Abstract
Objectives
Pancreatic ductal adenocarcinoma (PDAC) and autoimmune pancreatitis (AIP) are diseases with a highly analogous visual presentation that are difficult to distinguish by imaging. The purpose of this research was to create a radiomics-based prediction model using dual-time PET/CT imaging for the noninvasive classification of PDAC and AIP lesions.
Methods
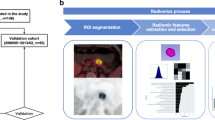
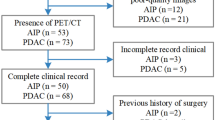
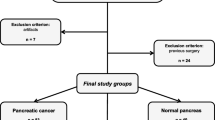
This retrospective study was performed on 112 patients (48 patients with AIP and 64 patients with PDAC). All cases were confirmed by imaging and clinical follow-up, and/or pathology. A total of 502 radiomics features were extracted from the dual-time PET/CT images to develop a radiomics decision model. An additional 12 maximum intensity projection (MIP) features were also calculated to further improve the radiomics model. The optimal radiomics feature set was selected by support vector machine recursive feature elimination (SVM-RFE), and the final classifier was built using a linear SVM. The performance of the proposed dual-time model was evaluated using nested cross-validation for accuracy, sensitivity, specificity, and area under the curve (AUC).
Results
The final prediction model was developed from a combination of the SVM-RFE and linear SVM with the required quantitative features. The multimodal and multidimensional features performed well for classification (average AUC: 0.9668, accuracy: 89.91%, sensitivity: 85.31%, specificity: 96.04%).
Conclusions
The radiomics model based on 2-[18F]fluoro-2-deoxy-D-glucose (2-[18F]FDG) PET/CT dual-time images provided promising performance for discriminating between patients with benign AIP and malignant PDAC lesions, which shows its potential for use as a diagnostic tool for clinical decision-making.
Key Points
• The clinical symptoms and imaging visual presentations of PDAC and AIP are highly similar, and accurate differentiation of PDAC and AIP lesions is difficult.
• Radiomics features provided a potential noninvasive method for differentiation of AIP from PDAC.
• The diagnostic performance of the proposed radiomics model indicates its potential to assist doctors in making treatment decisions.



Similar content being viewed by others
Materials availability
Requests for materials should be addressed to Chao Cheng, M.D. and Xiaodong Yang, Ph.D.
Abbreviations
- 2-[18F]FDG:
-
2-[18F]Fluoro-2-deoxy-D-glucose
- 2D:
-
Two dimensional
- 3D:
-
Three dimensional
- AIP:
-
Autoimmune pancreatitis
- CV:
-
Cross-validation
- MIP:
-
Maximal intensity projection
- PDAC:
-
Pancreatic ductal adenocarcinoma
- PET/CT:
-
Positron emission tomography/computerized tomography
- RI:
-
Mean retention index
- ROC:
-
Receiver operating characteristic
- ROI:
-
Region of interest
- SUVmax :
-
Maximum standardized uptake value
- SVM:
-
Support vector machine
- VOI:
-
Volume of interest
References
Ferrone CR, Pieretti-Vanmarcke R, Bloom JP et al (2012) Pancreatic ductal adenocarcinoma: long-term survival does not equal cure. Surgery 152:S43–S49
Vincent A, Herman J, Schulick R, Goggins M (2011) Pancreatic cancer. Lancet 378:607–620
Finkelberg DL, Sahani D, Deshpande V, Brugge WR (2006) Autoimmune pancreatitis. N Engl J Med 355:2670–2676
Komatsu K, Hamano H, Ochi Y et al (2005) High prevalence of hypothyroidism in patients with autoimmune pancreatitis. Dig Dis Sci 50:1052–1057
Kamisawa T, Egawa N, Nakajima H, Tsuruta K, Okamoto A, Kamata N (2003) Clinical difficulties in the differentiation of autoimmune pancreatitis and pancreatic carcinoma. Am J Gastroenterol 98:2694–2699
Carruthers MN, Khosroshahi A, Augustin T, Deshpande V, Stone JH (2015) The diagnostic utility of serum IgG4 concentrations in IgG4-related disease. Ann Rheum Dis 74:14–18
Pitchumoni CS, Chari S (2013) Ulcerative colitis and autoimmune pancreatitis. J Clin Gastroenterol 47:469–470
Dickerson LD, Farooq A, Bano F et al (2019) Differentiation of autoimmune pancreatitis from pancreatic cancer remains challenging. World J Surg 43:1604–1611
Meng Q, Xin L, Liu W et al (2015) Diagnosis and treatment of autoimmune pancreatitis in China: a systematic review. Plos One 10:e0130466
Vijayakumar A, Vijayakumar A (2013) Imaging of focal autoimmune pancreatitis and differentiating it from pancreatic cancer. ISRN Radiol. https://doi.org/10.5402/2013/569489
Lee TY, Kim MH, Park DH et al (2009) Utility of 18F-FDG PET/CT for differentiation of autoimmune pancreatitis with atypical pancreatic imaging findings from pancreatic cancer. AJR Am J Roentgenol 193:343–348
Ozaki Y, Oguchi K, Hamano H et al (2008) Differentiation of autoimmune pancreatitis from suspected pancreatic cancer by fluorine-18 fluorodeoxyglucose positron emission tomography. J Gastroenterol 43:144–151
Cheng MF, Guo YL, Yen RF et al (2018) Clinical utility of FDG PET/CT in patients with autoimmune pancreatitis: a case-control study. Sci Rep 8:1–8
Cheng MF, Guo YL, Chen YC et al (2016) Combined PET SUV measurements and texture analysis in differentiating pancreatic cancer from autoimmune pancreatitis. J Nucl Med 57:1594
Jian Z, Jia G, Zuo C, Jia N, Wang H (2017) 18F-FDG PET/CT helps differentiate autoimmune pancreatitis from pancreatic cancer. BMC Cancer 17:695
Lambin P, Rios-Velazquez E, Leijenaar R et al (2012) Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer 48:441–446
Alic L, Niessen WJ, Veenland JF (2014) Quantification of heterogeneity as a biomarker in tumor imaging: a systematic review. Plos One 9:e110300
Hawkins SH, Korecki JN, Balagurunathan Y et al (2014) Predicting outcomes of nonsmall cell lung cancer using CT image features. IEEE Access 2:1418–1426
Shen C, Liu Z, Guan M et al (2017) 2D and 3D CT radiomics features prognostic performance comparison in non-small cell lung cancer. Transl Oncol 10:886–894
Leger S, Zwanenburg A, Pilz K et al (2017) A comparative study of machine learning methods for time-to-event survival data for radiomics risk modeling. Sci Rep 7:1–11
Song J, Shi J, Dong D, Fang M et al (2018) A new approach to predict progression-free survival in stage IV EGFR-mutant NSCLC patients with EGFR-TKI therapy. Clin Cancer Res 24:3583–3592
Dong D, Zhang F, Zhong LZ et al (2019) Development and validation of a novel MR imaging predictor of response to induction chemotherapy in locoregionally advanced nasopharyngeal cancer: a randomized controlled trial substudy (NCT01245959). BMC Med 17:190
Chen Y, Chen TW, Wu CQ et al (2019) Radiomics model of contrast-enhanced computed tomography for predicting the recurrence of acute pancreatitis. Eur Radiol 29:4408–4417
Wu W, Pierce LA, Zhang Y et al (2019) Comparison of prediction models with radiological semantic features and radiomics in lung cancer diagnosis of the pulmonary nodules: a case-control study. Eur Radiol 29:6100–6108
Liu S, He J, Liu S et al (2020) Radiomics analysis using contrast-enhanced CT for preoperative prediction of occult peritoneal metastasis in advanced gastric cancer. Eur Radiol 30:239–246
Rizzo S, Botta F, Raimondi S et al (2018) Radiomics of high-grade serous ovarian cancer: association between quantitative CT features, residual tumour and disease progression within 12 months. Eur Radiol 28:4849–4859
Zhang Y, Cheng C, Liu Z et al (2019) Radiomics analysis for the differentiation of autoimmune pancreatitis and pancreatic ductal adenocarcinoma in 18F-FDG PET/CT. Med Phys 46:4520–4530
Parghane RV, Basu S (2017) Dual-time point 18F-FDG-PET and PET/CT for differentiating benign from malignant musculoskeletal lesions: opportunities and limitations[C]. Semin Nucl Med 47:373–391
Chen S, Harmon S, Perk T et al (2017) Diagnostic classification of solitary pulmonary nodules using dual time 18 F-FDG PET/CT image texture features in granuloma-endemic regions. Sci Rep 7:1–8
Sa R, Zhao HG, Dai YY, Guan F (2018) The role of dual time point PET/CT for distinguishing malignant from benign focal 18F-FDG uptake duodenal lesions. Medicine (Baltimore). https://doi.org/10.1097/MD.0000000000012521
Nakajo M, Jinguji M, Aoki M, Tani A, Sato M, Yoshiura T (2020) The clinical value of texture analysis of dual-time-point 18 F-FDG-PET/CT imaging to differentiate between 18 F-FDG-avid benign and malignant pulmonary lesions. Eur Radiol 30:1759–1769
Pieper S, Halle M, Kikinis R (2004) 3D Slicer. IEEE International Symposium on Biomedical Imaging: Nano to Macro 26:632–635
Kubota K (1985) Lung tumor imaging by positron emission tomography using C-11 L-methionine. J Nucl Med 26:37–42
Guyon I, Weston J, Barnhill S, Vapnik V (2002) Gene selection for cancer classification using support vector machines. Mach Learn 46:389–422
DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837–845
Chang CC, Lin CJ (2011) LIBSVM: a library for support vector machines. ACM Trans Intell Syst Technol 2:1–27
Kaalep A, Sera T, Oyen W et al (2018) EANM/EARL FDG-PET/CT accreditation-summary results from the first 200 accredited imaging systems. Eur J Nucl Med Mol Imaging 45:412–422
Houdu B, Lasnon C, Licaj I et al (2019) Why harmonization is needed when using FDG PET/CT as a prognosticator: demonstration with EARL-compliant SUV as an independent prognostic factor in lung cancer. Eur J Nucl Med Mol Imaging 46:421–428
Boellaard R, Delgado-Bolton R, Oyen W et al (2015) FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging 42:328–354
Kesner AL, Chung JH, Lind KE et al (2016) Validation of software gating: a practical technology for respiratory motion correction in PET. Radiology 281:239–248
Pan T (2018) Respiratory gating in PET/CT: a step in the right direction. J Nucl Cardiol 416–418
Lu Y, Fontaine K, Mulnix T et al (2018) Respiratory motion compensation for PET/CT with motion information derived from matched attenuation-corrected gated PET data. J Nucl Med 59:1480–1486
Acknowledgements
We thank to Yuquan Zhang for his helpful suggestions on this project and Fei Wang for the assistance in statistics and data visualization.
Funding
This study was funded by the National Natural Science Foundation of China (No. 61701492), the National Key Research and Development Program of China (No. 2016YFC0103502), the Wenzhou Science and Technology Foundation (No. ZS2017020), and as a medical artificial intelligence project by the Sun Yat-Sen Memorial Hospital (No.YXRGZN201905).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is Prof. Xiaodong Yang.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Study subjects or cohorts overlap
Some study subjects or cohorts have been previously reported in other journals.
The previous work published in the journal Medical Physics was named “Radiomics analysis for the differentiation of autoimmune pancreatitis and pancreatic ductal adenocarcinoma in 18F-FDG PET/CT.” Early work only focused on comparisons of feature selection strategies and classifiers using data from PET/CT single-time imaging. The number of patients was 45 AIP and 66 PDAC, which partially overlaps with our current case. Our current work is mainly to study the advantages of PET/CT dual-time imaging in distinguishing AIP and PDAC via radiomics analysis. Our number of patients is 48 AIP cases and 64 PDAC cases.
Methodology
• retrospective
• diagnostic or prognostic study
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 20 kb)
Rights and permissions
About this article
Cite this article
Liu, Z., Li, M., Zuo, C. et al. Radiomics model of dual-time 2-[18F]FDG PET/CT imaging to distinguish between pancreatic ductal adenocarcinoma and autoimmune pancreatitis. Eur Radiol 31, 6983–6991 (2021). https://doi.org/10.1007/s00330-021-07778-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-021-07778-0