Abstract
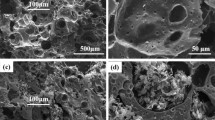
Recently, self-assembly method was applied for preparation of graphene foam. However, it is still a great challenge to obtain a three-dimensional graphene network with high performance (e.g., low density, high mechanical strength and high conductivity together) for the self-assembly method. Herein, a modified self-assembly method applied for preparation of graphene foam was investigated, in which, L-ascorbic acid and HI were firstly chosen as the reducing agent, and further reduced by hydrazine hydrate. The results demonstrated that the graphene foam showed high compressive strength (ca. 320 kPa), high electrical conductivity (20.6 S/m) and low density (14.7 mg/cm−1). Especially, the obtained compressive strength (ca. 320 kPa) is the highest value compared to the data of graphene foam reported in previous works. This phenomenon may be due to following three reasons: (1) the reaction between hydrazine hydrate and graphene brought some covalent bonds among graphene sheets; (2) graphene foam was achieved by high hydrophobicity and electrostatic repulsion which inhibit the restacking of graphene sheets; (3) the removal of the oxygen groups by hydrazine hydrate efficiently restores conjugation of sp2 regions and the π–π interaction in the cross-linking sites, which tightly bonds the sheets together. The obtained graphene foam not only had good porous structure and mechanical strength, but also showed excellent satisfactory double-layer capacitive behavior with good electrochemical cyclic stability and high specific capacitance of 171.0 F/g for application in electrode of supercapacitors and absorption capacities for the removal of various oils and dyes from water.









Similar content being viewed by others
References
Z.H. Tang, S.L. Shen, J. Zhuang, X. Wang, Angew. Chem. Int. Ed. 49, 4603 (2010)
Z.S. Wu, S.B. Yang, Y. Sun, K. Parvez, X.L. Feng, K. Mullen, J. Am. Chem. Soc. 134, 9082 (2012)
L. Estevez, A. Kelarakis, Q.M. Gong, E.H. Da’as, E.P. Giannelis, J. Am. Chem. Soc. 133, 6122 (2011)
Y.X. Xu, K.X. Sheng, C. Li, G.Q. Shi, ACS Nano 4, 4324 (2010)
X.T. Zhang, Z.Y. Sui, B. Xu, S.F. Yue, Y.J. Luo, W.C. Zhan, B. Liu, J. Mater. Chem. 21, 6494 (2011)
H.P. Cong, X.C. Ren, P. Wang, S.H. Yu, ACS Nano 6, 2693 (2012)
X. Jiang, Y.W. Ma, J.J. Li, Q.L. Fan, W. Huang, J. Phys. Chem. C 114, 22462 (2010)
Z.P. Chen, W.C. Ren, L.B. Gao, B.L. Liu, S.F. Pei, H.M. Cheng, Nat. Mater. 10, 424 (2011)
X.H. Cao, Y.M. Shi, W.H. Shi, G. Lu, X. Huang, Q.Y. Yan, Q.C. Zhang, H. Zhang, Small 7, 3163 (2011)
X.C. Dong, H. Xu, X.W. Wang, Y.X. Huang, M.B. Chan-Park, H. Zhang, L.H. Wang, W. Huang, P. Chen, ACS Nano 4, 3206 (2012)
X.C. Dong, X.W. Wang, L.H. Wang, H. Song, H. Zhang, W. Huang, P. Chen, A.C.S. Appl, Mater. Interfaces 4, 3129 (2012)
W.F. Chen, L.F. Yan, Nanoscale 3, 3132 (2011)
L.B. Zhang, G.Y. Chen, M. NejibHedhili, H.N. Zhang, P. Wang, Nanoscale 4, 7038 (2012)
S.F. Pei, J.P. Zhao, J.H. Du, W.C. Ren, H.M. Cheng, Carbon 48, 4466 (2010)
X.Z. Wu, J. Zhou, W. Xing et al., J. Mater. Chem. 22, 23186 (2012)
W.S. Hummers, R.E. Offeman, J. Am. Chem. Soc. 80, 1339 (1958)
X. Zhang, Z. Sui, B. Xu, S. Yue, Y. Luo, W. Zhan, B. Liu, J. Mater. Chem. 21, 6494 (2011)
Z. Sui, X. Zhang, Y. Lei, Y. Luo, Carbon 49, 4314 (2011)
J.H. Li, J.Y. Li, H. Meng, J. Mater. Chem. A 2, 2934 (2014)
H. Hu, Z.B. Zhao, W.B. Wan, Y. Gogotsi, J.S. Qiu, Adv. Mater. 25, 2219 (2013)
T. Wu, M.X. Chen, L. Zhang, X.Y. Xu, Y. Liu, J. Yan, W. Wang, J.P. Gao, J. Mater. Chem. A 1, 7612 (2013)
P. Lian, X. Zhu, S. Liang, Z. Li, W. Yang, H. Wang, Electrochim. Acta 55, 3909 (2010)
Z. Han, Z.H. Tang, P. Li, G.Z. Yang, Q.B. Zheng, J.H. Yang, Nanoscale 5, 5462 (2013)
H.Y. Sun, Z. Xu, C. Gao, Adv. Mater. 25, 2554 (2013)
S.T. Nguyena, H.T. Nguyen, A. Rinaldi, N.P.V. Nguyena, Z. Fan, H.M. Duong, Colloids and surfaces a: physicochem. Eng. Aspects 414, 352 (2012)
A.C. Ferrari, J.C. Meyer, V. Scardaci, C. Casiraghi, M. Lazzeri, F. Mauri, S. Piscanec, D. Jiang, K.S. Novoselov, S. Roth, A.K. Geim, Physical review letter 97, 187401 (2006)
Y.Y. Sun, W.H. Zhang, D.S. Li, L. Gao, C.L. Hou, Y.H. Zhang, Y.Q. Liu, J. Alloys Compd. 649, 579 (2015)
T.T. Liu, G.Z. Zhao, W.H. Zhang, H.J. Chi, C.L. Hou, Y.Y. Sun, J. Porous Mater. 22, 1573 (2015)
J. Zhang, H. Yang, G. Shen, P. Cheng, S. Guo, Reduction of graphene oxide via L-ascorbic acid. Chem. Commun. 46, 1112 (2010)
S.F. Pei, J.P. Zhao, J.H. Du, W.C. Ren, H.M. Cheng, Carbon 48, 4466 (2010)
I.K. Moon, J.H. Lee, R.S. Ruoff, H.Y. Lee, Nat. Commun. 1, 73 (2010)
H. Wang, D.S. Zhang, T.T. Yan, X.R. Wen, J.P. Zhang, L.Y. Shi, Q.D. Zhong, J. Mater. Chem. A 1, 11778–11789 (2013)
H.W. Liang, Q.F. Guan, L.F. Chen, Z. Zhu, W.J. Zhang, S.H. Yu, Angew. Chem. Int. Ed. 124, 5191 (2012)
E.A. Vogler, Adv. Colloids Interfaces 74, 69 (1998)
Acknowledgments
The authors are grateful for the support by National Natural Science Foundation of China under grants (11202006 and 11202007) and the Shanxi provincial natural science foundation of China (2014021018-6).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhang, W., Sun, Y., Liu, T. et al. Preparation of graphene foam with high performance by modified self-assembly method. Appl. Phys. A 122, 259 (2016). https://doi.org/10.1007/s00339-016-9684-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-016-9684-8