Abstract
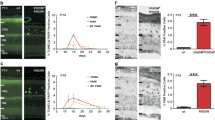
Retinitis pigmentosa (RP) is a neurodegenerative disorder that causes irreversible vision loss in over 1.5 million individuals world-wide. The genetic heterogeneity of RP necessitates a broad therapy that is able to provide treatment in a gene- and mutation- non-specific manner. In this study, we identify the therapeutic benefits of metabolic reprogramming by targeting pyruvate kinase M2 (PKM2) in a Pde6β preclinical model of RP. The genetic contributions of PKM2 inhibition in retinal degeneration were evaluated through histology and electroretinogram (ERG) followed by a statistical analysis using a linear regression model. Notably, PKM2 ablation resulted in thicker retinal layers in Pde6β-mutated mice as compared to the controls, suggesting greater photoreceptor survival. Consistent with these anatomical findings, ERG analyses revealed that the maximum b-wave is on average greater in Pkm2 knockout mice than in mice with intact Pkm2, indicating enhanced photoreceptor function. These rescue phenotypes from Pkm2 ablation in a preclinical model of RP indicate that a metabolome reprogramming may be useful in treating RP.




Similar content being viewed by others
Data availability
All data in this manuscript are available for share. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Anastasiou D (2011) Inhibition of pyruvate kinase M2 by reactive oxygen species contributes to cellular antioxidant responses. Science 334:1278–1283 https://doi.org/10.1126/science.1211485
Bainbridge JW (2008) Effect of gene therapy on visual function in Leber's congenital amaurosis. N Engl J Med 358:2231–2239 https://doi.org/10.1056/NEJMoa0802268
Barabas P, Cutler Peck C, Krizaj D (2010) Do calcium channel blockers rescue dying photoreceptors in the Pde6b ( rd1) mouse? Adv Exp Med Biol 664:491–499 https://doi.org/10.1007/978-1-4419-1399-9_56
Bird AC (1995) Retinal photoreceptor dystrophies: the LI Edward Jackson Memorial Lecture. Am J Opthalmol 119:543–562
Campochiaro PA, Mir TA (2018) The mechanism of cone cell death in Retinitis Pigmentosa. Prog Retin Eye Res 62:24–37. https://doi.org/10.1016/j.preteyeres.2017.08.004
Chinchore Y, Begaj T, Wu D, Drokhlyansky E, Cepko CL (2017) Glycolytic reliance promotes anabolism in photoreceptors. Elife. https://doi.org/10.7554/eLife.25946
Chinskey ND, Besirli CG, Zacks DN (2014) Retinal cell death and current strategies in retinal neuroprotection. Curr Opin Ophthalmol 25:228–233. https://doi.org/10.1097/ICU.0000000000000043
Cruz da L (2013) The Argus II epiretinal prosthesis system allows letter and word reading and long-term function in patients with profound vision loss. Br J Ophthalmol 97:632–636. https://doi.org/10.1136/bjophthalmol-2012-301525
Cruz da L (2016) Five-year safety and performance results from the argus II retinal prosthesis system clinical trial. Ophthalmology 123:2248–2254. https://doi.org/10.1016/j.ophtha.2016.06.049
Daiger SP, Bowne SJ, Sullivan LS (2007) Perspective on genes and mutations causing retinitis pigmentosa. Arch Ophthalmol 125:151–158. https://doi.org/10.1001/archopht.125.2.151
Davis RJ (2008) Functional rescue of degenerating photoreceptors in mice homozygous for a hypomorphic cGMP phosphodiesterase 6 b allele (Pde6bH620Q). Investig Ophthalmol Vis Sci 49:5067–5076. https://doi.org/10.1167/iovs.07-1422
Demetrius LA, Driver J (2013) Alzheimer's as a metabolic disease. Biogerontology 14:641–649. https://doi.org/10.1007/s10522-013-9479-7
Demetrius LA, Simon DK (2012) An inverse-Warburg effect and the origin of Alzheimer's disease. Biogerontology 13:583–594. https://doi.org/10.1007/s10522-012-9403-6
Dong G, Mao Q, Xia W, Xu Y, Wang J, Xu L, Jiang F (2016) PKM2 and cancer: the function of PKM2 beyond glycolysis. Oncol Lett 11:1980–1986. https://doi.org/10.3892/ol.2016.4168
Du J (2016) Reductive carboxylation is a major metabolic pathway in the retinal pigment epithelium. Proc Natl Acad Sci USA 113:14710–14715. https://doi.org/10.1073/pnas.1604572113
Duncan JL (2018) Inherited retinal degenerations: current landscape and knowledge gaps. Transl Vis Sci Technol 7:6. https://doi.org/10.1167/tvst.7.4.6
Dutta S, Sengupta P (2016) Men and mice: relating their ages. Life Sci 152:244–248. https://doi.org/10.1016/j.lfs.2015.10.025
Ferrari S, Iorio Di E, Barbaro V, Ponzin D, Sorrentino FS, Parmeggiani F (2011) Retinitis pigmentosa: genes and disease mechanisms. Curr Genomics 12:238–249. https://doi.org/10.2174/138920211795860107
Friedman DS (2004) Prevalence of age-related macular degeneration in the United States. Arch Ophthalmol 122:564–572. https://doi.org/10.1001/archopht.122.4.564
Hamel C (2006) Retinitis pigmentosa. Orphanet J Rare Dis 1:40. https://doi.org/10.1186/1750-1172-1-40
Hartong DT, Berson EL, Dryja TP (2006) Retinitis pigmentosa. Lancet 368:1795–1809 https://doi.org/10.1016/S0140-6736(06)69740-7
Iadevaia V, Huo Y, Zhang Z, Foster LJ, Proud CG (2012) Roles of the mammalian target of rapamycin, mTOR, in controlling ribosome biogenesis and protein synthesis. Biochem Soc Trans 40:168–172 https://doi.org/10.1042/BST20110682
Janisch KM, Kasanuki JM, Naumann MC, Davis RJ, Lin CS, Semple-Rowland S, Tsang SH (2009) Light-dependent phosphorylation of the gamma subunit of cGMP-phophodiesterase (PDE6gamma) at residue threonine 22 in intact photoreceptor neurons. Biochem Biophys Res Commun 390:1149–1153. https://doi.org/10.1016/j.bbrc.2009.10.106
Kanow MA (2017) Biochemical adaptations of the retina and retinal pigment epithelium support a metabolic ecosystem in the vertebrate eye. Elife. https://doi.org/10.7554/eLife.28899
Koch SF (2015) Halting progressive neurodegeneration in advanced retinitis pigmentosa. J Clin Invest 125:3704–3713. https://doi.org/10.1172/JCI82462
Maguire AM (2008) Safety and efficacy of gene transfer for Leber's congenital amauros is. N Engl J Med 358:224– 2248. https://doi.org/10.1056/NEJMoa0802315
Martinez-Pastor B, Mostoslavsky R (2012) Sirtuins, metabolism, and cancer. Front Pharmacol 3:22. https://doi.org/10.3389/fphar.2012.00022
McLaughlin ME, Ehrhart TL, Berson EL, Dryja TP (1995) Mutation spectrum of the gene encoding the beta subunit of rod phosphodiesterase among patients with autosomal recessive retinitis pigmentosa. Proc Natl Acad Sci USA 92:3249–3253
Molday RS, Moritz OL (2015) Photoreceptors at a glance. J Cell Sci 128:4039–4045. https://doi.org/10.1242/jcs.175687
Punzo C, Kornacker K, Cepko CL (2009) Stimulation of the insulin/mTOR pathway delays cone death in a mouse model of retinitis pigmentosa. Nat Neurosci 12:44–52
Punzo C, Xiong W, Cepko CL (2012) Loss of daylight vision in retinal degeneration: areoxidative stress and metabolic dysregulation to blame? J Biol Chem 287:1642–1648. https://doi.org/10.1074/jbc.R111.304428
Rajala A (2018) Pyruvate kinase M2 regulates photoreceptor structure, function, and viability. Cell Death Dis 9:240. https://doi.org/10.1038/s41419-018-0296-4
Rajala RV, Rajala A, Kooker C, Wang Y, Anderson RE (2016) The warburg effect mediator pyruvate kinase M2 expression and regulation in the retina. Sci Rep 6:37727. https://doi.org/10.1038/srep37727
Russell S (2017) Efficacy and safety of voretigene neparvovec (AAV2-hRPE65v2) in patients with RPE65-mediated inherited retinal dystrophy: a randomised, controlled, open-label, phase 3 trial. Lancet 390:849–860. https://doi.org/10.1016/S0140-6736(17)31868-8
Sancho-Pelluz J (2019) Mechanisms of neurodegeneration in a preclinical autosomal dominant retinitis pigmentosa knock-in model with a Rho(D190N) mutation. Cell Mol Life Sci 76:3657–3665. https://doi.org/10.1007/s00018-019-03090-9
Schmier JK, Jones ML, Halpern MT (2006) The burden of age-related macular degeneration. Pharmacoeconomics 24:319–334
Sohocki MM (2001) Prevalence of mutations causing retinitis pigmentosa and other inherited retinopathies. Hum Mutat 17:42–51. https://doi.org/10.1002/1098-1004(2001)17:1<42
Sun Q (2011) Mammalian target of rapamycin up-regulation of pyruvat kinase isoenzyme type M2 is critical for aerobic glycolysis and tumor growth. Proc Natl Acad Sci USA 108 4129–4134. https://doi.org/10.1073/pnas.1014769108
Takahashi VKL, Takiuti JT, Jauregui R, Tsang SH (2018) Gene therapy in inherited retinal degenerative diseases: a review. Ophthalmic Genet 39:560–568. https://doi.org/10.1080/13816810.2018.1495745
Tosi J, Davis RJ, Wang NK, Naumann M, Lin CS, Tsang SH (2011a) shRNA knockdown of guanylate cyclase 2e or cyclic nucleotide gated channel alpha 1 increases photoreceptor survival in a cGMP phosphodiesterase mouse model of retinitis pigmentosa. J Cell Mol Med 15:1778–1787. https://doi.org/10.1111/j.1582-4934.2010.01201.x
Tosi J et al (2011b) Lentivirus-mediated expression of cDNA and shRNA slows degeneration in retinitis pigmentosa. Exp Biol Med (Maywood). https://doi.org/10.1258/ebm.2011.011053
Tsang SH, Burns ME, Calvert PD, Gouras P, Baylor DA, Goff SP, Arshavsky VY (1998) Role for the target enzyme in deactivation of photoreceptor G protein in vivo. Science 282:117–121. https://doi.org/10.1126/science.282.5386.117
Tsang SH, Gouras P, Yamashita CK, Kjeldbye H, Fisher J, Farber DB, Goff SP (1996) Retinal degeneration in mice lacking the gamma subunit of the rod cGMP phosphodiesterase. Science 272:1026–1029
Tsang SH (2008) A novel mutation and phenotypes in phosphodiesterase 6 deficiency. Am J Ophthalmol 146:780–788. https://doi.org/10.1016/j.ajo.2008.06.017
Usui S (2009) Increased expression of catalase and superoxide dismutase 2 reduces cone cell death in retinitis pigmentosa. Mol Ther 17:778–786. https://doi.org/10.1038/mt.2009.47
Velez G (2017) Gene therapy restores mfrp and corrects axial eye length. Sci Rep 7:16151. https://doi.org/10.1038/s41598-017-16275-8
Venkatesh A, Ma S, Le YZ, Hall MN, Ruegg MA, Punzo C (2015) Activated mTORC1 promotes long-term cone survival in retinitis pigmentosa mice. J Clin Invest 125:1446–1458. https://doi.org/10.1172/JCI79766
Wert KJ, Davis RJ, Sancho-Pelluz J, Nishina PM, Tsang SH (2013) Gene therapy provides long-term visual function in a pre-clinical model of retinitis pigmentosa. Hum Mol Genet 22:558–567. https://doi.org/10.1093/hmg/dds466
Woodruff ML, Janisch KM, Peshenko IV, Dizhoor AM, Tsang SH, Fain GL (2008) Modulation of phosphodiesterase6 turnoff during background illumination in mouse rod photoreceptors. J Neurosci 28:2064–2074. https://doi.org/10.1523/JNEUROSCI.2973-07.2008
Wubben TJ, Pawar M, Smith A, Toolan K, Hager H, Besirli CG (2017) Photoreceptor metabolic reprogramming provides survival advantage in acute stress while causing chronic degeneration. Sci Rep 7:17863. https://doi.org/10.1038/s41598-017-18098-z
Xiong W, MacColl Garfinkel AE, Li Y, Benowitz LI, Cepko CL (2015) NRF2 promotes neuronal survival in neurodegeneration and acute nerve damage. J Clin Invest 125:1433–1445. https://doi.org/10.1172/JCI79735
Yang J (2012) Vigabatrin-induced retinal toxicity is partially mediated by signaling in rod and cone photoreceptors. PLoS ONE 7(8):e43889. https://doi.org/10.1371/journal.pone.0043889
Zadro-Lamoureux LA, Zacks DN, Baker AN, Zheng QD, Hauswirth WW, Tsilfidis C (2009) XIAP effects on retinal detachment-induced photoreceptor apoptosis [corrected]. Invest Ophthalmol Vis Sci 50:1448–1453. https://doi.org/10.1167/iovs.08-2855
Zhang L (2016a) Reprogramming metabolism by targeting sirtuin 6 attenuates retinal degeneration. J Clin Invest 126:4659–4673. https://doi.org/10.1172/JCI86905
Zhang L (2016b) Reprogramming towards anabolism impedes degeneration in a preclinical model of retinitis pigmentosa. Hum Mol Genet 25:4244–4255. https://doi.org/10.1093/hmg/ddw256
Zhang Z, Deng X, Liu Y, Liu Y, Sun L, Chen F (2019) PKM2, function and expression and regulation. Cell Biosci 9:52. https://doi.org/10.1186/s13578-019-0317-8
Acknowledgements
Jonas Children’s Vision Care and Bernard & Shirlee Brown Glaucoma Laboratory are supported by the National Institutes of Health [P30EY019007, R01EY018213, R01EY026682, R24EY027285, R24 EY28758-U01EY030580, National Cancer Institute Core [5P30CA013696], Foundation Fighting Blindness [TA-NMT-0116-0692-COLU], the Research to Prevent Blindness (RPB) Physician-Scientist Award, and unrestricted funds from RPB, New York, NY, USA. S.H.T. is a member of the RD-CURE Consortium and is supported by Kobi and Nancy Karp, the Crowley Family Fund, the Rosenbaum Family Foundation, the Tistou and Charlotte Kerstan Foundation, the Schneeweiss Stem Cell Fund, New York State [C029572], and the Gebroe Family Foundation.
Funding
Funding for this research was supported by the Global Ophthalmology Awards Program (GOAP), a Bayer-sponsored initiative committed to supporting ophthalmic research across the world. The Jonas Children’s Vision Care and Bernard & Shirlee Brown Glaucoma Laboratory are supported by the National Institutes of Health [P30EY019007, R01EY018213, R01EY024698, R01EY026682, R24EY027285, U01EY030580; R24 EY28758], National Cancer Institute Core [5P30CA013696], Foundation Fighting Blindness [TA-NMT-0116-0692-COLU], the Research to Prevent Blindness (RPB) Physician-Scientist Award, and unrestricted funds from RPB, New York, NY, USA. S.H.T. is a member of the RD-CURE Consortium and is supported by Kobi and Nancy Karp, the Crowley Family Fund, the Rosenbaum Family Foundation, the Tistou and Charlotte Kerstan Foundation, the Schneeweiss Stem Cell Fund, New York State [C029572], and the Gebroe Family Foundation.
Author information
Authors and Affiliations
Contributions
EZ and JR analyzed and interpreted the data and drafted the manuscript. SR and JO were major contributors in the critical revision of the manuscript. EZ, CH, XC, NW, and TY performed analysis of the variant effects of Pkm2 ablation on retinal morphology and function and were contributors to figure production. JR conducted the background literature review, provided figure descriptions and contributed to figure production. SHT was involved in the conception of this work and revising the manuscript for content. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest to disclose for any author.
Ethical approval
All experiments were approved by the Columbia University Institutional Animal Care and Use Committee (IACUC) prior to the start of the study. All mice were used and maintained in accordance with the Statement for the Use of Animals in Ophthalmic and Vision Research of the Association for Research in Vision and Ophthalmology and the Policy for the Use of Animals in Neuroscience Research of the Society for Neuroscience.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, E., Ryu, J., Levi, S.R. et al. PKM2 ablation enhanced retinal function and survival in a preclinical model of retinitis pigmentosa. Mamm Genome 31, 77–85 (2020). https://doi.org/10.1007/s00335-020-09837-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00335-020-09837-1