Abstract
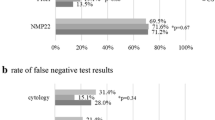
Despite several new urine markers urinary cytology remains the gold standard for the non-invasive detection of bladder carcinoma. The use of monoclonal antibodies against tumor associated antigens offers a promising approach to improve urinary cytology. The aim of this study was to compare fluorescence immunocytology (ImmunoCyt/Ucyt+ test), alone and in combination with the conventional cytology, with other urine markers. Urine samples from 126 patients undergoing cystoscopy were included in the study. Among them, 42 patients had urothelial carcinoma, two dysplasia, two other malignancies, and 78 had no evidence of bladder cancer. Urine samples were taken before any manipulation. We used the ImmunoCyt test and Papanicolaou staining for conventional cytology. The ImmunoCyt slides were examined under a fluorescence microscope. Evaluations of the tests were blinded to clinical and pathological data and were carried out by three independent observers. The results of cytology and ImmunoCyt were compared with the BTAstat, NMP22, Lewis X, 486p3/12, and Urovision tests. The sensitivity for the ImmunoCyt test was 78.3% and for conventional cytology 84.6%. The combination of ImmunoCyt and cytology showed a sensitivity of 89.1%. The specificity was 73.8% for the ImmunoCyt alone, 80.0% for the cytology, and 72.5% for the combination of ImmunoCyt and cytology. Sensitivities for the other tests were 68.8% for (FISH), 66.6% (BTA-Stat), 68.8% (486p3/12), 95.5% (Lewis X), and 71.1% for (NMP22). Specificity was 89.1% for (FISH), 78.2% (BTA-Stat), 76.4% (486p3/12), 32.8% (Lewis X), and 65.5% for (NMP22). Urinary cytology can be improved by immunostaining with monoclonal antibodies against tumor-associated antibodies. The combination of ImmunoCyt with conventional cytology offers a superior sensitivity to other commercial tests. The ImmunoCyt test provides a useful supplement to urinary cytology in the diagnosis of bladder cancer.
Similar content being viewed by others
References
Badalament RA, Fair WR, Whitmore WFJr, Melamed MR (1988) The relative value of cytometry and cytology in the management of bladder cancer: the Memorial Sloan-Kettering Cancer Center experience. Semin Urol 6:22–30
Leyh H, Marberger M, Conort P, Sternberg C, Pansadoro V, Pagano F, Bassi P, Boccon-Gibod L, Ravery V, Treiber U, Ishak L (1999) Comparison of the BTA stat test with voided urine cytology and bladder wash cytology in the diagnosis and monitoring of bladder cancer. Eur Urol 35:52–56
Chautard D, Daver A, Bocquillon V, Verriele V, Colls P, Bertrand G, Soret JY (2000) Comparison of the BTA TRAK test with voided urine cytology in the diagnosis and follow up of bladder tumors. Eur Urol 38:686–690
Ellis WJ, Blumenstein BA, Ishak LM, Enfield DL (1997) Clinical evaluation if the BTA TRAK assay and comparison to voided urine cytology and the Bard BTA test in patients with recurrent bladder tumors. The Multi Center Study Group. Urology 50:882–887
Pode D, Shapiro A, Wald M, Nativ O, Laufer M, Kaver I (1999) Noninvasive detection of bladder cancer with the BTA stat test. J Urol 161:443–446
Casella R, Huber P, Blochlinger A, Stoffel F, Dalquen P, Gasser TC, Lehmann K (2000) Urinary level of nuclear matrix protein 22 in the diagnosis of bladder cancer: experience with 130 patients with biopsy confirmed tumor. J Urol 164:1926–1928
Landman J, Chang Y, Kavaler E, Droller MJ, Liu BC (1998) Sensitivity and specificity of NMP22, telomerase, and BTA in the detection of human bladder cancer. Urology 52:398–302
Topsakal M, Karadeniz T, Anac M, Donmezer S, Besisik A (2001) Assessment of Fibrin-Fibrinogen Degradation Products (Accu Dx) test in bladder cancer patients. Eur Urol 39:287–289
Heicappell R, Schostak M, Mueller M, Miller K (2000) Evaluation of urinary bladder cancer antigen as a marker for diagnosis of transitional cell carcinoma of the bladder. Scand J Clin Lab Invest 60:275–282
Sanchez-Carbayo M, Herrero E, Megias J, Mira A, Espasa A, Chinchilla V, Soria F (1999) Initial evaluation of the diagnostic performance of the new urinary bladder cancer antigen test as a tumor marker for transitional cell carcinoma of the bladder. J Urol 161:1110–1115
Halling KC, King W, Sokolova IA, Meyer RG, Burkhardt HM, Halling AC, Cheville JC, Sebo TJ, Ramakumar S, Stewart CS, Pankratz S, O’Kane DJ, Seelig SA, Lieber MM, Jenkins RB (2000) A comparison of cytology and fluorescence in situ hybridization for the detection of urothelial carcinoma. J Urol 164:1768–1775
Jung I, Reeder JE, Cox C, Siddiqui JF, Oconnel MJ, Yang Z, Messing EM, Wheeless LL (1999) Chromosome 9 monosomy by fluorescence in situ hybridization of bladder irrigation specimens is a predictive of tumor recurrence. J Urol 162:1900–1903
Pycha A, Mian C, Posch B, Haitel A, El Baz M, Ghoneim MA, Marberger M (1998) Numerical aberrations of chromosomes 7, 9 and 17 in squamous cell and transitional cell cancer of the bladder: a comparative study performed by fluorescence in situ hybridization. J Urol 160:737–740
Klan R, Huland E, Baisch H, Huland H (1991) Sensitivity of urinary quantitative immunocytology with monoclonal antibody 486p3/12 in 241 unselected patients with bladder carcinoma. J Urol 145:495–497
Mian C, Pycha A, Wiener H, Haitel A, Lodde M, Marberger M (1999) ImmunoCyt: a new tool for detecting transitional cell cancer of the urinary tract. J Urol 161:1486–1489
Olsson H, Zackrisson B (2001) ImmunoCytTM a useful method in the follow-up protocol for patients with urinary bladder carcinoma. Scand J Urol Nephrol 35:280–282
Vriesema JLJ, Atsma F, Kiemeney ALM, Peelen WP, Witjes JA, Schalken JA (2001) Diagnostic efficacy of the ImmunoCyt test to detect superficial bladder cancer recurrence. Urology 58:367–371
Fradet Y, Lockhart C (1997) Performance characteristics of a new monoclonal antibody test for bladder cancer: ImmunoCyt. Can J Urol 4:400
Boman H, Hedelin H, Holmang S (2002) Four bladder tumor markers have a disappointingly low sensitivity for small size and low grade recurrence. J Urol 167:80–83
Lokeshwar VB, Soloway MS (2001) Current bladder tumor tests: does their projected utility fulfil clinical necessity? J Urol 165:1067–1977
Friedrich MG, Hellstern A, Toma, MI, Hammerer P, Huland H (2003) Are false positive urine tests in the diagnosis of bladder cancer really wrong—or do they predict tumour recurrence? Eur Urol 43:146–151
Pode D, Golijanin D, Sherman Y, Lebensart P, Shapiro A (1998) Immunostaining of Lewis X in cells from voided urine, cytopathology and ultrasound for noninvasive detection of bladder tumors. J Urol 159:389–392
Acknowledgements
The authors thank Mss Ellinghausen, Beutel and Bildhauer for technical assistance. In addition, the authors thank Dr. Jonathan C. Cheng for critically reading the manuscript
Author information
Authors and Affiliations
Corresponding author
Additional information
M.I. Toma, M.G. Friedrich contributed equally to this study
Rights and permissions
About this article
Cite this article
Toma, M.I., Friedrich, M.G., Hautmann, S.H. et al. Comparison of the ImmunoCyt test and urinary cytology with other urine tests in the detection and surveillance of bladder cancer. World J Urol 22, 145–149 (2004). https://doi.org/10.1007/s00345-003-0390-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-003-0390-8