2004) Screening plant growth promoting rhizobacteria for improving growth and yield of wheat. J Appl Microbiol 96:473–480
Kishore GK, Pande S, Podile AR (2005) Phylloplane bacteria increase seedling emergence, growth and yield of field-grown groundnut (Arachis hypogeae L.). Lett Appl Microbiol 40:260–268
Kloepper JW (1993) Plant growth-promoting rhizobacteria as biological control agents. In: Metting FB Jr (ed) Soil microbial ecology. Marcel Dekker, NewYork, pp 255–274
Krieg NR, Holt JG (1984) In: Murray RGE, Brenner DJ, Bryant MP, Holt JG, Krieg NR, Moulder JW, Pfennig N, Sneath PHA, Staley JT (eds) Bergey’s manual of systematic bacteriology, vol I. Williams and Wilkins, Baltimore, MD, USA
Manici LM, Caputo F, Babibi V (2004) Effect of green manure on Pythium spp. population and microbial communities in intensive cropping systems. Plant Soil 263:133–142
Normander B, Prosser JI (2000) Bacterial origin and community composition in the barley phytosphere as a function of habitat and presowing conditions. Appl Environ Microbiol 66:4372–4377
Pal KK, Tilak KVBR, Saxena AK, Dey R, Singh CS (2001) Suppression of maize root diseases caused by Macrophomina phaseolina, Fusarium moniliforme and Fusarium graminearum by plant growth promoting rhizobacteria. Microbiol Res 156:209–223
Pattern CL, Glick BR (2002) Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. Appl Environ Microbiol 68:3795–3801
Penrose DM, Moffatt BA, Glick BR (2001) Determination of 1-aminocyclopropane-1-carboxylic acid (ACC) to assess the effects of ACC deaminase-containing bacteria on roots of canola seedlings. Can J Microbiol 47:77–80
Postma J, Montenari M, van den Boogert PHJF (2003) Microbial enrichment to enhance disease suppressive activity of compost. Eur J Soil Biol 39:157–163
Raj SN, Deepak SA, Basavaraju P, Shetty HS, Reddy MS, Kloepper JW (2003) Comparative performance of formulations of plant growth promoting rhizobacteria in growth promotion and suppression of downy mildew in pearl millet. Crop Prot 22:579–588
Reid JD, Ogrydziak DM (1981) Chitinase-overproducing mutant of Serratia marcescens. Appl Environ Microbiol 41:64–669
Richardson AE, Hadobas PA (1997) Soil isolates of Pseudomonas spp. that utilize inositol phosphates. Can J Microbiol 43:509–516
Richardson AE, Hadobas PA, Hayes JE, O’Hara CP, Simpson RJ (2001) Utilization of phosphorous by pasture plants supplied with myo-inositol hexaphosphate is enhanced by the presence of soil microorganisms. Plant Soil 229:47–56
Renwick AR, Campbell A, Coe S (1991) Assessment of in vivo screening systems for potential biocontrol agents of Gaeumannomyces graminis. Plant Physiol 40:524–532
Rodriguez H, Fraga R (1999) Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol Adv 17:319–339
Sadfi N, Cherif M, Fliss I, Boudabbous A, Antoun H (2001) Evaluation of bacterial isolates from salty soils and Bacillus thuringensis strains for the biocontrol of Fusarium dry rot of potato tubers. J Plant Pathol 83:101–118
Sharma A, Johri BN (2003) Growth promoting influence of siderophore-producing Pseudomonas strains GRP3A and PRS9 in maize (Zea mays L.) under iron limiting conditions. Microbiol Res 158:243–248
Thimm T, Hoffmann A, Borkott H, Munch JN, Tebbe CC (1998) The gut of the soil microarthropod Folsomia candida (Collembola) is a frequently changeable but selective habitat and a vector for microorganisms. Appl Environ Microbiol 64:2660–2669
Xie H, Pasternak JJ, Glick BR (1996) Isolation and characterization of mutants of the plant growth-promoting rhizobacterium Pseudomonas putida GR12-2 that overproduce indole acetic acid. Curr Microbiol 32:67–71
Acknowledgements
We thank Dr. R P Thakur of ICRISAT for providing pathogenic fungal cultures, Dr. Neeru Narula of CCS Haryana Agricultural University, Hisar, Haryana, and Dr. G Naresh Kumar of M S University of Baroda, Gujarat for providing the reference strains used in these studies. Doctoral fellowship to Ms Hameeda Bee from the Jawaharlal Nehru Memorial Fund, New Delhi is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hameeda, B., Rupela, O.P., Reddy, G. et al. Application of plant growth-promoting bacteria associated with composts and macrofauna for growth promotion of Pearl millet (Pennisetum glaucum L.). Biol Fertil Soils 43, 221–227 (2006). https://doi.org/10.1007/s00374-006-0098-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-006-0098-1