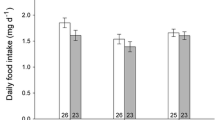
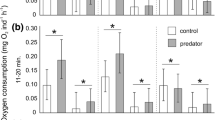
Abstract
We subjected fish from regions of high and low levels of predation pressure in four independent streams to a mild stressor and recorded their opercular beat rates. Fish from low-predation areas showed higher maximum, minimum and mean opercular beat frequencies than fish from high-predation regions. The change in opercular beat frequency (scope) was also significantly greater in fish from low- than in fish from high-predation regions. Under normal activity levels, however, low predation fish showed a reduced opercular beat frequency, which may be indicative of reduced activity levels or metabolic rate. Opercular beat frequency was negatively correlated with standard length as one would expect based on higher metabolic rates in smaller fish. We suggest that these contrasting stress responses are most likely the result of differential exposure to predators in fish from high- and low-predation areas. We argue that reduced stress responses in high-predation areas evolved to prevent excessive energy expenditure by modulating the fright response.





Similar content being viewed by others
References
Barcellos LJG, Nicolaiewsky S, de Souza SMG, Lulhier F (1999) Plasmatic levels of cortisol in the response to acute stress in Nile tilapia, Oreochromis niloticus (L), previously exposed to chronic stress. Aquacult Res 30:437–444
Barreto RE, Volpato GL (2004) Caution for using ventilatory frequency as an indicator of stress in fish. Behav Processes 66:43–51
Barreto RE, Luchiari AC, Macondes AL (2003) Ventilatory frequency indicates visual recognition of an allopatric predator in naïve Nile tilapia. Behav Processes 60:235–239
Basu N, Nakano T, Grau EG, Iwama GK (2001) The effects of cortisol on heat shock protein 70 levels in two fish species. Gen Comp Endocrinol 124:97–105
Brown C, Braithwaite VA (2004) Size matters: a test of boldness in eight populations of the poeciliid, Brachyraphis episcope. Anim Behav 68:1325–1329
Brown C, Braithwaite VA (2005) Effects of predation pressure on the cognitive ability of the poeciliid Brachyraphis episcopi. Behav Ecol 16:482–487. doi:10.1093/beheco/ari016
Brown C, Gardner C, Braithwaite VA (2004) Population variation in lateralised eye use in the poeciliid Brachyraphis episcopi. Proc R Soc Lond Ser B (Suppl) 271:S455–S457
Brown C, Jones FC, Braithwaite VA (2005) Environmental influences on boldness-shyness traits in the tropical poeciliid, Brachyraphis episcopi. Anim Behav (in press)
Caldji C, Francis D, Sharma S, Plotsky PM, Meaney MJ (2000) The effects of early rearing environment on the development of GABA A and central benzodiazepine receptor levels and novelty-induced fearfulness in the rat. Neuropsychopharmacology 22:219–229
Claireaux G, Lagardère JP (1999) Influence of temperature, oxygen and salinity on the metabolism of European sea bass. J Sea Res 42:157–168
Clarke A, Johnston NM (1999) Scaling of metabolic rate with body mass and temperature in teleost fish. J Anim Ecol 68:893–905. doi:10.1046/j.1365–2656.1999.00337.x
Clutton-Brock T (1989) A natural history of domesticated mammals. Cambridge University Press, Cambridge
Cooke SJ, Steinmetz J, Degner JF, Grant EC, Philipp DP (2003) Metabolic fright responses of different-sized largemouth bass (Micropterus salmoides) to two avian predators show variations in nonlethal energetic costs. Can J Zool 81:699–709
DeKoning ABL, Picard DJ, Bond SR, Schulte PM (2004) Stress and interpopulation variation in glycolytic enzyme activity and expression in a teleost fish Fundulus heteroclitus. Physiol Biochem Zool 77:18–26
Dunlap KD, Wingfield JC (1995) External and internal influences on indexes of physiological stress 1 Seasonal and population variation in adrenocortical secretion of free-living lizards, Sceloporus occidentalis. J Exp Zool 271:36–46
Ellis T, James JD, Stewart C, Scott AP (2004) A non-invasive stress assay based upon measurement of free cortisol released into the water by rainbow trout. J Fish Biol 65:1233–1252
Endler JA (1995) Multiple-trait coevolution and environmental gradients in guppies. Trends Ecol Evol 10:22–29
Espmark Y, Langvatn R (1985) Development and habituation of cardiac and behavioral-responses in young red deer calves (Cervus elaphus) exposed to alarm stimuli. J Mammal 66:702–711
Fevolden SE, Røed KH, Fjalestad K (2003) A combined salt and confinement stress enhances mortality in rainbow trout (Oncorhynchus mykiss) selected for high stress responsiveness. Aquaculture 216:67–76
Fraser DF, Gilliam JF (1992) Nonlethal impacts of predator invasion—Facultative suppression of growth and reproduction. Ecology 73: 959–970
Grantner A, Taborsky M (1998) The metabolic rates associated with resting, and with the performance of agonistic, submissive and digging behaviours in the cichlid fish Neolamprologus pulcher (Pisces: Cichlidae). J Comp Physiol B 168:427–433
Haase E, Donham RS (1980) Hormones and domestication. In: Epple A (ed) Avian Endocrinology. Academic, Santiago, pp 549–561
Haukenes AH, Barton BA (2004) Characterization of the cortisol response following an acute challenge with lipopolysaccharide in yellow perch and the influence of rearing density. J Fish Biol 64:851–862
Höjesjö J, Johnsson JI, Axelsson M (1999) Behavioural and heart rate responses to food limitation and predation risk: an experimental study on rainbow trout. J Fish Biol 55:1009–1019
Hughes GM (1960) A comparative study of gill ventilation in marine teleosts. J Exp Biol 37:28–45
Jennions MD, Telford SR (2002) Life-history phenotypes in populations of Brachyrhaphis episcopi (Poeciliidae) with different predator communities. Oecologia 132:44–50
Johnsson JI, Höjesjö J, Fleming IA (2001) Behavioural and heart rate responses to predation risk in wild and domesticated Atlantic salmon. Can J Fish Aquat Sci 58:788–794
Jones DR (1971) Theoretical analysis of factors which may limit the maximum oxygen uptake of fish: The oxygen cost of the cardiac and branchial pumps. J Theor Biol 32:341–349
Kunzl C, Sachser N (1999) The behavioural endocrinology of domestication: a comparison between the domestic Guinea pig (Gavia opera f porcellus) and its wild ancestor, the Cavy (Cavia apera). Horm Behav 35:28–37
Lepage O, Overli O, Petersson E, Jarvi T, Winberg S (2000) Differential stress coping in wild and domesticated sea trout. Brain Behav Evol 56:259–268
Lucas MC, Johnstone ADF, Priede IG (1993) Use of physiological telemetry as a method of estimating metabolism of fish in the natural environment. Trans Am Fish Soc 122:822–833
Majica CL (1998) Comparative ecology and historical biogeography of the genus Brachyraphis. Environ Biol Fishes 51:117–127
Metcalfe NB, Huntingford FA, Thorpe JE (1987) The influence of predation risk on the feeding motivation and foraging strategy of juvenile Atlantic salmon. Anim Behav 35:901–911
Miller RB (1954) Comparative survival of wild and hatchery-reared cutthroat trout in a stream. Trans Am Fish Soc 83:120–130
Olla BL, Davis MW, Schreck CB (1997) Effects of simulated trawling on sablefish and walleye pollock: The role of light intensity, net velocity and towing duration. J Fish Biol 50:1181–1194
Øverli O, Olsen RE, Lovik F, Ringo E (1999) Dominance hierarchies in Arctic charr, Salvelinus alpinus L: differential cortisol profiles of dominant and subordinate individuals after handling stress. Aquacult Res 30:259–264
Øverli O, Pottinger TG, Carrick TR, Overli E, Winberg S (2001) Brain monoaminergic activity in rainbow trout selected for high and low stress responsiveness. Brain Behav Evol 57:214–224
Pfeiffer W (1962) The fright reaction of fish. Biol Rev 37:495–511
Poole WR, Nolan DT, Wevers T, Dillane M, Cotter D, Tully O (2003) An ecophysiological comparison of wild and hatchery-raised Atlantic salmon (Salmo salar L) smolts from the Burrishoole system, western Ireland. Aquaculture 222:301–314
Pottinger TG, Carrick TR (1999) Modification of the plasma cortisol response to stress in rainbow trout by selective breeding. Gen Comp Endocrinol 116:122–132
Reed DH, Lowe EH, Briscoe DA, Frankham R (2003) Fitness and adaptation in a novel environment: Effect of inbreeding, prior environment, and lineage. Evolution 57:1822–1828
Robb T, Abrahams MV (2003) Variation in tolerance to hypoxia in a predator and prey species: an ecological advantage of being small?. J Fish Biol 62:1067–1081
Romero LM (2004) Physiological stress in ecology: Lessons from biomedical research. Trends Ecol Evol 19:249–255
Schmidt-Nielsen K (1997) Animal physiology: adaptation and environment. Cambridge University Press, Cambridge
Sutterland AM (1969) Effects of exercise on cardiac and ventilation frequency in three species of freshwater teleosts. Physiol Zool 42:36–52
Acknowledgements
We thank the Smithsonian Tropical Research Institute for their continued support and Fiona Burgess for her aid with husbandry. Thanks also to Felicity Brown for her help collecting the fish. The fish were collected and exported under licence from the Panamanian authorities (ANAM permit# 31503). This work was funded by NERC grant no NER/A/S/01/00608.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Heldmaier
Rights and permissions
About this article
Cite this article
Brown, C., Gardner, C. & Braithwaite, V.A. Differential stress responses in fish from areas of high- and low-predation pressure. J Comp Physiol B 175, 305–312 (2005). https://doi.org/10.1007/s00360-005-0486-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-005-0486-0