Abstract
Purpose
To investigate the clinical outcomes of metastatic prostate cancer patients and the relationship between nadir prostate-specific antigen (PSA) levels and different types of primary androgen deprivation therapy (PADT). This study utilized data from the Japan Study Group of Prostate Cancer registry, which is a large, multicenter, population-based database.
Methods
A total of 2982 patients treated with PADT were enrolled. Kaplan–Meier analysis was used to compare progression-free survival (PFS) and overall survival (OS) in patients treated using combined androgen blockade (CAB) and non-CAB therapies. The relationships between nadir PSA levels and PADT type according to initial serum PSA levels were also investigated.
Results
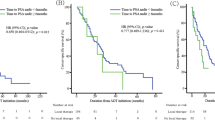
Among the 2982 enrolled patients, 2101 (70.5 %) were treated with CAB. Although CAB-treated patients had worse clinical characteristics, their probability of PFS and OS was higher compared with those treated with a non-CAB therapy. These results were due to a survival benefit with CAB in patients with an initial PSA level of 500–1000 ng/mL. Nadir PSA levels were significantly lower in CAB patients than in non-CAB patients with comparable initial serum PSA levels.
Conclusions
A small survival benefit for CAB in metastatic prostate cancer was demonstrated in a Japanese large-scale prospective cohort study. The clinical significance of nadir PSA levels following PADT was evident, but the predictive impact of PSA nadir on OS was different between CAB and non-CAB therapy.




Similar content being viewed by others
References
Huggins C, Hodges CV (1972) Studies on prostate cancer: i. the effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. CA Cancer J Clin 22(4):232–240
Mohler JL, Kantoff PW, Armstrong AJ, Bahnson RR, Cohen M, D’Amico AV, Eastham JA, Enke CA, Farrington TA, Higano CS, Horwitz EM, Kawachi MH, Kuettel M, Lee RJ, Macvicar GR, Malcolm AW, Miller D, Plimack ER, Pow-Sang JM, Richey S, Roach M 3rd, Rohren E, Rosenfeld S, Small EJ, Srinivas S, Stein C, Strope SA, Tward J, Walsh PC, Shead DA, Ho M (2014) National comprehensive cancer network (2013) Prostate cancer, version 1. J Natl Compr Canc Netw 11(12):1471–1479
Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T, Mason M, Matveev V, Wiegel T, Zattoni F, Mottet N, European Association of Urology (2014) EAU guidelines on prostate cancer. Part II: treatment of advanced, relapsing, and castration-resistant prostate cancer. Eur Urol 65(2):467–479
Prostate Cancer Trialists’ Collaborative Group (2000) Maximum androgen blockade in advanced prostate cancer: an overview of the randomised trials. Lancet 355(9241):1491–1498
Pagliarulo V, Bracarda S, Eisenberger MA, Mottet N, Schröder FH, Sternberg CN, Studer UE (2012) Contemporary role of androgen deprivation therapy for prostate cancer. Eur Urol 61(1):11–25
Akaza H, Usami M, Ogawa O, Kagawa S, Kitamura T, Tsukamoto T, Naito S, Hirao Y, Murai M, Yamanaka H (2004) Characteristics of patients with prostate cancer who have initially been treated by hormone therapy in Japan: J-CaP surveillance. Jap J Clin Oncol 34(6):329–336
Hinotsu S, Akaza H, Usami M, Ogawa O, Kagawa S, Kitamura T, Tsukamoto T, Naito S, Namiki M, Hirao Y, Murai M, Yamanaka H, Japan Study Group of Prostate Cancer (J-CaP) (2007) Current status of endocrine therapy for prostate cancer in Japan analysis of primary androgen deprivation therapy on the basis of data collected by J-CaP. Jap J Clin Oncol 37(10):775–781
Cooperberg MR, Hinotsu S, Namiki M, Ito K, Broering J, Carroll PR, Akaza H (2009) Risk assessment among prostate cancer patients receiving primary androgen deprivation therapy. J Clin Oncol 27(26):4306–4313
Kitagawa Y, Hinotsu S, Shigehara K, Nakashima K, Kawaguchi S, Yaegashi H, Mizokami A, Akaza H, Namiki M (2013) Japan Cancer of the Prostate Risk Assessment for combined androgen blockade including bicalutamide: clinical application and validation. Int J Urol 20(7):708–714
Shiota M, Yokomizo A, Takeuchi A, Imada K, Kiyoshima K, Inokuchi J, Tatsugami K, Naito S (2015) The oncological outcome and validation of Japan Cancer of the Prostate Risk Assessment score among men treated with primary androgen-deprivation therapy. J Cancer Res Clin Oncol 141(3):495–503
Yamaguchi Y, Hayashi Y, Ishizuya Y, Takeda K, Nakai Y, Arai Y, Nakayama M, Kakimoto K, Nishimura K (2015) A single-center study on predicting outcomes of primary androgen deprivation therapy for prostate cancer using the Japan Cancer of the Prostate Risk Assessment (J-CAPRA) score. Jpn J Clin Oncol 45(2):197–201
Akaza H, Hinotsu S, Usami M, Ogawa O, Kitamura T, Suzuki K, Tsukamoto T, Naito S, Namiki M, Hirao Y, Murai M (2013) Evaluation of primary androgen deprivation therapy in prostate cancer patients using J-CAPRA risk score. Prostate Int 1(2):81–88
Mastuoka T, Kawai K, Kimura T, Kojima T, Onozawa M, Miyazaki J, Nishiyama H, Hinotsu S, Akaza H (2014) Long-term outcomes of combined androgen blockade therapy in stage IV prostate cancer. J Cancer Res Clin Oncol 141(4):759–765
Sugihara T, Yu C, Kattan MW, Yasunaga H, Ihara H, Onozawa M, Hinotsu S, Akaza H (2014) Long-term survival of extremely advanced prostate cancer patients diagnosed with prostate-specific antigen over 500 ng/mL. Jpn J Clin Oncol 44(12):1227–1232
Hussain M, Tangen CM, Higano C, Schelhammer PF, Faulkner J, Crawford ED, Wilding G, Akdas A, Small EJ, Donnelly B, MacVicar G, Raghavan D, Southwest Oncology Group Trial 9346 (INT-0162) (2006) Absolute prostate-specific antigen value after androgen deprivation is a strong independent predictor of survival in new metastatic prostate cancer: data from southwest oncology group trial 9346 (INT-0162). J Clin Oncol 24(24):3984–3990
Kwak C, Jeon SJ, Park MS, Lee E, Lee SE (2002) Prognostic significance of the nadir prostate specific antigen level after hormone therapy for prostate cancer. J Urol 168(3):995–1000
Morote J, Trilla E, Esquena S, Abascal JM, Reventos J (2004) Nadir prostate-specific antigen best predicts the progression to androgen-independent prostate cancer. Int J Cancer 108(6):877–881
Kitagawa Y, Ueno S, Izumi K, Mizokami A, Hinotsu S, Akaza H, Namiki M (2014) Nadir prostate-specific antigen (PSA) level and tome to PSA nadir following primary androgen deprivation therapy as independent prognostic factors in a Japanese large-scale prospective cohort study (J-CaP). J Cancer Res Clin Oncol 140(4):673–679
International Union Against Cancer (1997) Urologic Tumors: Prostate. In: Sobin LH, Wittekind CH (eds) TNM Classification of Malignant Tumours, 5th edn. Wiley, New York, pp 170–173
Choueiri TK, Xie W, D’Amico AV, Ross RW, Hu JC, Pomerantz M, Regan MM, Taplin ME, Kantoff PW, Sartor O, Oh WK (2009) Time to prostate-specific antigen nadir independently predicts overall survival in patients who have metastatic hormone-sensitive prostate cancer treated with androgen-deprivation therapy. Cancer 115(5):981–987
Usami M, Akaza H, Arai Y, Hirano Y, Kagawa S, Kanetake H, Naito S, Sumiyoshi Y, Takimoto Y, Terai A, Yoshida H, Ohashi Y (2007) Bicalutamide 80 mg combined with a luteinizing hormone-releasing hormone agonist (LHRH-A) versus LHRH-A monotherapy in advanced prostate cancer: findings from a phase III randomized, double-blind, multicenter trial in Japanese patients. Prostate Cancer Prostat Dis 10(2):194–201
Akaza H, Hinotsu S, Usami M, Arai Y, Kanetake H, Naito S, Hirao Y, Study Group for the Combined Androgen Blockade Therapy of Prostate Cancer (2009) Combined androgen blockade with bicalutamide for advanced prostate cancer: long-term follow-up of a phase 3, double-blind, randomized study for survival. Cancer 115(15):3437–3445
Hori S, Jabbar T, Kachroo N, Vasconcelos JC, Robson CN, Gnanapragasam VJ (2011) Outcomes and predictive factors for biochemical relapse following primary androgen deprivation therapy in men with bone scan negative prostate cancer. J Cancer Res Clin Oncol 137(2):235–241
Acknowledgments
We thank Dr. Michiyuki Usami, Dr. Osamu Ogawa, Dr. Tadaichi Kitamura, Dr. Kazuhiro Suzuki, Dr. Taiji Tsukamoto, Dr. Seiji Naito, Dr. Yoshihiko Hirao, Dr. Masaru Murai, and the Japan Study Group of Prostate Cancer (J-CaP) for providing clinical data from the J-CaP.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Ethical standard
This study has been approved by Japanese multicenter institutional review board for research (UMIN 000000570). There is no patient identifying information included in this manuscript.
Rights and permissions
About this article
Cite this article
Kitagawa, Y., Ueno, S., Izumi, K. et al. Clinical outcomes and nadir prostate-specific antigen (PSA) according to initial PSA levels in primary androgen deprivation therapy for metastatic prostate cancer. World J Urol 34, 319–327 (2016). https://doi.org/10.1007/s00345-015-1621-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-015-1621-5