Abstract
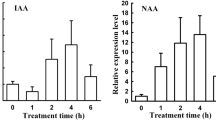
Overexpression of the IAGLU gene from maize (ZmIAAGLU) in Arabidopsis thaliana, under the control of the CaMV 35S promoter, inhibited root but not hypocotyl growth of seedlings in four different transgenic lines. Although hypocotyl growth of seedlings and inflorescence growth of mature plants was not affected, the leaves of mature plants were smaller and more curled as compared to wild-type and empty vector transformed plants. The rosette diameter in transgenic lines with higher ZmIAGLU expression was also smaller compared to the wild type. Free indole-3-acetic acid (IAA) levels in the transgenic plants were comparable to the wild type, even though a decrease in free IAA levels might be expected from overexpression of an IAA-conjugate–forming enzyme. IAA-glucose levels, however, were increased in transgenic lines compared to the wild type, indicating that the ZmIAGLU gene product is active in these plants. In addition, three different 35SZmIAGLU lines showed less inhibition of root growth when cultivated on increasing concentrations of IAA but not indole-3-butyric acid (IBA) and 2,4-dichlorophenoxyacetic acid (2,4-D). Feeding IAA to transgenic lines resulted in increased IAA-glucose synthesis, whereas the levels of IAA-aspartate and IAA-glutamine formed were reduced compared to the wild type. Our results show that IAA homeostasis can be altered by heterologous overexpression of a conjugate-forming gene from maize.







Similar content being viewed by others
References
Baldi BG, Maher BR, Cohen JD. 1989. Hydrolysis of indole-3-acetic acid esters exposed to mild alkaline conditions. Plant Physiol 91:9–12
Bandurski RS, Cohen JD, Slovin JP, Reinecke D. 1995. Auxin biosynthesis and metabolism. In: Davies PJ (ed) Plant hormones: physiology, biochemistry, and molecular biology. Kluwer Academic Publishers, Dordrecht, The Netherlands, 39–65
NM Barratt W Dong DA Gage V Magnus CD Town (1999) ArticleTitleMetabolism of exogenous auxin by Arabidopsis thaliana: identification of the conjugate N-(indol-3-ylacetyl)-glutamine and initiation of a mutant screen Physiol Plant 105 207–217 Occurrence Handle1:CAS:528:DyaK1MXis1GgtL8%3D Occurrence Handle10.1034/j.1399-3054.1999.105204.x
B Bartel GR Fink (1995) ArticleTitleILR1, an amidohydrolase that releases active indole-3-acetic acid from conjugates Science 268 1745–1748 Occurrence Handle1:CAS:528:DyaK2MXmsFCitro%3D Occurrence Handle10.1126/science.7792599 Occurrence Handle7792599
Bialek K, Cohen JD. 1986. Isolation and partial characterization of the major amide-linked conjugate of indole-3-acetic acid from Phaseolus vulgaris L. Plant Physiol 80:99–104
JJ Campanella J Ludwig-Mueller CD Town (1996) ArticleTitleIsolation and characterization of mutants of Arabidopsis thaliana with increased resistance to growth inhibition by IAA-amino acid conjugates Plant Physiol 112 735–746 Occurrence Handle1:CAS:528:DyaK28XmsFKksb8%3D Occurrence Handle10.1104/pp.112.2.735 Occurrence Handle8883385 Occurrence Handle157998
JJ Campanella J Ludwig-Müller V Bakllamaja V Sharma A Cartier (2003) ArticleTitleILR1 and sILR1 IAA amidohydrolase homologs differ in expression pattern and substrate specificity Plant Growth Regul 41 215–223 Occurrence Handle1:CAS:528:DC%2BD3sXps1anu7c%3D Occurrence Handle10.1023/B:GROW.0000007501.27878.aa
Chen K-H, Miller AN, Patterson GW, Cohen JD. 1988. A rapid and simple procedure for purification of indole-3-acetic acid prior to GC-SIM-MS analysis. Plant Physiol 86:822–825
J-C Chou WW Mulbry JD Cohen (1998) ArticleTitleThe gene for indole-3-acetyl-L-aspartic acid hydrolase from Enterobacter agglomerans: molecular cloning, nucleotide sequence, and expression in Escherichia coli Mol Gen Genet 259 172–178 Occurrence Handle1:CAS:528:DyaK1cXns1Ggtbk%3D Occurrence Handle10.1007/s004380050802 Occurrence Handle9747708
JD Cohen (1984) ArticleTitleConvenient apparatus for the generation of small amounts of diazomethane J. Chromatogr 303 193–196 Occurrence Handle1:CAS:528:DyaL2MXisVSkug%3D%3D Occurrence Handle10.1016/S0021-9673(01)96061-3
JD Cohen RS Bandurski (1982) ArticleTitleChemistry and physiology of the bound auxins Annu Rev Plant Physiol 33 403–430 Occurrence Handle1:CAS:528:DyaL38Xkt1Slt74%3D Occurrence Handle10.1146/annurev.pp.33.060182.002155
Cohen JD, Baldi BG, Slovin JP. 1986. 13C6-?Benzene ring?-indole-3-acetic acid: a new internal standard for quantitative mass spectral analysis of IAA in plants. Plant Physiol 80:14–19
JD Cohen JP Slovin AM Hendrickson (2003) ArticleTitleTwo genetically discrete pathways convert tryptophan to auxin: more redundancy in auxin biosynthesis Trends Plant Sci 8 197–199 Occurrence Handle1:CAS:528:DC%2BD3sXjvFWls7k%3D Occurrence Handle10.1016/S1360-1385(03)00058-X Occurrence Handle12758033
RT Davies DH Goetz J Lasswell MN Anderson B Bartel (1999) ArticleTitleIAR3 encodes an auxin conjugate hydrolase from Arabidopsis Plant Cell 11 365–376 Occurrence Handle1:CAS:528:DyaK1MXitlehs7o%3D Occurrence Handle10.1105/tpc.11.3.365 Occurrence Handle10072397 Occurrence Handle144182
W Domagalski A Schulze RS Bandurski (1987) ArticleTitleIsolation and characterization of esters of indole-3-acetic acid from the liquid endosperm of the horse chestnut (Aesculus species) Plant Physiol. 84 1107–1113 Occurrence Handle1:CAS:528:DyaL2sXlvFCqtr0%3D Occurrence Handle10.1104/pp.84.4.1107 Occurrence Handle11539676 Occurrence Handle1056736
Glass NL, Kosuge T. 1986. Cloning of the gene for indoleacetic acid-lysine synthetase from Pseudomonas syringae ssp. savastanoi. J Bacteriol 166:598–603
Graham R, Thornburg R. 1997. DNA sequence of UDP-glucose:indole-3-acetate-beta-D-glucosyltransferase from Arabidopsis thaliana (accession no. U81293) (PGR97-044). Plant Physiol 113:1004
WM Gray A Ostin G Sandberg CP Romano M Estelle (1998) ArticleTitleHigh temperature promotes auxin-mediated hypocotyl elongation in Arabidopsis Proc Natl Acad Sci USA. 95 7197–7202 Occurrence Handle1:CAS:528:DyaK1cXjslymt7w%3D Occurrence Handle10.1073/pnas.95.12.7197 Occurrence Handle9618562 Occurrence Handle22781
N Ilić J Normanly JD Cohen (1996) ArticleTitleQuantification of free plus conjugated indoleacetic acid in Arabidopsis requires correction for nonenzymatic conversion of indolic nitriles Plant Physiol 111 781–788 Occurrence Handle10.1104/pp.111.3.781
Iyer M, Slovin JP, Epstein E, Cohen JD. 1999. An unexpected change in free IAA levels and alteration of fruit ripening in tomatoes transformed with the iaglu gene. Plant Biol 1999, abstract no. 704
Iyer M, Slovin JP, Epstein E, Cohen JD. 2005. Transgenic tomato plants with a modified ability to synthesize indole-3-acetyl-β-1-0-D-glucose. J. plant Growth Regul 24 (this issue)
RG Jackson EK Lim Y Li M Kowalczyk G Sandberg et al. (2001) ArticleTitleIdentification and biochemical characterization of an Arabidopsis indole-3-acetic acid glucosyltransferase J Biol Chem 276 4350–4356 Occurrence Handle1:CAS:528:DC%2BD3MXht1GnsLc%3D Occurrence Handle10.1074/jbc.M006185200 Occurrence Handle11042207
RG Jackson M Kowalczyk Y Li G Higgins J Ross et al. (2002) ArticleTitleOver-expression of an Arabidopsis gene encoding a glucosyltransferase of indole-3-acetic acid: phenotypic characterisation of transgenic lines Plant J 32 573–583 Occurrence Handle1:CAS:528:DC%2BD38Xps1Kru7s%3D Occurrence Handle10.1046/j.1365-313X.2002.01445.x Occurrence Handle12445128
Jakubowska A, Kowalczyk S, Leznicki AJ. 1993. Enzymatic hydrolysis of 4-O and 6-O-indolo-3-ylacetyl-beta-D-glucose in plant tissues. J Plant Physiol 142:61–66
CP Keller R Stahlberg LS Barkawi JD Cohen (2004) ArticleTitleLong-term inhibition by auxin of leaf blade expansion in bean and Arabidopsis Plant Physiol. 134 1217–1226 Occurrence Handle1:CAS:528:DC%2BD2cXis1Sjtbw%3D Occurrence Handle10.1104/pp.103.032300 Occurrence Handle14988474 Occurrence Handle389946
JM Kesy RS Bandurski (1990) ArticleTitlePartial purification and characterization of indol-3-yl-acetylglucose-myo-inositol indol-3-yl-acetyltransferase (indoleacetic acid-inositol synthase) Plant Physiol. 94 1598–1604 Occurrence Handle1:CAS:528:DyaK3MXhtFOgtLw%3D Occurrence Handle10.1104/pp.94.4.1598 Occurrence Handle11537469 Occurrence Handle1077426
DG Kneller FE Cohen R Langridge (1990) ArticleTitleImprovements in protein secondary structure prediction by an enhanced neural network J Mol Biol 214 171–182 Occurrence Handle1:CAS:528:DyaK3cXkvFCgtr4%3D Occurrence Handle10.1016/0022-2836(90)90154-E Occurrence Handle2370661
J Kyte RF Doolittle (1982) ArticleTitleA simple method for displaying the hydropathic character of a protein J Mol Biol 157 105–142 Occurrence Handle1:CAS:528:DyaL38Xks1yjtro%3D Occurrence Handle10.1016/0022-2836(82)90515-0 Occurrence Handle7108955
UK Laemmli (1970) ArticleTitleCleavage of structural proteins during the assembly of the head of bacteriophage T4 Nature 227 680–685 Occurrence Handle1:CAS:528:DC%2BD3MXlsFags7s%3D Occurrence Handle10.1038/227680a0 Occurrence Handle5432063
S LeClere R Tellez RA Rampey SPT Matsuda B Bartel (2002) ArticleTitleCharacterization of a family of IAA-amino acid conjugate hydrolases from Arabidopsis J Biol Chem 277 20446–20452 Occurrence Handle1:CAS:528:DC%2BD38XkslWgtL8%3D Occurrence Handle10.1074/jbc.M111955200 Occurrence Handle11923288
AJ Leznicki RS Bandurski (1988) ArticleTitleEnzymic synthesis of indole-3-acetyl-1-0-β-D-glucose. II. Metabolic characteristics of the enzyme Plant Physiol 88 1481–1485 Occurrence Handle1:CAS:528:DyaL1MXntFKhsA%3D%3D Occurrence Handle10.1104/pp.88.4.1481 Occurrence Handle11537439 Occurrence Handle1055783
K Ljung RP Bhalerao G Sandberg (2001) ArticleTitleSites and homeostatic control of auxin biosynthesis in Arabidopsis during vegetative growth Plant J 28 465–474 Occurrence Handle1:CAS:528:DC%2BD38XmtVGhuw%3D%3D Occurrence Handle10.1046/j.1365-313X.2001.01173.x Occurrence Handle11737783
R Löw T Rausch (1994) ArticleTitleSensitive nonradioactive Northern blots using alkaline transfer of total RNA and PCR-amplified biotinylated probes BioTechniques 17 1026–1030 Occurrence Handle7873169
Ludwig-Müller J, Hilgenberg W. 1992. Tryptophan oxidizing enzyme and basic peroxidase isoenzymes in Arabidopsis thaliana (L.) Heynh.: are they identical? Plant Cell Physiol 33:1115–1125
J Ludwig-Müller S Sass EG Sutter M Wodner E Epstein (1993) ArticleTitleIndole-3-butyric acid in Arabidopsis thaliana. I. Identification and quantification Plant Growth Regul 13 179–187 Occurrence Handle10.1007/BF00024260
Ludwig-Müller J, Schubert B, Pieper K. 1995. Regulation of IBA synthetase by drought stress and abscisic acid. J Exp Bot 46:423–432
J Ludwig-Müller E Epstein W Hilgenberg (1996) ArticleTitleAuxin-conjugate hydrolysis in Chinese cabbage: characterization of an amidohydrolase and its role during the clubroot disease Physiol Plant 97 627–634 Occurrence Handle10.1111/j.1399-3054.1996.tb00525.x
L Michalczuk RS Bandurski (1982) ArticleTitleEnzymic synthesis of 1-O-indol-3-ylacetyl-β-D-glucose and indol-3-ylacetyl-myo-inositol Biochem J 207 273–281 Occurrence Handle1:CAS:528:DyaL3sXhslGns7Y%3D Occurrence Handle10.1042/bj2070273 Occurrence Handle6218801 Occurrence Handle1153857
Murashige T, Skoog F. 1962. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473-497
JH Oetiker G Aeschbacher (1997) ArticleTitleTemperature-sensitive plant cells with shunted indole-3-acetic acid conjugation Plant Physiol 114 1385–1395 Occurrence Handle1:CAS:528:DyaK2sXlsleisbc%3D Occurrence Handle10.1104/pp.114.4.1385 Occurrence Handle12223777 Occurrence Handle158431
A Östin M Kowalczyk RP Bhalerao G Sandberg (1998) ArticleTitleMetabolism of indole-3-acetic acid in Arabidopsis Plant Physiol 118 285–296 Occurrence Handle10.1104/pp.118.1.285 Occurrence Handle9733548 Occurrence Handle34867
Peterson GL. 1977. A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal Biochem 83:346–356
Rampey RA, LeClere S, Kowalczyk M, Ljung K, Sandberg G, Bartel B. 2004. A family of auxin-conjugate hydrolases that contributes to free indole-3-acetic acid levels during Arabidopsis germination 135:1–11
FF Roberto H Klee F White R Nordeen T Kosuge (1990) ArticleTitleExpression and fine structure of the gene encoding indole-3-acetyl-L-lysine synthetase from Pseudomonas savastanoi Proc Natl Acad Sci USA 87 5797–5801 Occurrence Handle1:CAS:528:DyaK3MXhtlWq Occurrence Handle10.1073/pnas.87.15.5797 Occurrence Handle2377619 Occurrence Handle54415
CP Romano MB Hein HJ Klee (1991) ArticleTitleInactivation of auxin in tobacco transformed with the indoleacetic acid–lysine synthetase gene of Pseudomonas savastanoi Genes Dev 5 438–446 Occurrence Handle1:CAS:528:DyaK3MXhsl2ltL4%3D Occurrence Handle10.1101/gad.5.3.438 Occurrence Handle2001843
Sambrook J, Russell D 2001. Molecular cloning: a laboratory manual (3rd edition). Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
C Simmons F Migliaccio P Masson T Caspar D Soll (1995) ArticleTitleA novel root gravitropism mutant of Arabidopsis thaliana exhibiting altered auxin physiology Physiol Plant 93 790–798 Occurrence Handle1:CAS:528:DyaK2MXovFCms7w%3D Occurrence Handle10.1111/j.1399-3054.1995.tb05133.x Occurrence Handle11540162
Slovin JP, Bandurski RS, Cohen JD. 1999. Auxin. In: Hoykaas PJJ, Hall MA, Libbenga KR, eds,Biochemistry and molecular biology of plant hormones. Elsevier, Amsterdam, 115–140
Spena A, Prinsen E, Fladung M, Schulze SC, van Onckelen H. 1991. The indoleacetic acid-lysine synthetase gene of Pseudomonas syringae subsp. savastanoi induces developmental alterations in transgenic tobacco and potato plants. Mol Gen Genet 227:205–212
PE Staswick I Tiryaki ML Rowe (2002) ArticleTitleJasmonate response locus JAR1 and several related Arabidopsis genes encode enzymes of the firefly luciferase superfamily that show activity on jasmonic, salicylic and indole-3-acetic acids in an assay for adenylation Plant Cell 14 1405–1415 Occurrence Handle1:CAS:528:DC%2BD38Xlt1alsbo%3D Occurrence Handle10.1105/tpc.000885 Occurrence Handle12084835 Occurrence Handle150788
PE Staswick B Serban M Rowe I Tiryaki MT Maldonado et al. (2005) ArticleTitleCharacterization of an Arabidopsis enzyme family that conjugates amino acids to indole-3-acetic acid Plant Cell 17 616–627 Occurrence Handle1:CAS:528:DC%2BD2MXhsFKqs7g%3D Occurrence Handle10.1105/tpc.104.026690 Occurrence Handle15659623 Occurrence Handle548830
JD Szerszen K Szczyglowski RS Bandurski (1994) ArticleTitleiaglu, a gene from Zea mays involved in conjugation of growth hormone indole-3-acetic acid Science 265 1699–1701 Occurrence Handle1:CAS:528:DyaK2MXps1SktA%3D%3D Occurrence Handle10.1126/science.8085154 Occurrence Handle8085154
E Sztein JD Cohen I de Garcia la Fuente TJ Cooke (1999) ArticleTitleAuxin metabolism in mosses and liverworts Am J Bot 86 1544–1555 Occurrence Handle1:CAS:528:DyaK1MXotVOrsrY%3D Occurrence Handle10.2307/2656792
YY Tam J Normanly (2002) ArticleTitleOverexpression of a bacterial indole-3-acetyl-L-aspartic acid hydrolase in Arabidopsis thaliana Physiol Plant 115 513–522 Occurrence Handle1:CAS:528:DC%2BD38XlvFKhtrg%3D Occurrence Handle10.1034/j.1399-3054.2002.1150405.x Occurrence Handle12121457
YY Tam E Epstein J Normanly (2000) ArticleTitleCharacterization of auxin conjugates in Arabidopsis. Low steady-state levels of indole-3-acetyl-aspartate, indole-3-acetyl-glutamate, and indole-3-acetyl-glucose Plant Physiol 123 589–595 Occurrence Handle1:CAS:528:DC%2BD3cXktlWntLc%3D Occurrence Handle10.1104/pp.123.2.589 Occurrence Handle10859188 Occurrence Handle59026
Valvekens D, van Montagu M, van Lijsebettens M. 1988. Agrobacterium tumefaciens–mediated transformation of Arabidopsis thaliana root explants by using kanamycin selection. Proc Natl Acad Sci USA 85:5536–5540
A Walz S Park JP Slovin J Ludwig-Müller Y Momonoki et al. (2002) ArticleTitleA gene encoding a protein modified by the phytohormone indoleacetic acid Proc Natl Acad Sci USA 99 1718–1723 Occurrence Handle1:CAS:528:DC%2BD38Xht1Cltb8%3D Occurrence Handle10.1073/pnas.032450399 Occurrence Handle11830675 Occurrence Handle122257
Yamagishi E, Gong Z, Yamazaki M, Saito K. 1998. Molecular cloning of a cDNA encoding a novel UDP-glucose glucosyltransferase homologue from Arabidopsis thaliana (accession no. AB016819) (PGR98-187). Plant Physiol 118:1102
Acknowledgments
This work was supported by a grant from the Deutsche Forschungsgemeinschaft to J. Ludwig-Müller (Lu 500/2-3) and U.S. Department of Energy grant DE-FG02-00ER15079 and U.S. National Science Foundation grant IBN 0111530 to J. Cohen. The ZmIAAGLU cDNA was obtained from Prof. Dr. Robert S. Bandurski. The maize IAA-glucose synthase antibody was a gift from Dr. Mariusz Kowalczyk. Vectors pCGN1761ENX and pCIB200 were provided by Dr. Eric Ward. We thank Mrs. Kerstin Pieper for excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ludwig-Müller, J., Walz, A., Slovin, J.P. et al. Overexpression of Maize IAGLU in Arabidopsis thaliana Alters Plant Growth and Sensitivity to IAA but not IBA and 2,4-D. J Plant Growth Regul 24, 127–141 (2005). https://doi.org/10.1007/s00344-004-0006-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-004-0006-6