Abstract
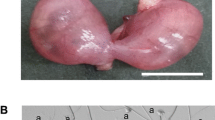
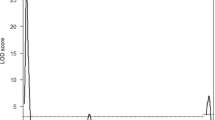
Spontaneous testicular teratomas (STTs) composed by various kinds of tissues are derived from primordial germ cells (PGCs) in the fetal testes of the mouse. In contrast, intra-testicular grafts of the mouse strain (129/Sv-Ter (+/+)) fetal testes possessed the ability to develop the experimental testicular teratomas (ETTs), indistinguishable from the STTs at a morphological level. In this study, linkage analysis was performed for exploration of possible candidate genes involving in ETT development using F2 intercross fetuses derived from [LTXBJ × 129/Sv-Ter (+/+)] F1 hybrids. Linkage analysis with selected simple sequence length polymorphisms along chromosomes 18 and 19, which have been expected to contain ETT-susceptibility loci, demonstrated that a novel recessive candidate gene responsible for ETT development is located in 1.1 Mb region between the SSLP markers D18Mit81 and D18Mit184 on chromosome 18 in the 129/Sv-Ter (+/+) genetic background. Since this locus is different from the previously known loci (including Ter, pgct1, and Tgct1) for STT development, we named this novel gene “experimental testicular teratoma 1 (ett1)”. To resolve the location of ett1 independently from other susceptibility loci, ett1 loci was introduced in a congenic strain in which the distal segment of chromosome 18 in LTXBJ strain mice had been replaced by a 1.99 Mbp genomic segment of the 129/Sv-Ter (+/+) mice. Congenic males homozygous for the ett1 loci were confirmed to have the ability to form ETTs, indicating that this locus contain the gene responsible for ETTs. We listed candidate genes included in this region, and discussed about their possible involvement in induction of ETTs.



Similar content being viewed by others
References
Anderson PD, Nelson VR, Tesar PJ, Nadeau JH (2009) Genetic factors on mouse chromosome 18 affecting susceptibility to testicular germ cell tumors and permissiveness to embryonic stem cell derivation. Cancer Res 69:9112–9117
Asada Y, Varnum DS, Frankel WN, Nadeau JH (1994) A mutation in the Ter gene causing increased susceptibility to testicular teratomas maps to mouse chromosome 18. Nat Genet 6:363–368
Bertagna X (1994) Proopiomelanocortin-derived peptides. Endocrinol Metab Clin N Am 23:467–485
Bhattacharya R, Cabral F (2004) A ubiquitous beta-tubulin disrupts microtubule assembly and inhibits cell proliferation. Mol Biol Cell 15:3123–3131
Blin N, Stafford DW (1976) A general method for isolation of high molecular weight DNA from eukaryotes. Nucleic Acids Res 3:2303–2308
Bultman SJ, Michaud EJ, Woychik RP (1992) Molecular characterization of the mouse agouti locus. Cell 71:1195–1204
Cleveland DW (1987) The multitubulin hypothesis revisited: what have we learned? J Cell Biol 104:381–383
Corradi JP, Ravyn V, Robbins AK, Hagan KW, Peters MF, Bostwick R, Buono RJ, Berrettini WH, Furlong ST (2005) Alternative transcripts and evidence of imprinting of GNAL on 18p11.2. Mol Psychiatry 10:1017–1025
Cryns K, Shamir A, Shapiro J, Daneels G, Goris I, Van Craenendonck H, Straetemans R, Belmaker RH, Agam G, Moechars D, Steckler T (2007) Lack of lithium-like behavioral and molecular effects in IMPA2 knockout mice. Neuropsychopharmacology 32:881–891
Dinulescu DM, Cone RD (2000) Agouti and agouti-related protein: analogies and contrasts. J Biol Chem 275:6695–6698
Fujita M, Maeda Y, Ra M, Yamaguchi Y, Taguchi R, Kinoshita T (2009) GPI glycan remodeling by PGAP5 regulates transport of GPI-anchored proteins from the ER to the Golgi. Cell 139:352–365
Gantz I, Konda Y, Tashiro T, Shimoto Y, Miwa H, Munzert G, Watson SJ, DelValle J, Yamada T (1993a) Molecular cloning of a novel melanocortin receptor. J Biol Chem 268:8246–8250
Gantz I, Miwa H, Konda Y, Shimoto Y, Tashiro T, Watson SJ, DelValle J, Yamada T (1993b) Molecular cloning, expression, and gene localization of a fourth melanocortin receptor. J Biol Chem 268:15174–15179
Gantz I, Shimoto Y, Konda Y, Miwa H, Dickinson CJ, Yamada T (1994) Molecular cloning, expression, and characterization of a fifth melanocortin receptor. Biochem Biophys Res Commun 200:1214–1220
Hanaoka K, Hayasaka M, Noguchi T, Kato Y (1991) The stem cells of a primordial germ cell-derived teratocarcinoma have the ability to form viable mouse chimeras. Differentiation 48:83–87
Heaney JD, Michelson MV, Youngren KK, Lam MY, Nadeau JH (2009) Deletion of eIF2beta suppresses testicular cancer incidence and causes recessive lethality in agouti-yellow mice. Hum Mol Genet 18:1395–1404
Hiser L, Aggarwal A, Young R, Frankfurter A, Spano A, Correia JJ, Lobert S (2006) Comparison of beta-tubulin mRNA and protein levels in 12 human cancer cell lines. Cell Motil Cytoskelet 63:41–52
Hoek KS (2007) DNA microarray analyses of melanoma gene expression: a decade in the mines. Pigment Cell Res 20:466–484
Huang YW, Luo J, Weng YI, Mutch DG, Goodfellow PJ, Miller DS, Huang TH (2010) Promoter hypermethylation of CIDEA, HAAO and RXFP3 associated with microsatellite instability in endometrial carcinomas. Gynecol Oncol 117:239–247
Huszar D, Lynch CA, Fairchild-Huntress V, Dunmore JH, Fang Q, Berkemeier LR, Gu W, Kesterson RA, Boston BA, Cone RD, Smith FJ, Campfield LA, Burn P, Lee F (1997) Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88:131–141
Inohara N, Koseki T, Chen S, Wu X, Nunez G (1998) CIDE, a novel family of cell death activators with homology to the 45 kDa subunit of the DNA fragmentation factor. EMBO J 17:2526–2533
Ishida M, Sunamura M, Furukawa T, Lefter LP, Morita R, Akada M, Egawa S, Unno M, Horii A (2008) The PMAIP1 gene on chromosome 18 is a candidate tumor suppressor gene in human pancreatic cancer. Dig Dis Sci 53:2576–2582
Kierszenbaum AL, Tres LL (2001) Primordial germ cell-somatic cell partnership: a balancing cell signaling act. Mol Reprod Dev 60:277–280
Kublaoui BM, Holder JL Jr, Tolson KP, Gemelli T, Zinn AR (2006) SIM1 overexpression partially rescues agouti yellow and diet-induced obesity by normalizing food intake. Endocrinology 147:4542–4549
Lam MY, Heaney JD, Youngren KK, Kawasoe JH, Nadeau JH (2007) Trans-generational epistasis between Dnd1Ter and other modifier genes controls susceptibility to testicular germ cell tumors. Hum Mol Genet 16:2233–2240
Leandro-Garcia LJ, Leskela S, Landa I, Montero-Conde C, Lopez-Jimenez E, Leton R, Cascon A, Robledo M, Rodriguez-Antona C (2010) Tumoral and tissue-specific expression of the major human beta-tubulin isotypes. Cytoskeleton (Hoboken) 67:214–223
Matin A, Collin GB, Asada Y, Varnum D, Nadeau JH (1999) Susceptibility to testicular germ-cell tumours in a 129.MOLF-Chr 19 chromosome substitution strain. Nat Genet 23:237–240
Miller MW, Duhl DM, Vrieling H, Cordes SP, Ollmann MM, Winkes BM, Barsh GS (1993) Cloning of the mouse agouti gene predicts a secreted protein ubiquitously expressed in mice carrying the lethal yellow mutation. Genes Dev 7:454–467
Mountjoy KG, Robbins LS, Mortrud MT, Cone RD (1992) The cloning of a family of genes that encode the melanocortin receptors. Science 257:1248–1251
Nicholson JR, Kohler G, Schaerer F, Senn C, Weyermann P, Hofbauer KG (2006) Peripheral administration of a melanocortin 4-receptor inverse agonist prevents loss of lean body mass in tumor-bearing mice. J Pharmacol Exp Ther 317:771–777
Noguchi T, Noguchi M (1985) A recessive mutation (ter) causing germ cell deficiency and a high incidence of congenital testicular teratomas in 129/Sv-ter mice. J Natl Cancer Inst 75:385–392
Noguchi M, Watanabe C, Kobayashi T, Kuwashima M, Sakurai T, Katoh H, Moriwaki K (1996) The ter mutation responsible for germ cell deficiency but not testicular nor ovarian teratocarcinogenesis in ter/ter congenic mice. Dev Growth Differ 38:59–69
Pearson K (1900) On the criterion that a given system of deviations from the probable in the case of a correlated system of variables is such that it can be reasonably supposed to have arisen from random sampling. Lond, Edinb, Dublin Philos Mag J Sci 50:157–175
Regenass U, Friedrich TD, Stevens LC (1982) Experimental induction of testicular teratomas in dissociated-reaggregated chimaeric gonads. J Embryol Exp Morphol 72:153–167
Sakurai T, Katoh H, Moriwaki K, Noguchi T, Noguchi M (1994) The ter primordial germ cell deficiency mutation maps near Grl-1 on mouse chromosome 18. Mamm Genome 5:333–336
Shibue T, Takeda K, Oda E, Tanaka H, Murasawa H, Takaoka A, Morishita Y, Akira S, Taniguchi T, Tanaka N (2003) Integral role of Noxa in p53-mediated apoptotic response. Genes Dev 17:2233–2238
Sjoholt G, Gulbrandsen AK, Lovlie R, Berle JO, Molven A, Steen VM (2000) A human myo-inositol monophosphatase gene (IMPA2) localized in a putative susceptibility region for bipolar disorder on chromosome 18p11.2: genomic structure and polymorphism screening in manic-depressive patients. Mol Psychiatry 5:172–180
Skinner MK (1991) Cell-cell interactions in the testis. Endocr Rev 12:45–77
Smith AI, Funder JW (1988) Proopiomelanocortin processing in the pituitary, central nervous system, and peripheral tissues. Endocr Rev 9:159–179
Stevens LC (1964) Experimental production of testicular teratomas in mice. Proc Natl Acad Sci USA 52:654–661
Stevens LC (1967a) The biology of teratomas. Adv Morphog 6:1–31
Stevens LC (1967b) Origin of testicular teratomas from primordial germ cells in mice. J Natl Cancer Inst 38:549–552
Stevens LC (1973) A new inbred subline of mice (129-terSv) with a high incidence of spontaneous congenital testicular teratomas. J Natl Cancer Inst 50:235–242
Stevens LC (1975) Teratocarcinogenesis and spontaneous parthenogenesis in mice. Symp Soc Dev Biol 93–106
Stevens LC, Hummel KP (1957) A description of spontaneous congenital testicular teratomas in strain 129 mice. J Natl Cancer Inst 18:719–747
Stevens LC, Little CC (1954) Spontaneous testicular teratomas in an inbred strain of mice. Proc Natl Acad Sci USA 40:1080–1087
Stevens LC, Varnum DS (1974) The development of teratomas from parthenogenetically activated ovarian mouse eggs. Dev Biol 37:369–380
Stevens LC, Varnum DS, Eicher EM (1977) Viable chimaeras produced from normal and parthenogenetic mouse embryos. Nature 269:515–517
Strathmann M, Wilkie TM, Simon MI (1989) Diversity of the G-protein family: sequences from five additional alpha subunits in the mouse. Proc Natl Acad Sci USA 86:7407–7409
Takabayashi S, Sasaoka Y, Yamashita M, Tokumoto T, Ishikawa K, Noguchi M (2001) Novel growth factor supporting survival of murine primordial germ cells: evidence from conditioned medium of ter fetal gonadal somatic cells. Mol Reprod Dev 60:384–396
Tanaka N, Kyuuma M, Sugamura K (2008) Endosomal sorting complex required for transport proteins in cancer pathogenesis, vesicular transport, and non-endosomal functions. Cancer Sci 99:1293–1303
Tao YX (2010) The melanocortin-4 receptor: physiology, pharmacology, and pathophysiology. Endocr Rev 31:506–543. doi:10.1210/er.2009-0037
Valente LJ, Gray DH, Michalak EM, Pinon-Hofbauer J, Egle A, Scott CL, Janic A, Strasser A (2013) p53 efficiently suppresses tumor development in the complete absence of its cell-cycle inhibitory and proapoptotic effectors p21, Puma, and Noxa. Cell Rep 3:1339–1345
Weidinger G, Stebler J, Slanchev K, Dumstrei K, Wise C, Lovell-Badge R, Thisse C, Thisse B, Raz E (2003) dead end, a novel vertebrate germ plasm component, is required for zebrafish primordial germ cell migration and survival. Curr Biol 13:1429–1434
Youngren KK, Nadeau JH, Matin A (2003) Testicular cancer susceptibility in the 129.MOLF-Chr19 mouse strain: additive effects, gene interactions and epigenetic modifications. Hum Mol Genet 12:389–398
Youngren KK, Coveney D, Peng XN, Bhattacharya C, Schmidt LS, Nickerson ML, Lamb BT, Deng JM, Behringer RR, Capel B, Rubin EM, Nadeau JH, Matin A (2005) The Ter mutation in the dead end gene causes germ cell loss and testicular germ cell tumours. Nature 435:360–364. doi:10.1038/nature03595
Zhou Z, Yon Toh S, Chen Z, Guo K, Ng CP, Ponniah S, Lin SC, Hong W, Li P (2003) Cidea-deficient mice have lean phenotype and are resistant to obesity. Nat Genet 35:49–56
Acknowledgments
This work was supported by JSPS KAKENHI Grant Number 12680809, 14380381 and 16650092 (to MN). This work also supported by Grants-in-Aid for Scientific Research in Priority Areas from the Ministry of Education, Culture, Sports, Science and Technology of Japan for preservation of special lines of mice to Shizuoka University.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Miyazaki, T., Ikeda, Y., Kubo, I. et al. Identification of genomic locus responsible for experimentally induced testicular teratoma 1 (ett1) on mouse Chr 18. Mamm Genome 25, 317–326 (2014). https://doi.org/10.1007/s00335-014-9529-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00335-014-9529-8