Abstract
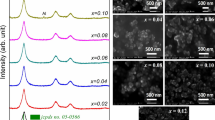
A comprehensive study aimed to investigate the role of sodium hydroxide precursor concentration on the physical properties of the ZnO nanoparticles. A simple wet chemical approach was employed to synthesize ZnO nanoparticles by keeping the Zn precursor concentration constant and sodium hydroxide concentration varied. XRD analysis confirms the prepared ZnO nanoparticles having a hexagonal wurtzite phase whose crystallite sizes are from 24 to 34 nm. Good crystalline ZnO nanoparticles are realized when the Zn-to-OH precursor concentration is greater than 1:1 molar ratio. UV–Vis spectroscopic studies reveal the optical bandgap of the ZnO nanoparticles can be tailored considerably by varying the alkali hydroxide concentration. The variation in Urbach energy values emphasizes the existence of localized states originating from the lattice disorder and defects. The room temperature photoluminescence analysis confirms the presence of defects in the prepared nanoparticles. Surface morphological investigation of the synthesized nanoparticle samples was investigated using SEM.









Similar content being viewed by others
References
F. Rahman, Zinc oxide light-emitting diodes: a review. Opt. Eng. 58, 1 (2019). https://doi.org/10.1117/1.oe.58.1.010901
S.S. Shin, S.J. Lee, S.I. Seok, Exploring wide bandgap metal oxides for perovskite solar cells. APL Mater. 7, 022401 (2019). https://doi.org/10.1063/1.5055607
O.B. Ajayi, M.S. Akanni, J.N. Lambi, C. Jeynes, J.F. Watts, Compositional studies of various metal oxide coatings on glass. Thin Solid Films 185, 123–136 (1990). https://doi.org/10.1016/0040-6090(90)90012-3
J.C. Védrine, Heterogeneous catalysis on metal oxides. Catalysts 7, 341 (2017)
R. Mallampati, S. Valiyaveettil, Biomimetic metal oxides for the extraction of nanoparticles from water. Nanoscale 5, 3395–3399 (2013). https://doi.org/10.1039/c3nr34221b
C. Wang, L. Yin, L. Zhang, D. Xiang, R. Gao, Metal oxide gas sensors: sensitivity and influencing factors. Sensors 10, 2088–2106 (2010)
N. Chidhambaram, K. Ravichandran, Fabrication of ZnO/g-C3N4 nanocomposites for enhanced visible light driven photocatalytic activity. Mater. Res. Express 4, 075037 (2017). https://doi.org/10.1088/2053-1591/aa7abd
P.A. Rodnyi, I.V. Khodyuk, Optical and luminescence properties of zinc oxide (Review). Opt. Spectrosc. (English Transl) Opt. Spektrosk. 111, 776–785 (2011). https://doi.org/10.1134/s0030400x11120216
N. Chidhambaram, Augmented antibacterial efficacies of the aluminium doped ZnO nanoparticles against four pathogenic bacteria. Mater. Res. Express 6, 075061 (2019). https://doi.org/10.1088/2053-1591/ab1804
J.N. Hasnidawani, H.N. Azlina, H. Norita, N.N. Bonnia, S. Ratim, E.S. Ali, Synthesis of ZnO nanostructures using sol–gel method synthesis of ZnO nanostructures using sol–gel method. Procedia Chem. 19, 211–216 (2016). https://doi.org/10.1016/j.proche.2016.03.095
J. Li, Z. Wu, Y. Bao, Y. Chen, C. Huang, N. Li, S. He, Z. Chen, Wet chemical synthesis of ZnO nanocoating on the surface of bamboo timber with improved mould-resistance. J. Saudi Chem. Soc. 21, 920–928 (2017). https://doi.org/10.1016/j.jscs.2015.12.008
S.S. Kumar, P. Venkateswarlu, V.R. Rao, G.N. Rao, Synthesis, characterization and optical properties of zinc oxide nanoparticles. Int. Nano Lett. 4, 2–6 (2014). https://doi.org/10.1186/2228-5326-3-30
H.S.M. Abd-Rabboh, M. Eissa, S.K. Mohamed, M.S. Hamdy, Synthesis of ZnO by thermal decomposition of different precursors: photocatalytic performance under UV and visible light illumination. Mater. Res. Express (2019). https://doi.org/10.1088/2053-1591/ab04ff
S.T. Navale, V.V. Jadhav, K.K. Tehare, R.U.R. Sagar, C.S. Biswas, M. Galluzzi, W. Liang, V.B. Patil, R.S. Mane, F.J. Stadler, Solid-state synthesis strategy of ZnO nanoparticles for the rapid detection of hazardous Cl2. Sens. Actuat. B Chem. 238, 1102–1110 (2017). https://doi.org/10.1016/j.snb.2016.07.136
D.B. Bharti, A.V. Bharati, Synthesis of ZnO nanoparticles using a hydrothermal method and a study its optical activity. Luminescence 32, 317–320 (2017). https://doi.org/10.1002/bio.3180
P. Rai, W.K. Kwak, Y.T. Yu, Solvothermal synthesis of ZnO nanostructures and their morphology-dependent gas-sensing properties. ACS Appl. Mater. Interfaces 5, 3026–3032 (2013). https://doi.org/10.1021/am302811h
K. Ravichandran, N. Chidhambaram, S. Gobalakrishnan, Copper and Graphene activated ZnO nanopowders for enhanced photocatalytic and antibacterial activities. J. Phys. Chem. Solids 93, 82–90 (2016). https://doi.org/10.1016/j.jpcs.2016.02.013
S.V. Elangovan, V. Chandramohan, N. Sivakumar, T.S. Senthil, Synthesis and characterization of ZnO nanoparticles at different molarity concentrations for photocatalytic applications. Desalin. Water Treat. 57, 9671–9678 (2016). https://doi.org/10.1080/19443994.2015.1035340
A. Bagabas, A. Alshammari, M.F.A. Aboud, H. Kosslick, Room-temperature synthesis of zinc oxide nanoparticles in different media and their application in cyanide photodegradation. Nanoscale Res. Lett. 8, 1–10 (2013). https://doi.org/10.1186/1556-276X-8-516
B. Choudhury, M. Dey, A. Choudhury, Defect generation, d–d transition, and band gap reduction in Cu-doped TiO2 nanoparticles. Int. Nano Lett. 3, 25 (2013). https://doi.org/10.1186/2228-5326-3-25
O.R. Vasile, E. Andronescu, C. Ghitulica, B.S. Vasile, O. Oprea, E. Vasile, R. Trusca, Synthesis and characterization of nanostructured zinc oxide particles synthesized by the pyrosol method. J. Nanopart. Res. 14, 1–13 (2012). https://doi.org/10.1007/s11051-012-1269-7
J.H. Lin, R.A. Patil, R.S. Devan, Z.A. Liu, Y.P. Wang, C.H. Ho, Y. Liou, Y.R. Ma, Photoluminescence mechanisms of metallic Zn nanospheres, semiconducting ZnO nanoballoons, and metal-semiconductor Zn/ZnO nanospheres. Sci. Rep. 4, 1–8 (2014). https://doi.org/10.1038/srep06967
H. Zeng, G. Duan, Y. Li, S. Yang, X. Xu, W. Cai, Blue luminescence of ZnO nanoparticles based on non-equilibrium processes: defect origins and emission controls. Adv. Funct. Mater. 20, 561–572 (2010). https://doi.org/10.1002/adfm.200901884
L. Dai, X.L. Chen, W.J. Wang, T. Zhou, B.Q. Hu, Growth and luminescence characterization of large-scale zinc oxide nanowires. J. Phys. Condens. Matter 15, 2221–2226 (2003). https://doi.org/10.1088/0953-8984/15/13/308
N.H. Alvi, K. ul Hasan, O. Nur, M. Willander, The origin of the red emission in n-ZnO nanotubes/p-GaN white light emitting diodes. Nanoscale Res. Lett. 6, 1–7 (2011). https://doi.org/10.1186/1556-276X-6-130
U.S.U. Thampy, C.R. Krishna, C.V. Reddy, B. Babu, Y.P. Reddy, P.S. Rao, R.V.S.S.N. Ravikumar, Spectral investigations on Cu2+-doped ZnO nanopowders. Appl. Magn. Reson. 41, 69–78 (2011). https://doi.org/10.1007/s00723-011-0234-4
M. Vafaee, M.S. Ghamsari, Preparation and characterization of ZnO nanoparticles by a novel sol–gel route. Mater. Lett. 61, 3265–3268 (2007). https://doi.org/10.1016/j.matlet.2006.11.089
S. Kumar, K. Asokan, R.K. Singh, S. Chatterjee, D. Kanjilal, A.K. Ghosh, Investigations on structural and optical properties of ZnO and ZnO:Co nanoparticles under dense electronic excitations. RSC Adv. 4, 62123–62131 (2014). https://doi.org/10.1039/c4ra09937k
K. Ravichandran, N. Chidhambaram, T. Arun, S. Velmathi, S. Gobalakrishnan, Realizing cost-effective ZnO:Sr nanoparticles@graphene nanospreads for improved photocatalytic and antibacterial activities. RSC Adv. 6, 67575–67585 (2016). https://doi.org/10.1039/C6RA08697G
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nilavazhagan, S., Anbuselvan, D., Santhanam, A. et al. Effect of an alkali hydroxide concentration on the structural, optical, and surface morphological properties of ZnO nanoparticles. Appl. Phys. A 126, 279 (2020). https://doi.org/10.1007/s00339-020-3462-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-020-3462-3