Abstract
Objectives
Invasively measured fractional flow reserve (FFR) is associated with outcome in heart transplant (HTx) patients. Coronary computed tomography angiography (CCTA)–derived FFR (FFRct) provides additional functional information from anatomical CT images. We describe the first use of FFRct in HTx patients.
Methods
HTx patients underwent CCTA with FFRct to screen for cardiac allograft vasculopathy. FFRct was measured distal to each coronary stenosis > 30% and FFRct ≤ 0.8 indicated hemodynamically significant stenosis. FFRct was also measured at the most distal location of each vessel. Overall distal FFRct was calculated as the mean of the distal values in the left, right, and circumflex coronary artery in each patient.
Results
Seventy-three patients (age 56 (42–65) years, 63% males) at 11 (8–16) years after HTx were included. Eighteen (25%) patients had a focal hemodynamically significant stenosis (stenosis > 30% with FFRct ≤ 0.8). In the 55 patients without a hemodynamically significant focal FFRct stenosis (FFRct > 0.80), the distal left anterior descending artery FFRct was < 0.90 in 74% of the patients and 10 (18%) patients had ≥ 1 coronary artery with a distal FFRct ≤ 0.8, including 1 with a distal FFRct ≤ 0.8 in all coronaries. Overall distal FFRct in patients without focal stenosis was 0.88 (0.86–0.91), 0.87 (0.86–0.90), and 0.88 (0.86–0.91) (median with 25th–75th percentile) at 5–9, 10–14, or ≥ 15 years post-transplantation, respectively (p = 0.93).
Conclusions
FFRct performed on CCTA scans of HTx patients demonstrated that 25% of patients had a focal coronary stenosis with FFRct ≤ 0.8. Even without a focal stenosis, FFRct values are often abnormal in HTx patients.
Key Points
• This is the first report describing the use of FFRct in in heart transplant patients.
• FFRct identifies patients after heart transplantation with hemodynamically significant coronary stenosis.
• Even without a focal stenosis, FFRct values are often abnormal in heart transplant patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cardiac allograft vasculopathy (CAV) is an accelerated fibroproliferative disease that affects the coronary arteries in heart transplant (HTx) patients leading to coronary stenoses [1, 2]. Data from the International Society for Heart and Lung Transplantation (ISHLT) show that almost 50% of patients have CAV at 10 years post-transplant [3, 4]. CAV is the third to fourth leading cause of death amongst HTx patients and accounts for 1 in 8 deaths in those that survive the first year after HTx [3]. Medical treatment can slow CAV progression. But ultimately, CAV progresses and usually revascularization, and in select cases, even retransplantation is needed [1]. Patients with CAV seldom present with classical symptoms of angina because the transplanted heart is denervated [1]. The ISHLT currently recommends annual or biannual invasive coronary angiography (ICA) to evaluate for the development of CAV [5]. Beyond anatomical evaluation invasive fractional flow reserve (FFR) measurements have been shown to provide complementary information and has been shown to be an independent predictor of death and retransplantation [6].
Coronary computed tomography angiography (CCTA) is a reliable alternative to ICA for CAV detection [7]. Technology to calculate FFR values based on CCTA images (FFRct) has become commercially available and is validated in multiple studies in chest pain patients [8,9,10]. The use of FFRct in the follow-up of HTx patients has not been reported yet. The aim of this study is to describe the initial results of CCTA with FFRct analysis in a cohort of HTx patients.
Materials and methods
Patient selection
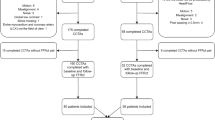
All HTx patients from one hospital that participated in the Assessing Diagnostic Value of Non-invasive FFRct in Coronary Care (ADVANCE EXTEND) registry were included [8]. The institutional ethics committee approved the study. All patients provided informed consent. Patients with a stent in the left main, a stent in two or more major coronary arteries or a metallic stent in the left coronary system could not be enrolled in the ADVANCE EXTEND registry as these scans cannot be processed for FFRct analysis. For each patient, we recorded the following: patient demographics, angina symptom status, current medication use, and clinical outcomes at 1 year including any additional test that was performed for coronary ischemia testing, coronary revascularization, and the occurrence of major adverse cardiac events (MACE) defined as myocardial infarction, all-cause mortality, or unplanned hospitalization for acute coronary syndrome leading to revascularization. The CAV score prior to the FFRct analysis was determined by combining all available information including findings at ICA and stress scintigraphy closest to the CCTA with FFRct.
In our hospital, patients undergo an annual CCTA starting the 5th year post-transplant for CAV surveillance. Invasive coronary angiography is routinely performed one and four years post-transplant and thereafter only when clinically indicated (e.g., stenosis detected on CCTA).
CCTA acquisition
A contrast-enhanced CCTA examination was performed according to the normal clinical routine on a dual-source CT scanner (Force or Drive, Siemens Healthineers) using a prospectively ECG-triggered acquisition mode. Automated tube voltage and tube current selection were used. Contrast injection was generally done using a test-bolus injection with 10–15 ml of contrast media (iopromide 370 mg/ml, Bayer) followed by a 20-ml saline chaser. The CCTA was then performed using 50–70 ml of contrast material followed by a 25-ml saline chaser. Flow rate was 5.4 ml/s. Beta-blockers to lower the heart rate at the time of scanning were administered in conjunction with the treating cardiologist. Sublingual nitroglycerine was administered just prior to scanning in each patient.
FFRct analysis
All CCTA scans were sent to Heartflow for FFRct analysis. In brief, the analysis is based on defining the coronary artery boundaries, subsequently extracting a 3D model of the coronary arteries which is used to perform computational fluid dynamics calculations. Ultimately, a 3D coronary model is generated that can be interrogated at each point in a coronary artery to provide the FFR value at that specific location. In case a modeled stenosis, > 30% is present in a coronary artery; the FFR value distal to the stenosis is automatically supplied on the FFRct report and recorded [11]. A FFRct value of ≤ 0.8 was considered positive and to constitute a hemodynamically significant stenosis (Fig. 1a). Also, in each patient, the FFRct value at the most distal point in each of the three major coronary arteries (right coronary artery (RCA), left anterior descending (LAD), and left circumflex (LCx)) was recorded and the overall FFRct was calculated in each patient as the mean of the three FFRct values (Fig. 1b). A distal FFRct value of ≤ 0.8 without focal coronary stenosis was not considered hemodynamically significant stenosis. In the case of a total occlusion, a FFRct value of 0.5 was used for analysis similar to a previous study using FFRct [11]. The volume-to-mass ratio (V/M, volume of the coronary arteries divided by the left ventricular myocardial mass) was derived from the FFRct segmentation [12].
a CT-derived fractional flow reserve (FFRct) coronary tree with a focal stenosis in the right coronary artery (RCA) and FFRct value of 0.64; b FFRct coronary tree in a patient without a significant stenosis with distal FFR measurements in the tree major coronary arteries: RCA 0.93, left anterior descending (LAD) 0.87, and left circumflex (LCx) 0.95; with a mean distal FFRct of 0.92
Statistical analysis
Data are presented as absolute numbers, means with standard deviation or median with 25th–75th percentile where appropriate. Patient subgroups were compared using Mann–Whitney-U, Kruskal–Wallis, chi-square, or Fisher’s exact tests depending on the type of data. For analysis of time after HTx, patients were divided into three groups: up to 10 years, 10 to 15 years, and 15 or more years after HTx. A p-value of < 0.05 was considered significant.
Results
Patients
Seventy-three HTx patients (46 males (63%), age at time of CCTA 56 (42–65) years, age at HTx 43 (26–54) years) who were 11 (8–16) years after HTx were included (Table 1). The CAV score prior to CCTA was CAV 0 in 60 (82%) patients, CAV 1 in 3 (4%) patients, and CAV 2 in 10 (14%) patients. Fifteen (21%) patients were on mammalian target of rapamycin receptor inhibitor (mTORi). Sixty patients (82%) were on statins and/or ezetimibe. Beta-blockers were administered prior to the CT scan in 51 (70%) patients. The median heart rate of the patients included in the study was 74 (66–84) beats per minute.
FFRct analysis
FFRct could be calculated in all patients in 214 coronary arteries including one with a stent (image quality was very good in this vessel and the stent created almost no artifacts). The LAD could not be analyzed in 3 patients and the LCx in 2 patients due to the presence of metallic stents. Eighteen (25%) patients had ≥ 1 focal coronary stenosis of > 30% with a FFRct value of ≤ 0.8. In the 55/73 (75%) patients without a focal stenosis with FFRct ≤ 0.8, the distal LAD FFRct was available for 54 vessels of which 40 (74%) had a distal LAD FFRct value of < 0.90. Ten out of the 55 (18%) patients had ≥ 1 coronary artery with a distal FFRct ≤ 0.8, including 1 patient that had an overall distal FFRct of ≤ 0.8. The V/M was available in 70 patients and was 25.4 (21.5–35.8) for the entire cohort.
Focal stenosis with FFRct ≤ 0.8 vs. no focal stenosis
Compared to those without a hemodynamically significant focal stenosis, patients with a focal stenosis and FFRct ≤ 0.8 had significantly longer time after HTx (15 (11–18) years vs. 10 (7–15) years, p = 0.02); had higher CAV scores prior to CCTA (p = 0.02); and more often had a coronary stent (p = 0.03) (Table 2). In patients with at least one focal stenosis > 30% with focal FFRct ≤ 0.8, the overall distal FFRct in the other coronary arteries (so only including those vessels without a focal > 30% stenosis with FFRct ≤ 0.8) was similar to patients that had no focal stenosis: 0.88 (0.86–0.91) vs. 0.88 (0.82–0.90), respectively (p = 0.15) (Table 3).
Time after HTx
Patients at a longer time since HTx more often had a coronary stenosis with FFRct ≤ 0.8 (Table 4). The overall distal FFRct as well as per vessel and the V/M did not differ significantly between groups. In the 55 patients without a focal stenosis with FFR ≤ 0.8, both the mean distal FFR and per vessel separately did not differ over time (Table 5).
Follow-up
At 1-year follow-up, 13 (18%) patients had undergone an additional test to assess the coronary arteries including 13 ICA, of which 8 with invasive FFR measurements, 1 stress scintigraphy, and 1 cardiac magnetic resonance scan (Tables 2 and 4). Additional testing was performed more frequently in case of a focal stenosis with FFRct ≤ 0.8 (p < 0.001) and in patients at a longer time after HTx (p = 0.001).
Three patients that underwent additional testing did not have a focal stenosis with FFRct ≤ 0.8 (Fig. 2). The ICA was requested based on CCTA findings before FFRct results were reviewed. The FFRct and ICA results of all 13 patients are detailed in the electronic supplementary material. Three patients did not have a focal stenosis with FFRct < 0.8 but did undergo ICA and one received revascularization of the LAD (FFRct 0.83) (electronic supplementary material). In the 10 patients with a FFRct ≤ 0.8 who underwent ICA, 7 underwent revascularization as the coronary stenosis was demonstrated to be significant either by an invasive FFR measurement ≤ 0.8, visual interpretation of the ICA, or findings at optical coherence tomography during the same session (Fig. 2). Of the 3 patients with a FFRct ≤ 0.8 who underwent ICA but were not revascularized, one did show chronic total occlusions, but these could not be treated; the other 2 patients did not show significant stenoses.
Patient with a focal stenosis in the right coronary artery (RCA) and mid left anterior descending (LAD) of > 30% with FFRct value of 0.92 and 0.85, respectively (b, d). Invasive coronary angiography (ICA) was performed that confirmed the FFRct finding of non-significant stenosis with an invasive FFR value of 0.96 in the RCA and 0.83 in the LAD (a, c). Patient with a focal stenosis in the RCA and LAD of > 30% with FFRct value of 0.94 and 0.76 respectively (f, h). ICA confirmed the lesion in the RCA to be non-significant (invasive FFR 0.95) and stenoses in the LAD (e, g). Additional stress scintigraphy demonstrated ischemia in the LAD territory and the LAD was stented in a subsequent session with good result (i)
Three patients had a MACE during follow-up, one being an iatrogenic infarction due to no reflow after stenting, one patient suffered a stroke after LAD stenting and later died of carcinoma, and one patient died of sudden cardiac death of unknown cause 10 months post-CCTA although the CCTA showed no abnormalities, and the patient had a coronary calcium score of 0.
Discussion
We describe the first cohort of HTx patients that underwent FFRct analysis of CCTA performed for routine annual screening for CAV. In our analysis, patients with a coronary stenosis and FFRct ≤ 0.8 were at a longer time since transplantation and more often underwent additional testing for coronary ischemia and revascularization.
Cardiac allograft vasculopathy
CAV constitutes a serious complication affecting HTx patients. The cause of CAV is likely multifactorial consisting of both alloimmune dependent and independent factors and has both donor and recipient related risk factors [13]. The ISHLT recommends (bi)annual ICA for screening but other techniques are used as well depending on local expertise and preferences including amongst others stress scintigraphy, intravascular ultrasound (IVUS), stress echocardiography, and CCTA [5, 14, 15]. Ideally, a technique would not only detect anatomical findings of CAV but also be able to predict its occurrence before structural changes become apparent. When medical treatment to slow down CAV progression should be initiated is a clinical dilemma, starting too early unnecessarily exposes the patient to side effects of the medication whereas starting too late appears to be ineffective due to a different plaque composition [1].
FFR measurements in transplant patients
Several studies describe invasive FFR measurements in HTx patients [6, 16,17,18]. Fearon et al. described 53 patients without angiographic disease at a mean of 3.1 ± 3.7 years post-transplant and showed the mean FFR in the LAD was 0.88 ± 0.07 and below the normal threshold of 0.94 in 75% of cases, less than 0.80 in 15% of patients, and even less than 0.75 in 6% [16]. In the same study, IVUS was performed and invasive FFR measurements showed a strong correlation with indexes of plaque burden [16]. Hence, invasive FFR measurements seem to detect CAV before anatomical changes become apparent on ICA. In our study, similar results were found. The 28 patients in our study at 5–9 years post-HTx (and thus closest in time after HTx compared to the invasive group mentioned above) had a LAD FFRct of 0.87 (0.82–0.91). Reported invasive FFR measurements in HTx patients were performed in the distal two thirds of the LAD [6]. This likely corresponds well to the distal FFRct as vessels are segmented down to 1.8 mm. FFRct, however, enables calculation of FFR values for each location in all coronary arteries which is a major advantage and provides more comprehensive information. Yang et al. showed that patients with baseline invasive FFR of < 0.90 have a significantly lower (42% vs. 79%) event-free survival of death or retransplantation at a mean of 4.5 ± 3.5 years follow-up [6]. FFR may therefore help to identify patients at increased risk of severe morbidity and mortality and tailor medication use accordingly. In our series, the overall distal FFR was only slightly lower at longer times since HTx and the difference was not statistically significant. Three patients suffered a MACE during follow-up in our study due to iatrogenic infarct, a carcinoma, and cardiac death with unknown cause. Our follow-up period is however limited to 1 year. With longer clinical follow-up including CCTA scans, we can assess how FFRct changes over time relate to outcome. Based on data from invasive FFR measurements, there may be two different mechanisms that lead to a reduction in FFR especially in the first year after HTx [18]. Patients with negative remodeling of the coronary arteries causing reduction in vessel volume without a change in plaque volume in the first year after HTx showed a decrease in invasive FFR from 0.88 ± 0.06 to 0.84 ± 0.07 [18]. Alternatively, patients without a decrease in vessel volume but an increase in plaque volume also showed reduction in invasive FFR from 0.89 ± 0.05 to 0.85 ± 0.05 [18]. This indicates changes in vessel volume due to negative remodeling itself without plaque progression may contribute to myocardial ischemia in HTx patients. Interestingly, we found a small reduction in mean distal FFRct at longer times after HTx albeit not statistically significant. The coronary artery volume and V/M are readily available from the image segmentation to compute FFRct values and may provide valuable additional information in HTx patients regarding vessel remodeling over time [12].
Heart transplant patients with CAV may have microvascular dysfunction to some degree. In subgroups of patients prone to have microvascular dysfunction (e.g., hypertension and diabetes) FFRct accuracy is not affected [19, 20]. With microvascular disease, there may be diminished coronary flow reserve with a normal FFR. However, it does not imply that FFR is less accurate, only that there may not be epicardial disease appropriate for revascularization.
Limitations
Given the cross-sectional nature of the study, potentially, the patients that were at a longer time after transplantation represents a group that has less severe CAV and/or slower progression of CAV.
FFRct has not been validated against invasive FFR in HTx patients. FFRct is however extensively validated in stable chest pain patients demonstrating good correlation and reproducibility of measurements [21]. A general limitation of FFRct analysis in HTx patients is that some patients cannot undergo either CCTA due to poor kidney function or FFRct analysis due to the presence of stents in two or more major coronary arteries. The follow-up time of 1 year in our study is relatively limited. Invasive FFR in heart transplant patients carries prognostic information as described by Yang et al. [6]. Longer follow-up is needed to establish if FFRct provides similar prognostic value in heart transplant patients.
Future outlook
Anatomical coronary artery assessment on CCTA combined with additional FFRct analysis, V/M, plaque analysis, and quantification from the same CCTA dataset could provide a comprehensive non-invasive assessment in HTx patients. It will be necessary to evaluate changes over time of the above-mentioned parameters and their relation to the occurrence of adverse events during longer term follow-up. Potentially, the current CAV grading classification may even be altered and/or expanded using these parameters to more accurately identify which patients will develop CAV and how the disease will progress with the goal to determine the optimal time point to adjust medical therapy. Newer drugs like PCSK9 inhibitors have only been described in small case series without a long follow-up and at the moment, it is not known if this is a sensible adjunctive therapy in this patient category [22]. Maybe CCTA and FFRct may play a role in the evaluation of the effect of new therapies.
In conclusion, FFRct was successfully performed on CCTA scans of HTx patients and demonstrated that more than a quarter of patients had a focal coronary stenosis with FFRct ≤ 0.8. Even in the absence of a focal stenosis, FFRct values are often abnormal in HTx patients.
Abbreviations
- CAV:
-
Cardiac allograft vasculopathy
- CCTA:
-
Coronary computed tomography angiography
- CMV:
-
Cytomegalovirus
- CNI:
-
Calcineurin inhibitor
- FFR:
-
Fractional flow reserve
- FFRct:
-
Coronary computed tomography angiography–derived fractional flow reserve
- HTx:
-
Heart transplant
- ICA:
-
Invasive coronary angiography
- ISHLT:
-
International society for heart and lung transplantation
- IVUS:
-
Intravascular ultrasound
- LAD:
-
Left anterior descending
- LCx:
-
Left circumflex
- MACE:
-
Major adverse cardiovascular events
- mTORi:
-
Mammalian target of rapamycin receptor inhibitor
- RCA:
-
Right coronary artery
- SPECT:
-
Single-photon emission computed tomography
- V/M:
-
Volume-to-mass ratio
References
Chih S, Chong AY, Mielniczuk LM, Bhatt DL, Beanlands RSB (2016) Allograft vasculopathy the Achilles’ heel of heart transplantation. J Am Coll Cardiol 68:80–91
Mehra MR, Crespo-Leiro MG, Dipchand A et al (2010) International Society for Heart and Lung Transplantation working formulation of a standardized nomenclature for cardiac allograft vasculopathy-2010. J Hear Lung Transplant 29:717–727
ISHLT. ISHLT 2018 slides adult heart transplantation statistics. https://ishltregistries.org/registries/slides.asp.
Yusen RD, Edwards LB, Kucheryavaya AY et al (2014) The registry of the international society for heart and lung transplantation: thirty-first adult lung and heart-lung transplant report - 2014; Focus theme: Retransplantation. J Hear Lung Transplant 33:1009–1024
Costanzo MR, Dipchand A, Starling R et al (2010) The international society of heart and lung transplantation guidelines for the care of heart transplant recipients. J Hear Lung Transplant 29:914–956
Yang HM, Khush K, Luikart H et al (2016) Invasive assessment of coronary physiology predicts late mortality after heart transplantation. Circulation 133:1945–1950
Wever-Pinzon O, Romero J, Kelesidis I et al (2014) Coronary computed tomography angiography for the detection of cardiac allograft vasculopathy: a meta-analysis of prospective trials. J Am Coll Cardiol 63:2005–2006
Fairbairn TA, Nieman K, Akasaka T et al (2018) Real-world clinical utility and impact on clinical decision-making of coronary computed tomography angiography-derived fractional flow reserve: lessons from the ADVANCE Registry. Eur Heart J 39:3701–3711
Nørgaard BL, Leipsic J, Gaur S et al (2014) Diagnostic performance of noninvasive fractional flow reserve derived from coronary computed tomography angiography in suspected coronary artery disease: The NXT trial (Analysis of Coronary Blood Flow Using CT Angiography: Next Steps). J Am Coll Cardiol 63:1145–1155
Min JK, Leipsic J, Pencina MJ et al (2012) Diagnostic accuracy of fractional flow reserve from anatomic CT angiography. JAMA 308:1237–1245
Driessen RS, Danad I, Stuijfzand WJ et al (2019) Comparison of coronary computed tomography angiography, fractional flow reserve, and perfusion imaging for ischemia diagnosis. J Am Coll Cardiol 73:161–173
Taylor CA, Gaur S, Leipsic J et al (2017) Journal of Cardiovascular Computed Tomography Effect of the ratio of coronary arterial lumen volume to left ventricle myocardial mass derived from coronary CT angiography on fractional fl ow reserve. J Cardiovasc Comput Tomogr 11:429–436
Schmauss D, Weis M (2008) Cardiac allograft vasculopathy: recent developments. Circulation 117:2131–2141
Olymbios M, Kwiecinski J, Berman DS, Kobashigawa JA (2018) Imaging in heart transplant patients. JACC Cardiovasc Imaging 11:1514–1530
Veenis JF, Boiten HJ, van Den Berge JC et al (2019) Prediction of long-term (> 10 year) cardiovascular outcomes in heart transplant recipients: value of stress technetium-99m tetrofosmin myocardial perfusion imaging. J Nucl Cardiol 26:845–852
Fearon WF, Nakamura M, Lee DP et al (2003) Simultaneous assessment of fractional and coronary flow reserves in cardiac transplant recipients: physiologic investigation for transplant arteriopathy (PITA study). Circulation 108:1605–1610
Kobayashi Y, Kobayashi Y, Yang HM et al (2018) Long-term prognostic value of invasive and non-invasive measures early after heart transplantation. Int J Cardiol 260:31–35
Fearon WF, Felix R, Hirohata A et al (2017) The effect of negative remodeling on fractional flow reserve after cardiac transplantation. Int J Cardiol 241:283–287
Nous F, Coenen A, Boersma E et al (2019) Comparison of the diagnostic performance of coronary computed tomography angiography derived fractional flow reserve in patients with versus without diabetes mellitus. Am J Cardiol 123:537–543
Eftekhari A, Min J, Achenbach S et al (2017) Fractional flow reserve derived from coronary computed tomography angiography : diagnostic performance in hypertensive and diabetic patients. Eur Heart J Cardiovasc Imaging 18:1351–1360
Koo BK, Erglis A, Doh JH et al (2011) Diagnosis of ischemia-causing coronary stenoses by noninvasive fractional flow reserve computed from coronary computed tomographic angiograms: results from the prospective multicenter DISCOVER-FLOW (Diagnosis of Ischemia-Causing Stenoses Obtained Via Noni. J Am Coll Cardiol 58:1989–1997
Jennings DL, Jackson R, Farr M (2019) PCSK9 inhibitor use in heart transplant recipients: a case series and review of the literature. Transplantation 104:e38–e39
Funding
This study has received funding from Heartflow.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is R. Budde, MD, PHD.
Conflict of interest
The authors of this manuscript declare relationships with the following companies: Heartflow, Siemens. Dr. Budde reports institutional research support from HeartFlow and Siemens Healthineers. Dr. Nous has received travel reimbursement from Heartflow, Inc. Dr. Nieman reports non-financial support from Heartflow Inc. Dr. Koweek serves as a consultant to Heartflow, Inc. Dr. Leipsic serves as a consultant to, and has stock options in, Heartflow, Inc. The other authors report no conflicts.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional review board approval was obtained.
Methodology
• Retrospective
• cross-sectional study
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Budde, R.P.J., Nous, F.M.A., Roest, S. et al. CT-derived fractional flow reserve (FFRct) for functional coronary artery evaluation in the follow-up of patients after heart transplantation. Eur Radiol 32, 1843–1852 (2022). https://doi.org/10.1007/s00330-021-08246-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-021-08246-5