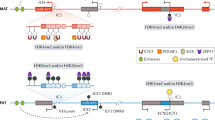
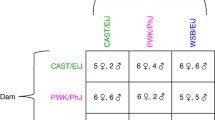
Abstract
Genomic imprinting, where the effects of alleles depend on their parent-of-origin, can be an important component of the genetic architecture of complex traits. Although there has been a rapidly increasing number of studies of genetic architecture that have examined imprinting effects, none have examined whether imprinting effects depend on genetic background. Such effects are critical for the evolution of genomic imprinting because they allow the imprinting state of a locus to evolve as a function of genetic background. Here we develop a two-locus model of epistasis that includes epistatic interactions involving imprinting effects and apply this model to scan the mouse genome for loci that modulate the imprinting effects of quantitative trait loci (QTL). The inclusion of imprinting leads to nine orthogonal forms of epistasis, five of which do not appear in the usual two-locus decomposition of epistasis. Each form represents a change in the imprinting status of one locus across different classes of genotypes at the other locus. Our genome scan identified two different locus pairs that show complex patterns of epistasis, where the imprinting effect at one locus changes across genetic backgrounds at the other locus. Thus, our model provides a framework for the detection of genetic background-dependent imprinting effects that should provide insights into the background dependence and evolution of genomic imprinting. Our application of the model to a genome scan supports this assertion by identifying pairs of loci that show reciprocal changes in their imprinting status as the background provided by the other locus changes.




Similar content being viewed by others
References
Alleman M, Doctor J (2000) Genomic imprinting in plants: observations and evolutionary implications. Plant Mol Biol 43:147–161
Álvarez-Castro JM, Carlborg O (2007) A unified model for functional and statistical epistasis and its application to quantitative trait loci analysis. Genetics 176:1151–1167
Arnaud P, Feil R (2006) MEDEA takes control of its own imprinting. Cell 124:468–470
Chai CK (1956) Analysis of quantitative inheritance of body size in mice. I. Hybridization and maternal influence. Genetics 41:167–178
Chaudhuri S, Messing J (1994) Allele-specific parental imprinting of dzr1, a posttranscriptional regulator of zein accumulation. Proc Natl Acad Sci USA 91:4867–4871
Cheverud JM (2000) Detecting epistasis among quantitative trait loci. In: Wolf JB, Brodie EDIII, Wade MJ (eds) Epistasis and the evolutionary process. Oxford University Press, New York, pp 58–81
Cheverud JM, Routman EJ, Duarte FAM, Swinderen BV, Cothran K et al (1996) Quantitative trait loci for murine growth. Genetics 142:1305–1319
Cheverud JM, Hager R, Roseman C, Fawcett G, Wang B et al (2008) Genomic imprinting effects on adult body composition in mice. Proc Natl Acad Sci USA 105:4253–4258
Choi Y, Gehring M, Johnson L, Hannon M, Harada JJ et al (2002) DEMETER, a DNA glycosylase domain protein, is required for endosperm gene imprinting and seed viability in Arabidopsis. Cell 110:33–42
Cockett NE, Jackson SP, Shay TL, Farnir F, Berghmans S, Snowder GD (1996) Polar overdominance at the ovine callipyge locus. Science 273:236–238
Davis E, Caiment F, Tordoir X, Cavaillé J, Ferguson-Smith A, Cockett N, Georges M, Charlier C (2005) RNAi-mediated allelic trans-interaction at the imprinted Rtl1/Peg11 locus. Curr Biol 15:743–749
de Koning D-J, Rattink AP, Harlizius B, van Arendonk JAM, Brascamp EW, Groenen MAM (2000) Genome-wide scan for body composition in pigs reveals important role of imprinting. Proc Natl Acad Sci USA 97:7947–7950
de Koning D-J, Bovenhuis H, Van Arendonk JAM (2002) On the detection of imprinted quantitative trait loci in experimental crosses of outbred species. Genetics 161:931–938
Falconer DS, Mackay TFC (1996) Introduction to quantitative genetics, 4th edn. Longman, Essex
Freking BA, Murphy SK, Wylie AA, Rhodes SJ, Keele JW, Leymaster KA, Jirtle RL, Smith TPL (2002) Identification of the single base change causing the callipyge muscle hypertrophy phenotype, the only known example of polar overdominance in mammals. Genome Res 12:1496–1506
Goodale HD (1938) A study of the inheritance of body weight in the albino mouse by selection. J Hered 29:101–112
Grossniklaus U (2005) Genomic imprinting in plants: a predominantly maternal affair. In: Meyer P (ed) Annual plant reviews: plant epigenetics. Blackwell, Sheffield, pp 174–200
Hager R, Cheverud JM, Wolf JB (2008) Maternal effects as the cause of parent-of-origin effects that mimic genomic imprinting. Genetics 178:1755–1762
Hager R, Cheverud JM, Wolf JB (2009) Relative contribution of additive, dominance and imprinting effects to phenotypic variation in body size and growth between divergent selection lines of mice. Evolution Int J org Evolution 63:1118–1128
Hrbek T, Alves de Brito R, Wang B, Pletscher LS, Cheverud JM (2006) Genetic characterization of a new set of recombinant inbred lines (LGXSM) formed from the intercross of SM/J and LG/J inbred mouse strains. Mamm Genome 17:417–429
Kao C-H, Zeng Z-B (2002) Modeling epistasis of quantitative trait loci using Cockerham’s model. Genetics 160:1243–1261
Kermicle JL (1978) Imprinting of gene action in maize endosperm. In: Walden DB (ed) Maize breeding and genetics. Wiley, New York, pp 357–371
Knott SA, Marklund L, Haley CS, Andersson K, Davies W, Ellegren H, Fredholm M, Hansson I, Hoyheim B, Lundstrom K, Moller M, Andersson L (1998) Multiple marker mapping of quantitative trait loci in a cross between outbred wild boar and large white pigs. Genetics 149:1069–1080
Kramer MG, Vaughn TT, Pletscher LS, King-Ellison K, Adams E, Cheverud JM (1998) Genetic variation for body weight gain and composition in the intercross of Large (LG/J) and Small (SM/J) inbred strains of mice. Genet Mol Biol 21:211–218
Lander ES, Botstein D (1989) Mapping Mendelian factors underlying quantitative traits using RFLP linkage maps. Genetics 121:185–189
Leamy LJ, Klingenberg CP, Sherratt E, Wolf JB, Cheverud JM (2008) A search for quantitative trait loci exhibiting imprinting effects on mouse mandible size and shape. Heredity 101:518–526
Li J, Ji L (2005) Adjusting multiple testing in multilocus analysis using the eigenvalues of a correlaton matrix. Heredity 95:221–227
Li J, Jiang T (2003a) Efficient inference of haplotypes from genotypes on a pedigree. J Bioinform Comput Biol 1:41–69
Li J, Jiang T (2003b) Efficient rule-based haplotyping algorithms for pedigree data. In: Proceedings of the 7th annual international conference on research computational molecular biology, pp 197–206
Luedi PP, Hartemink AJ, Jirtle RL (2005) Genome-wide prediction of imprinted murine genes. Genome Res 15:875–884
Luedi PP, Dietrich FS, Weidman JR, Bosko JM, Jirtle RL, Hartemink AJ (2007) Computational and experimental identification of novel human imprinted genes. Genome Res 17:1723–1730
Lynch M, Walsh B (1998) Genetics and analysis of quantitative traits. Sinauer Associates, Sunderland
MacArthur J (1944) Genetics of body size and related characters. I. Selection of small and large races of the laboratory mouse. Am Naturalist 78:142–157
Mantey C, Brockmann GA, Kalm E, Reinsch N (2005) Mapping and exclusion mapping of genomic imprinting effects in mouse F2 families. J Hered 96:329–338
Morison IM, Ramsay JP, Spencer HG (2005) A census of mammalian imprinting. Trends Genet 21:457–465
Nezer C, Moreau L, Brouwers B, Coppieters W, Detilleaux J, Hanset R, Karim L, Kvasz A, Leroy P, Georges M (1999) An imprinted QTL with major effect on muscle mass and fat deposition maps to the IGF2 locus in pigs. Nat Genet 21:155–156
Reik W, Walter J (2001) Genomic imprinting: parental influence on the genome. Nat Rev Genet 2:21–32
Reilly KM, Tuskan RG, Christy E, Loisel DA, Ledger J, Bronson RT, Smith CD, Tsang S, Munroe DJ, Jacks T (2004) Susceptibility to astrocytoma in mice mutant for Nf1 and Trp53 is linked to chromosome 11 and subject to epigenetic effects. Proc Natl Acad Sci USA 35:13008–13013
Reilly KM, Broman KW, Bronson RT, Tsang S, Loisel DA, Christy E, Sun J, Diehl J, Munroe DJ, Tuskan RG (2006) An imprinted locus epistatically influences Nstr1 and Nstr2 to control resistance to nerve sheath tumors in a neuorofibromatosis type 1 mouse model. Cancer Res 66:62–68
Spencer HG, Williams JM (1997) The evolution of genomic imprinting: two modifier-locus models. Theor Popul Biol 51:23–35
Templeton AR (2000) Epistasis and complex traits. In: Wolf JB, Brodie EDIII, Wade MJ (eds) Epistasis and the evolutionary process. Oxford University Press, New York, pp 41–57
Van Cleve J, Feldman MW (2007) Sex-specific viability, sex linkage and dominance in genomic imprinting. Genetics 176:1101–1118
Van Laere A-S, Nguyen M, Braunschweig M, Nezer C, Collette C, Moreau L, Archibald AL, Haley CS, Buys N, Tally M, Andersson G, Georges M, Andersson L (2003) A regulatory mutation in IGF2 causes a major QTL effect on muscle growth in the pig. Nature 425:832–836
Vaughn TT, Pletscher LS, Peripato A, King-Ellison K, Adams E, Erikson C, Cheverud JM (1999) Mapping quantitative trait loci for murine growth—a closer look at genetic architecture. Genet Res 74:313–322
Vongs A, Kakutani T, Martienssen RA, Richards EJ (1993) Arabidopsis thaliana DNA methylation deficient mutants. Science 260:1926–1928
Walrath JC, Fox K, Truffer E, Alvord WG, Quiñones OA, Reilly KM (2009) Chr 19A/J modifies tumor resistance in a sex- and parent-or-origin-specific manner. Mamm Genome 20:214–223
Wilkins JF, Haig D (2002) Parental modifiers, antisense transcripts and loss of imprinting. Proc R Soc B Biol Sci 269:1841–1846
Wolf JB (2001) Gene interactions from maternal effects. Evolution Int J org Evolution 54:1882–1898
Wolf JB, Brodie EDIII, Wade MJ (2000) Epistasis and the evolutionary process. Oxford University Press, Oxford
Wolf JB, Hager R, Cheverud JM (2008a) Genomic imprinting effects on complex traits: a phenotype-based perspective. Epigenetics 3:295–299
Wolf JB, Cheverud JM, Roseman C, Hager R (2008b) Genome-wide analysis reveals a complex pattern of genomic imprinting in mice. PLoS Genet 4:e1000091
Xiao W, Gehring M, Choi Y, Margossian L, Pu H, Harada JJ, Goldberg RB, Pennell RI, Fischer RL (2003) Imprinting of the MEA polycomb gene is controlled by antagonism between MET1 methyltransferase and DME glycosylase. Dev Cell 5:891–901
Zeng Z-B, Wang T, Zou W (2005) Modeling quantitative trait loci and interpretation of models. Genetics 169:1711–1725
Acknowledgments
We thank Larry Leamy and Charles Roseman for insightful discussions during the development of this work and Reinmar Hager and Will Pitchers for help with the haplotype reconstruction. This work was supported by a grant from the Biotechnology and Biological Sciences Research Council (UK) to JBW and by NIH grant DK055736 to JMC.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendices
Appendix 1: model details
The order of the vector of two-locus genotypic values (G AB) is constructed using the Kronecker product of the two single-locus genotypic value vectors, where G AB = G B ⊗ G A (cf. Appendix 1 in Álvarez-Castro and Carlborg 2007). The Kronecker product gives the order of the genotypic values, not the actual products (i.e., multiplication) of values. Since each single-locus genotypic value has two subscripts corresponding to the maternally and paternally inherited alleles at the locus, the two-locus genotypic values simply contain the combination of subscripts taken from the two single-locus genotypic values, with the A locus subscripts appearing first. Thus, in the resulting vector of two-locus genotypic values, G ijkl, the first two subscripts refer to the two A locus alleles (i = paternally inherited allele, j = maternally inherited allele) and the last two refer to the B locus alleles (k = maternally inherited allele, l = paternally inherited allele). This can be visualized as
Like the matrix of two-locus genotypic values, the structure of the vector of two-locus genetic effects (E AB) is defined by the Kronecker product of the two single-locus vectors (E A and E B), E AB = E B ⊗ E A, but with the reference point in each vector first replaced with a 1 (Álvarez-Castro and Carlborg 2007). The products of single-locus effects in the resulting vector are replaced with corresponding epistasis terms (e.g., a A a B would be replaced with aa, which corresponds to the interaction between additive effects of the two loci, see below), resulting in a vector that contains a total of 16 terms, 9 of which correspond to epistasis between the two loci:
Appendix 2: extension of the model to arbitrary allele frequencies
In this appendix we extend the model to populations with arbitrary allele frequencies that are in Hardy–Weinberg equilibrium. This approach follows the general two-allele model presented in Zeng et al. (2005), which has been called the G2A model by Álvarez-Castro and Carlborg (2007). It can be extended to any population using the framework in Álvarez-Castro and Carlborg (2007). We do not present a full description of the G2A model since a full description and justification is presented by Zeng et al. (2005). The G2A model differs from the model presented in the text above in that the values of the genotypic index variables (coefficients) depend on allele frequencies at the loci (to maintain orthogonality across allele frequencies). When the two alleles are at equal frequency, the G2A model reduces to the Cockerham model presented in the text.
Because the genotypic index variables depend on allele frequency, we denote the frequency of the A 1 allele as p 1 and the frequency of the A 2 allele as p 2. Under the G2A model the additive and dominance effect design variables, w A and v A, respectively are defined as (extended to consider the two heterozygotes separately)
The inclusion of the imprinting genotypic index variable in the G2A model is straightforward because the two heterozygotes are always at equal frequency at Hardy-Weinberg equilibrium. As a result, the value of the imprinting genotypic index variable u A does not depend on allele frequencies and therefore is orthogonal from the additive and the dominance genotypic index variables at all frequencies. The value of the genotypic index variable for the G2A model matches that given in Eq. 3. This makes the genetic-effect design matrix for the G2A model:
and the corresponding vector of genetic effects:
The reference point for the model remains the mean of the genotypic values but is now weighted by their frequency. In the G2A model, the measure of the additive effect a A is frequency-dependent and now represents the average effect of an allele substitution rather than the typical frequency-independent additive effect or additive genotypic value. The dominance and imprinting effects, however, are independent of allele frequencies for the single-locus model (but not for multiple loci Zeng et al. 2005).
The two-locus extension of the G2A model for ordered genotypes follows the extension of the single-locus Cockerham model to the two-locus case. The structure of the two-locus vectors of genotypic values (G AB) and genetic effects (E AB) match those in Appendix 1. To extend the model to two loci, one-first needs to define the allele frequencies at the B locus; therefore, we denote the frequency of the B 1 allele as q 1 and the frequency of the B 2 allele as q 2 = 1 − q 1. Using these allele frequencies, a genetic-effect design matrix for locus B can be constructed following the structure in Eq. 15:
As in the Cockerham model presented in the text, the two single-locus genetic-effect design matrices (Eqs. 15 and 17) can be used to derive the genotypic index values for the two-locus model as S AB = S B ⊗ S A. The resulting matrix, which is given in Supplementary Table 2, is analogous to that presented in Table 1 of Zeng et al. (2005), but with the additional dimensions added for the ordered genotypes. As in the model of unordered genotypes from Zeng et al. (2005), the values of all coefficients involving additive and dominance effects (including all epistatic terms involving interactions between these effects) depend on allele frequencies. However, coefficients for the imprinting effects of both loci (i A and i B) and their interaction (ii) do not depend on allele frequencies. The inverse of the two-locus genetic-effect design matrix defines the two-locus genetic effects in terms of two-locus genotypic values. The inverse of the two-locus genetic-effect design matrix, \( {\mathbf{S}}_{AB}^{ - 1} \), which gives the definitions of the two-locus genetic effects in terms of the genotypic values, is given in Supplementary Table 3.
Rights and permissions
About this article
Cite this article
Wolf, J.B., Cheverud, J.M. A framework for detecting and characterizing genetic background-dependent imprinting effects. Mamm Genome 20, 681–698 (2009). https://doi.org/10.1007/s00335-009-9209-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00335-009-9209-2