Abstract
Purpose
To analyze the utilization of Active Surveillance (AS) and Watchful Waiting (WW) in the daily routine setting, since both are non-invasive treatment options for localized prostate cancer (PCa), which are used in a curative (AS) or palliative (WW) setting. Since differentiation of both strategies is not always clear, patients were compared with respect to the inclusion criteria, frequency of follow-up examinations (Prostate Specific Antigen = PSA tests, rebiopsies), and initiation of a deferred treatment.
Methods
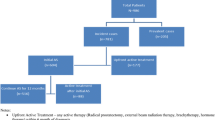
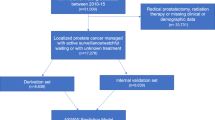
HAROW is a non-interventional, health-service research study on the management of localized PCa in the community setting. Of 3169 patients, prospectively enrolled from 2008 to 2013 with a mean follow-up of 28.2 months, 468 chose AS and 126 WW. Treating urologists reported clinical variables, information on therapy and clinical course of disease.
Results
AS patients were significantly younger and had more low-risk tumors. No differences were seen in the number of PSA tests during follow-up: mean number of PSA tests was 6.08 for AS- and 5.18 for WW patients, more than four PSA tests were reported in 63.9% AS- and 59.5% WW patients (p = 0.136). At least one re-biopsy was performed in 39.7% AS- and 9.5% WW patients (p < 0.001). Discontinuation rates were 23.9% (n = 112) for AS and 11.9% (n = 15) for WW. Most of the AS patients opted for a curative treatment (prostatectomy = 65, radiotherapy = 30), whereas 12 WW patients received a palliative hormone therapy and three patients received radiotherapy.
Conclusions
Physicians seem to distinguish clearly between AS and WW in terms of inclusion criteria and deferred therapy, whereas this differentiation tends to become indistinct in terms of follow-up examinations.



Similar content being viewed by others
References
Deutsche Krebsgesellschaft, Deutsche Krebshilfe, AWMF: Interdisziplinäre Leitlinie der Qualität S3 zur Früherkennung, Diagnose und Therapie der verschiedenen Stadien des Prostatakarzinoms. www.awmf.org/leitlinien/detail/ll/043–022OL.html. Accessed 26 Nov 2017
Mottet N, Bellmunt J, Bolla M et al (2017) EAU-ESTRO-SIOG guidelines on prostate cancer. part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol 71:618–629
Klotz L, Vesprini D, Sethukavalan P et al (2015) Long-term follow-up of a large active surveillance cohort of patients with prostate cancer. J Clin Oncol 33:272–277
Hamdy FC, Donovan JL, Lane JA et al (2016) 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med 375:1415–1424
Thomsen FB, Brasso K, Klotz LH et al (2014) Active surveillance for clinically localized prostate cancer—a systematic review. J Surg Oncol 109:830–835
Womble PR, Montie JE, Ye Z et al (2015) Contemporary use of initial active surveillance among men in Michigan with low-risk prostate cancer. Eur Urol 67:44–50
Weerakoon M, Papa N, Lawrentschuk N et al (2015) The current use of active surveillance in an Australian cohort of men: a pattern of care analysis from the Victorian Prostate Cancer Registry. BJU Int 115(Suppl 5):50
Loeb S, Folkvaljon Y, Curnyn C et al (2016) Uptake of active surveillance for very-low-risk prostate cancer in Sweden. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2016.3600. (Epub ahead of print)
Weissbach L, Stuerzebecher S, Mumperow E, Klotz T, Schnell D (2016) HAROW: the first comprehensive prospective observational study comparing treatment options in localized prostate cancer. World J Urol 34:641–647
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40:373–383
Choo R, Klotz L, Danjoux C et al (2002) Feasibility study: watchful waiting for localized low to intermediate grade prostate carcinoma with selective delayed intervention based on prostate specific antigen, histological and/or clinical progression. J Urol 167:1664–1669
Parker C (2004) Active surveillance: towards a new paradigm in the management of early prostate cancer. Lancet Oncol 5:101–106
van den Bergh RC, Roemeling S, Roobol MJ, Roobol W, Schröder FH, Bangma CH (2007) Prospective validation of active surveillance in prostate cancer: the PRIAS study. Eur Urol 52:1560–1563
Heidenreich A, Aus G, Bolla M et al (2008) EAU guidelines on prostate cancer. Eur Urol 53:68–80
Berges R, Ebert T, Schafhauser W, Schultze-Seemann W, Braun M, Herden J, Weib P, Garcia Schürmann M, Reimann M, Bornhof C, Oberpenning F, Baur P, Zumbé J, Gronau E, Diederichs W, Brinkman OA, Goepel M, Göll A, Hoefner K, Kriegmair M, Laabs S, Planz B, Platz G, Heidenreich A (2015) Treatment mapping of prostate cancer in DVPZ prostate centers in Germany. Urologe A [Article in German] 54(1546):1548–1554
Cooperberg MR, Carroll PR (2015) Trends in management for patients with localized prostate cancer, 1990–2013. JAMA 314:80–82
Wilt TJ, Jones KM, Barry MJ et al (2017) Follow-up of prostatectomy versus observation for early prostate cancer. N Engl J Med 377:132–142
Bill-Axelson A, Holmberg L, Garmo H et al (2014) Radical prostatectomy or watchful waiting in early prostate cancer. N Engl J Med 370:932–942
Loeb S, Walter D, Curnyn C, Gold HT, Lepor H, Makarov DV (2016) How active is active surveillance? intensity of follow-up during active surveillance for prostate cancer in the United States. J Urol 196:721–726
Funding
The HAROW study was initiated and conducted by the Foundation of Men’s Health (Berlin, Germany). Gazprom Germany sponsored the Foundation of Men’s Health by providing an unconditional Grant for data collection and data management.
Author information
Authors and Affiliations
Contributions
JH Project development, Data analysis, Manuscript writing. LW Project development, Data collection, Data analysis, Manuscript editing
Corresponding author
Ethics declarations
Conflict of interest
Jan Herden and Lothar Weissbach declare that they have no conflicts of interest.
Ethical approval
The HAROW study was approved by the ethics committee of the Bavarian State Board of Physicians. The study was registered under study ID 479 at the DKSR (German Cancer Study Registry; February 2008). All procedures performed in studies involving human participants were in accordance with the ethical standards of the Ethics Committee of the State Chamber of Medicine in Bavaria and with the 1964 Helsinki Declaration and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Herden, J., Weissbach, L. Utilization of Active Surveillance and Watchful Waiting for localized prostate cancer in the daily practice. World J Urol 36, 383–391 (2018). https://doi.org/10.1007/s00345-018-2175-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-018-2175-0