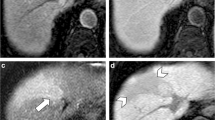
Abstract
Objectives
This study assesses the risk of progression of Liver Imaging Reporting and Data System (LI-RADS) categories, and the effects of inter-exam changes in modality or radiologist on LI-RADS categorization.
Methods
Clinical LI-RADS v2014 CT and MRI exams at our institution between January 2014 and September 2017 were retrospectively identified. Untreated LR-1, LR-2, LR-3, and LR-4 observations with at least one follow-up exam were included. Three hundred and seventy-two observations in 214 patients (149 male, 65 female, mean age 61 ± 10 years) were included during the study period (715 exams total). Cumulative incidence curves for progression to malignant LI-RADS categories (LR-5 or LR-M) and to LR-4 or higher were generated for each index category and compared using log-rank tests with a resampling extension. Relationships between inter-exam changes in LI-RADS category and modality or radiologist, adjusted for inter-exam time intervals, were modeled using mixed effect logistic regressions.
Results
Median inter-exam follow-up interval and total follow-up duration were 123 and 227 days, respectively. Index LR-1, LR-2, LR-3, and LR-4 differed significantly in their cumulative incidences of progression to malignant categories (p < 0.0001), which were 0%, 2%, 7%, and 32% at 6 months, respectively. Index LR-1, LR-2, and LR-3 differed significantly in cumulative incidences of progression to LR-4 or higher (p = 0.003). MRI-MRI exam pairs had more stable LI-RADS categorization compared to CT-CT (OR = 0.460, p = 0.0018).
Conclusions
LI-RADS observations demonstrate increasing risk of progression to malignancy with increasing category ranging from 0% for LR-1 to 32% for LR-4 at 6 months. Inter-exam modality changes are associated with LI-RADS category changes.
Key Points
• While the majority of LR-2 observations remain stable over long-term follow-up, LR-3 and especially LR-4 observations have a higher risk for category progression.
• Category transitions between sequential exams using different modalities (CT vs. MRI) may reflect modality differences rather than biological change. MRI, especially with the same type of contrast agent, may provide the most reproducible categorization, although this needs additional validation.
• In a clinical practice setting, in which radiologists refer to prior imaging and reports, there was no significant association between changes in radiologist and changes in LI-RADS categorization.





Similar content being viewed by others
Abbreviations
- HCC:
-
Hepatocellular carcinoma
- IQR:
-
Inter-quartile range
- OR:
-
Odds ratio
- LI-RADS:
-
Liver Imaging Reporting and Data System
References
Heimbach JK, Kulik LM, Finn RS et al (2018) AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology 67:358–380
Mitchell DG, Bruix J, Sherman M, Sirlin CB (2015) LI-RADS (liver imaging reporting and data system): summary, discussion, and consensus of the LI-RADS management working group and future directions. Hepatology 61:1056–1065
Kielar AZ, Elsayes KM, Chernyak V, Tang A, Sirlin CB (2018) LI-RADS version 2018: what is new and what does this mean to my radiology reports? Abdom Radiol (NY). https://doi.org/10.1007/s00261-018-1730-x
Elsayes KM, Kielar AZ, Agrons MM et al (2017) Liver imaging reporting and data system: an expert consensus statement. J Hepatocell Carcinoma 4:29–39
Wald C, Russo MW, Heimbach JK, Hussain HK, Pomfret EA, Bruix J (2013) New OPTN/UNOS policy for liver transplant allocation: standardization of liver imaging, diagnosis, classification, and reporting of hepatocellular carcinoma. Radiology 266:376–382
Tang A, Bashir MR, Corwin MT et al (2018) Evidence supporting LI-RADS major features for CT- and MR imaging-based diagnosis of hepatocellular carcinoma: a systematic review. Radiology 286:29–48
Chen N, Motosugi U, Morisaka H et al (2016) Added value of a gadoxetic acid-enhanced hepatocyte-phase image to the LI-RADS system for diagnosing hepatocellular carcinoma. Magn Reson Med Sci 15:49–59
Choi SH, Byun JH, Kim SY et al (2016) Liver imaging reporting and data system v2014 with gadoxetate disodium-enhanced magnetic resonance imaging: validation of LI-RADS category 4 and 5 criteria. Invest Radiol 51:483–490
Cha DI, Jang KM, Kim SH, Kang TW, Song KD (2017) Liver imaging reporting and data system on CT and gadoxetic acid-enhanced MRI with diffusion-weighted imaging. Eur Radiol 27:4394–4405
Abd Alkhalik Basha M, Abd El Aziz El Sammak D, El Sammak AA (2017) Diagnostic efficacy of the Liver Imaging-Reporting and Data System (LI-RADS) with CT imaging in categorising small nodules (10–20 mm) detected in the cirrhotic liver at screening ultrasound. Clin Radiol 72:901.e1–901.e11
Kim YY, An C, Kim S, Kim MJ (2018) Diagnostic accuracy of prospective application of the Liver Imaging Reporting and Data System (LI-RADS) in gadoxetate-enhanced MRI. Eur Radiol 28:2038–2046
Liu W, Qin J, Guo R et al (2018) Accuracy of the diagnostic evaluation of hepatocellular carcinoma with LI-RADS. Acta Radiol 59:140–146
Tanabe M, Kanki A, Wolfson T et al (2016) Imaging outcomes of Liver Imaging Reporting and Data System version 2014 category 2, 3, and 4 observations detected at CT and MR imaging. Radiology 281:129–139
Choi JY, Cho HC, Sun M, Kim HC, Sirlin CB (2013) Indeterminate observations (Liver Imaging Reporting and Data System category 3) on MRI in the cirrhotic liver: fate and clinical implications. AJR Am J Roentgenol 201:993–1001
Burke LMB, Sofue K, Alagiyawanna M et al (2016) Natural history of liver imaging reporting and data system category 4 nodules in MRI. Abdom Radiol (NY) 41:1758–1766
Sofue K, Burke LMB, Nilmini V et al (2017) Liver imaging reporting and data system category 4 observations in MRI: risk factors predicting upgrade to category 5. J Magn Reson Imaging 46:783–792
Law CG, Brookmeyer R (1992) Effects of mid-point imputation on the analysis of doubly censored data. Stat Med 11:1569–1578
Mitchell DG, Bashir MR, Sirlin CB (2017) Management implications and outcomes of LI-RADS-2, -3, -4, and -M category observations. Abdom Radiol (NY) 43:143–148
Zhang YD, Zhu FP, Xu X et al (2016) Liver Imaging Reporting and Data System:: substantial discordance between CT and MR for imaging classification of hepatic nodules. Acad Radiol 23:344–352
Chernyak V, Flusberg M, Law A, Kobi M, Paroder V, Rozenblit AM (2017) Liver imaging reporting and data system: discordance between computed tomography and gadoxetate-enhanced magnetic resonance imaging for detection of hepatocellular carcinoma major features. J Comput Assist Tomogr 42(1). https://doi.org/10.1097/RCT.0000000000000642
Fowler KJ, Tang A, Santillan C et al (2018) Interreader reliability of LI-RADS version 2014 algorithm and imaging features for diagnosis of hepatocellular carcinoma: a large international multireader study. Radiology 286:173–185
Davenport MS, Khalatbari S, Liu PS et al (2014) Repeatability of diagnostic features and scoring systems for hepatocellular carcinoma by using MR imaging. Radiology 272:132–142
Barth BK, Donati OF, Fischer MA et al (2016) Reliability, validity, and reader acceptance of LI-RADS—an in-depth analysis. Acad Radiol 23:1145–1153
Ehman EC, Behr SC, Umetsu SE et al (2016) Rate of observation and inter-observer agreement for LI-RADS major features at CT and MRI in 184 pathology proven hepatocellular carcinomas. Abdom Radiol (NY) 41:963–969
Zhang YD, Zhu FP, Xu X et al (2016) Classifying CT/MR findings in patients with suspicion of hepatocellular carcinoma: comparison of liver imaging reporting and data system and criteria-free Likert scale reporting models. J Magn Reson Imaging 43:373–383
Lee CS, Nagy PG, Weaver SJ, Newman-Toker DE (2013) Cognitive and system factors contributing to diagnostic errors in radiology. AJR Am J Roentgenol 201:611–617
Bruno MA, Walker EA, Abujudeh HH (2015) Understanding and confronting our mistakes: the epidemiology of error in radiology and strategies for error reduction. Radiographics 35:1668–1676
Acknowledgements
A part of these results was presented at the 2017 RSNA Annual Meeting as a paper presentation.
Funding
The authors acknowledge grant support from National Institutes of Health T32 EB005970-09.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Cheng William Hong, MD MS.
Conflict of interest
The authors of this manuscript declare relationships with the following companies:
Claude B Sirlin, MD, is a member of the external advisory board of AMRA Medical and Guerbet and speaker for GE Healthcare and Bayer (fees paid to University of California Regents); a consultant for Boehringer Ingelheim; receives research grants from Gilead, GE Healthcare, Siemens, GE MRI, Bayer AMRI, GE Digital, GE US, and ACR Innovation; gives education presentations for Medscape; and performs contracted research for ICON Medical Imaging/Enanta, Philips, Gilead, Shire, Virtualscopics, Intercept, and Synageva.
Statistics and biometry
Tanya Wolfson, MA, and Anthony Gamst, PhD, kindly provided statistical advice for this manuscript. Both of these authors have significant statistical expertise.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• observational
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 24 kb)
Rights and permissions
About this article
Cite this article
Hong, C.W., Park, C.C., Mamidipalli, A. et al. Longitudinal evolution of CT and MRI LI-RADS v2014 category 1, 2, 3, and 4 observations. Eur Radiol 29, 5073–5081 (2019). https://doi.org/10.1007/s00330-019-06058-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-019-06058-2