Abstract
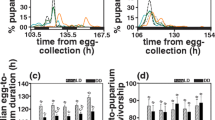
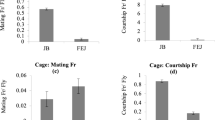
Although, circadian clocks are believed to be involved in the regulation of life-history traits such as pre-adult development time and lifespan in fruit flies Drosophila melanogaster, there is very little unequivocal evidence either to support or refute this. Here we report the results of a long-term study aimed at examining the role of circadian clocks in the temporal regulation of pre-adult development in D. melanogaster. We employed laboratory selection protocol for faster pre-adult development on four large, outbred, random mating populations of Drosophila. We assayed pre-adult development time and circadian period of locomotor activity rhythm of these flies at regular intervals of 5–10 generations. After 50 generations of selection, the overall egg-to-adult duration in the selected stocks was reduced by ~29 h (~12.5 %) relative to controls, with the selected populations showing a concurrent reduction in time taken to hatching, pupation and wing pigmentation, by ~2, ~16, and ~25.2 h, respectively. Furthermore, selected populations showed a concomitant reduction in the circadian period of locomotor activity rhythm, implying that circadian clocks and development time are correlated. Thus, our study provides the first ever unequivocal evidence for the evolution of circadian clocks as a correlated response to selection for faster pre-adult development, suggesting that circadian clocks and development are linked in fruit flies D. melanogaster.





Similar content being viewed by others
References
Ashburner M, Golic KG, Hawley RS (2005) Drosophila: a laboratory handbook, 2nd edn. Cold Spring Harbor Laboratory Press, New York, pp 162–164
Bakker K (1959) Feeding period, growth and pupation in larvae of Drosophila melanogaster. Entomol Exp Appl 2:171–186
Chippindale AK, Hoang DT, Service PM, Rose MR (1994) The evolution of development in Drosophila melanogaster selected for postponed senescence. Evolution 48:1880–1899
Chippindale AK, Alipaz JA, Chen HW, Rose MR (1997) Experimental evolution of accelerated development in Drosophila. 1. Developmental speed and larval survival. Evolution 51:1536–1551
de Moed GH, Kruitwagen CLJJ, de Jong G, Scharloo W (1999) Critical weight for the induction of pupariation in Drosophila melanogaster: genetic and environmental variation. J Evol Biol 12:852–858
Harano T, Miyatake T (2011) Independence of genetic variation between circadian rhythm and development time in the seed beetle, Callosobruchus chinensis. J Insect Physiol 57:415–420
Harker JE (1965) The effect of a biological clock on the developmental rate of Drosophila pupae. J Exp Biol 43:323–337
Konopka RJ, Pittendrigh CS, Orr D (1989) Reciprocal behaviour associated with altered homeostasis and photosensitivity of Drosophila clock mutants. J Neurogenet 6:1–10
Kumar S, Kumar D, Paranjpe DA, Akarsh CR, Sharma VK (2007) Selection on the timing of adult emergence results in altered circadian clocks in fruit flies Drosophila melanogaster. J Exp Biol 210:906–918
Kyriacou CP, Hall JC (1980) Circadian rhythm mutations in Drosophila affect short-term fluctuations in the male’s courtship song. Proc Nat Acad Sci USA 77:6929–6933
Kyriacou CP, Oldroyd M, Wood J, Sharp M, Hill M (1990) Clock mutation alters developmental timing in Drosophila. Heredity 64:395–401
Lakowski B, Hekimi S (1996) Determination of life-span in Caenorhabditis elegans by four clock genes. Science 272:1010–1013
Lorenz LJ, Hall JC, Rosbash M (1989) Expression of a Drosophila mRNA is under circadian clock control during pupation. Development 1:8697–8800
Miyatake T (1996) Comparison of adult life history traits in lines artificially selected for long and short larval and pupal developmental periods in the melon fly, Bactrocera cucurbitae (Diptera: Tephritidae). App Entomol Zool 31:335–343
Miyatake T (1997) Correlated responses to selection for developmental period in Bactrocera cucurbitae (Diptera: Tephritidae): time of mating and daily activity rhythms. Behav Genet 27:489–498
Miyatake T (2002) Circadian rhythm and time of mating in Bactrocera cucurbitae (Diptera: Tephritidae) selected for age at reproduction. Heredity 88:302–306
Moss GE (2007) Heterochronic genes and nature of the developmental time. Curr Biol 17:425–434
Mueller LD (1997) Theoretical and empirical examination of density-dependent selection. Annu Rev Ecol Syst 28:269–288
Mueller LD, Ayala FJ (1981) Trade-off between r-selection and k-selection in Drosophila populations. Proc Nat Acad Sci USA 78:1303–1305
Mukherjee N, Kannan NN, Yadav P, Sharma VK (2012) A model based on oscillatory threshold and build-up of a developmental substance can explain gating of adult emergence in fruit flies Drosophila melanogaster. J Exp Biol 215:2960–2968
Nunney L (1990) Drosophila on oranges: colonization, competition and coexistence. Ecology 71:1904–1915
Nunney L (1996) The response to selection for fast larval development in Drosophila melanogaster and its effect on adult weight: an example of a fitness trade-off. Evolution 50:1193–1204
Paranjpe DA, Anitha D, Chandrashekaran MK, Joshi A, Sharma VK (2005) Possible role of eclosion rhythm in mediating the effects of light-dark environments on pre-adult development in Drosophila melanogaster. BMC Dev Biol 5:5
Partridge L, Fowler K (1992) Direct and correlated responses to selection on age at reproduction in Drosophila melanogaster. Evolution 46:76–91
Pittendrigh CS (1981) Circadian systems: entrainment. In: Aschoff J (ed) Handbook of behavioral neurobiology, Biological Rhythms, vol 4. Plenum Press, New York, pp 95–124
Prasad NG, Joshi A (2003) What have two decades of laboratory life-history evolution studies on Drosophila melanogaster taught us? J Genet 82:45–76
Prasad NG, Shakarad M, Gohil VM, Sheeba V, Rajamani M, Joshi A (2000) Evolution of reduced pre-adult viability and larval growth rate in laboratory populations of Drosophila melanogaster selected for shorter development time. Genet Res 76:249–259
Prasad NG, Shakarad M, Anitha D, Rajamani M, Joshi A (2001) Correlated responses to selection for faster development and early reproduction in Drosophila: the evolution of larval traits. Evolution 55:1363–1372
Qiu J, Hardin PE (1996) Developmental state and circadian clock interact to influence the timing of eclosion in Drosophila melanogaster. J Biol Rhythms 11:75–86
Robertson FW (1963) The ecological genetics of growth in Drosophila: the genetic correlation between the duration of the larval period and body size in relation to larval diet. Genet Res 4:74–92
Sang JH (1956) The quantitative nutritional requirements of Drosophila melanogaster. J Exp Biol 33:45–72
Sharma VK, Joshi A (2002) Clocks, genes and evolution: the evolutionary genetics of circadian organization. In: Kumar V (ed) Biological clocks. Narosa, New Delhi, pp 5–23
Shimizu T, Miyatake T, Watari Y, Arai T (1997) A gene pleiotropically controlling developmental and circadian periods in the melon fly, Bactrocera cucurbitae (Diptera: Tephritidae). Heredity 70:600–605
StatSoft Inc (1995) STATISTICA™ Vol. I General conventions and Statistics Tulsa; StatSoft Inc
Wong A, Boutis P, Hekimi S (1995) Mutations in the clk-1 gene of Caenorhabditis elegans affect developmental and behavioural timing. Genetics 139:1247–1259
Zwaan BJ, Bijlsma R, Hoekstra RF (1995) Artificial selection for development time in Drosophila melanogaster in relation to the evolution of aging: direct and correlated responses. Evolution 49:635–648
Acknowledgments
We thank Sheeba Vasu and two anonymous reviewers for carefully reading our manuscript and suggesting some very useful changes. We also thank Madhumohan, Nisha, Madhumala, Rajanna, Manjesh and Muniraju for assisting us during the experiments and while maintaining stocks.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Heldmaier.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yadav, P., Sharma, V.K. Correlated changes in circadian clocks in response to selection for faster pre-adult development in fruit flies Drosophila melanogaster . J Comp Physiol B 183, 333–343 (2013). https://doi.org/10.1007/s00360-012-0716-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-012-0716-1