Abstract
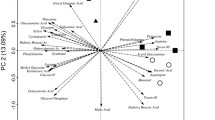
The present study compares the community structures of microbiota at different habitats in Japanese rice fields by comparing their phospholipid fatty acid (PLFA) compositions to understand the contribution of different habitats to microbiological diversity. The data were collected from four neighboring rice fields. Comparison was made for the PLFA compositions extracted from the floodwater, percolating water, rice soils under flooded and drained conditions, rice straw (RS) placed in flooded and drained rice soils, RS in the composting process, and RS compost placed in a flooded rice field. Average amounts of PLFAs were 33 μg L−1 in the floodwater, 17.1 μg L−1 in the percolating water from plow layers, 34.6 μg L−1 in the percolating water from subsoil layers, 108 μg g−1 dry weight basis (dw) in flooded rice soils, 382 μg g−1 dw in RS materials, 2,510 μg g−1 dw in RS composts, 2,850 μg g−1 dw in RS composts after application to a flooded rice soil, 222 μg g−1 wet weight basis (ww) in RS in drained rice soils, and 284 μg g−1 ww in RS in flooded rice soils. The total amount of PLFAs to the soil depth of 10 cm was estimated to be about 12 g m−2. The PLFA compositions were different from each other depending on the habitats. Rice soils were characterized by the predominance of actinomycetes and Gram-positive bacteria in comparison with the other habitats. In contrast, the microbial communities in the floodwater and percolating water were characterized by the predominance of Gram-negative bacteria and eukaryotes (presumably algae), and Gram-negative bacteria, respectively. The microbial community of RS materials was dominated by fungi. Gram-positive bacteria became predominant in RS after application to flooded rice soils, while RS placed in a drained rice field after harvesting rice was characterized by the predominance of Gram-negative bacteria and fungi. The community structures at respective habitats were stable and specific, irrespective of the season of sampling and the duration of decomposition of RS.





Similar content being viewed by others
References
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Cahyani VR, Watanabe A, Matsuya K, Asakawa S, Kimura M (2002) Succession of microbiota estimated by phospholipid fatty acid analysis and changes in organic constituents during the composting process of rice straw. Soil Sci Plant Nutr 48:735–743
Cahyani VR, Matsuya K, Asakawa S, Kimura M (2003) Succession and phylogenetic composition of bacterial communities responsible for the composting process of rice straw estimated by PCR-DGGE analysis. Soil Sci Plant Nutr 49:619–630
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131:479–491
Herrmann RF, Shann JF (1997) Microbial community changes during composting of municipal solid waste. Microb Ecol 33:78–85
Hirota T (2001) Statistical macros for Excel. http://www.tuat.ac.jp/~ethology/Columbo/Stat/
Ito Y, Sato K (2002) Problems around the indices of species diversity for comparison of different communities. Seibutsukagaku 53:204–220
Kimura M (2000) Anaerobic microbiology in waterlogged rice fields. In: Bollag JM, Stotzky G (eds) Soil biochemistry, vol 10. Marcel Dekker, New York, pp 35–138
Kimura M, Kishi H, Okabe A, Maie N (2001a) Phospholipid fatty acid composition of microbiota in the percolating water from a rice paddy microcosm. Soil Sci Plant Nutr 47:533–545
Kimura M, Miyaki M, Fujinaka K, Maie N (2001b) Microbiota responsible for the decomposition of rice straw in a submerged paddy soil estimated from phospholipid fatty acid composition. Soil Sci Plant Nutr 47:569–578
Kroppenstedt RM (1992) The genus Nocardiopsis. In: Balows A, Trüper HG, Dworkin M, Harder W, Schleifer K-H (eds) The prokaryotes, 2nd edn, vol 2. Springer, Berlin Heidelberg New York, pp 1139–1156
Lechevalier H, Lechevalier MP (1988) Chemotaxonomic use of lipids — an overview. In: Ratledge C, Wilkinson SG (eds) Microbial lipid, vol 1. Academic Press, London, pp 869–902
Lösel DM (1988) Fungal lipids. In: Ratledge C, Wilkinson SG (eds) Microbial lipids, vol 1. Academic Press, London, pp 699–806
Lueders T, Friedrich M (2000) Archaeal population dynamics during sequential reduction processes in rice field soil. Appl Environ Microbiol 66:2732–2742
Nakamura A, Tun CC, Asakawa S, Kimura M (2003) Microbial community responsible for the decomposition of rice straw in a paddy field: estimation by phospholipid fatty acid analysis. Biol Fertil Soils 38:288–295
Nannipieri P, Acher J, Ceccherini MT, Landi L, Pietramellara G, Renella G (2003) Microbial diversity and soil functions. Eur J Soil Sci 54:655–670
Okabe A, Oike H, Toyota K, Kimura M (2000a) Comparison of phospholipid fatty acid composition in floodwater and plow layer soil during the rice cultivation period in a Japanese paddy field. Soil Sci Plant Nutr 46:893–904
Okabe A, Toyota K, Kimura M (2000b) Seasonal variations of phospholipid fatty acid composition in the floodwater of a Japanese paddy field under a long-term fertilizer trial. Soil Sci Plant Nutr 46:177–188
O'Leary WM, Wilkinson SG (1988) Gram-positive bacteria. In: Ratledge C, Wilkinson SG (eds) Microbial lipids, vol 1. Academic Press, London, pp 117–202
Shimizu M, Hayashi M, Matsuya K, Murase J, Kimura M (2002a) Comparison of microbial communities in percolating water from plow layer and subsoil layer estimated by phospholipid fatty acid (PLFA) analysis. Soil Sci Plant Nutr 48:877–881
Shimizu M, Nakajima Y, Matsuya K, Kimura M (2002b) Comparison of phospholipid fatty acid composition in percolating water, floodwater, and the plow layer soil during the rice cultivation period in a Japanese paddy field. Soil Sci Plant Nutr 48:595–600
Takai Y, Kamura T (1966) The mechanism of reduction in waterlogged paddy soil. Folia Microbiol 11:304–313
Tanahashi T, Murase J, Matsuya K, Asakawa S, Kimura M (2004) Microbial communities responsible for the decomposition of rice straw compost in a Japanese rice paddy field determined by phospholipid fatty acid (PLFA) analysis. Soil Sci Plant Nutr 50:1229–1236
Vestal JR, White DC (1989) Lipid analysis in microbial ecology: quantitative approaches to the study of microbial communities. BioScience 39:535–541
White DC, Davis WM, Nickels JS, King JD, Bobbie RJ (1979) Determination of the sedimentary microbial biomass by extractable lipid phosphate. Oecologia 40:51–62
Wilkinson SG (1988) Gram-negative bacteria. In: Ratledge C, Wilkinson SG (eds) Microbial lipids, vol 1. Academic Press, London, pp 299–488
Zelles L (1999) Fatty acid patterns of phospholipids and lipopolysaccharides in the characterisation of microbial communities in soil: a review. Biol Fertil Soils 29:111–129
Acknowledgements
We thank the coworkers in our laboratory for their valuable comments and also the staff members of Aichi-ken Anjo Research and Extension Center for allowing us to use the experimental fields for soil sampling. We also appreciate A. Nakanishi, T. Torimaru, and N. Tomaru of the Graduate School of Bioagricultural Sciences, Nagoya University, for their help in the AMOVA analysis. This study was supported in part by a Grant-in Aid for Scientific Research (B; No. 11460031) from the Japan society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kimura, M., Asakawa, S. Comparison of community structures of microbiota at main habitats in rice field ecosystems based on phospholipid fatty acid analysis. Biol Fertil Soils 43, 20–29 (2006). https://doi.org/10.1007/s00374-005-0057-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-005-0057-2