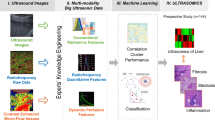
Abstract
Objectives
To develop a machine learning model based on quantitative ultrasound (QUS) parameters to improve classification of steatohepatitis with shear wave elastography in rats by using histopathology scoring as the reference standard.
Methods
This study received approval from the institutional animal care committee. Sixty male Sprague-Dawley rats were either fed a standard chow or a methionine- and choline-deficient diet. Ultrasound-based radiofrequency images were recorded in vivo to generate QUS and elastography maps. Random forests classification models and a bootstrap method were used to identify the QUS parameters that improved the classification accuracy of elastography. Receiver-operating characteristic analyses were performed.
Results
For classification of not steatohepatitis vs borderline or steatohepatitis, the area under the receiver-operating characteristic curve (AUC) increased from 0.63 for elastography alone to 0.72 for a model that combined elastography and QUS techniques (p < 0.001). For detection of liver steatosis grades 0 vs ≥ 1, ≤ 1 vs ≥ 2, ≤ 2 vs 3, respectively, the AUCs increased from 0.70, 0.65, and 0.69 to 0.78, 0.78, and 0.75 (p < 0.001). For detection of liver inflammation grades 0 vs ≥ 1, ≤ 1 vs ≥ 2, ≤ 2 vs 3, respectively, the AUCs increased from 0.58, 0.77, and 0.78 to 0.66, 0.84, and 0.87 (p < 0.001). For staging of liver fibrosis grades 0 vs ≥ 1, ≤ 1 vs ≥ 2, and ≤ 2 vs ≥ 3, respectively, the AUCs increased from 0.79, 0.92, and 0.91 to 0.85, 0.98, and 0.97 (p < 0.001).
Conclusion
QUS parameters improved the classification accuracy of steatohepatitis, liver steatosis, inflammation, and fibrosis compared to shear wave elastography alone.
Key Points
• Quantitative ultrasound and shear wave elastography improved classification accuracy of liver steatohepatitis and its histological features (liver steatosis, inflammation, and fibrosis) compared to elastography alone.
• A machine learning approach based on random forest models and incorporating local attenuation and homodyned-K tissue modeling shows promise for classification of nonalcoholic steatohepatitis.
• Further research should be performed to demonstrate the applicability of this multi-parametric QUS approach in a human cohort and to validate the combinations of parameters providing the highest classification accuracy.




Similar content being viewed by others
Abbreviations
- AUC:
-
Area under the receiver-operating characteristic curve
- CAP:
-
Controlled attenuation parameter
- HKD:
-
Homodyned-K distributions
- HPS:
-
Hematoxylin phloxine saffron
- IQR:
-
Inter-quartile range
- MCD:
-
Methionine and choline deficient
- NAFLD:
-
Nonalcoholic fatty liver disease
- NASH:
-
Nonalcoholic steatohepatitis
- QUS:
-
Quantitative ultrasound
- ROC:
-
Receiver operating characteristic
- ROI:
-
Region of interest
- SH:
-
Steatohepatitis
References
Vernon G, Baranova A, Younossi ZM (2011) Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther 34:274–285
Kleiner DE, Brunt EM, Van Natta M et al (2005) Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 41:1313–1321
Sanyal AJ, Brunt EM, Kleiner DE et al (2011) Endpoints and clinical trial design for nonalcoholic steatohepatitis. Hepatology 54:344–353
Fernández-Salazar L, Velayos B, Aller R, Lozano F, Garrote JA, González JM (2011) Percutaneous liver biopsy: patients’ point of view. Scand J Gastroenterol 46:727–731
Myers RP, Fong A, Shaheen AA (2008) Utilization rates, complications and costs of percutaneous liver biopsy: a population-based study including 4275 biopsies. Liver Int 28:705–712
Reeder SB, Cruite I, Hamilton G, Sirlin CB (2011) Quantitative assessment of liver fat with magnetic resonance imaging and spectroscopy. J Magn Reson Imaging 34:spcone
Tang A, Desai A, Hamilton G et al (2015) Accuracy of MR imaging-estimated proton density fat fraction for classification of dichotomized histologic steatosis grades in nonalcoholic fatty liver disease. Radiology 274:416–425
Yokoo T, Serai SD, Pirasteh A et al (2018) Linearity, bias, and precision of hepatic proton density fat fraction measurements by using MR imaging: a meta-analysis. Radiology 286:486–498
Petitclerc L, Sebastiani G, Gilbert G, Cloutier G, Tang A (2017) Liver fibrosis: review of current imaging and MRI quantification techniques. J Magn Reson Imaging 45:1276–1295
Singh S, Venkatesh SK, Wang Z et al (2015) Diagnostic performance of magnetic resonance elastography in staging liver fibrosis: a systematic review and meta-analysis of individual participant data. Clin Gastroenterol Hepatol 13:440–451 e446
Salameh N, Larrat B, Abarca-Quinones J et al (2009) Early detection of steatohepatitis in fatty rat liver by using MR elastography. Radiology 253:90–97
Yin M, Glaser KJ, Manduca A et al (2017) Distinguishing between hepatic inflammation and fibrosis with MR Elastography. Radiology 284:694–705
Zhang E, Wartelle-Bladou C, Lepanto L, Lachaine J, Cloutier G, Tang A (2015) Cost-utility analysis of nonalcoholic steatohepatitis screening. Eur Radiol 25:3282–3294
Park CC, Nguyen P, Hernandez C et al (2017) Magnetic resonance elastography vs transient elastography in detection of fibrosis and noninvasive measurement of steatosis in patients with biopsy-proven nonalcoholic fatty liver disease. Gastroenterology 152:598–607.e592
de Lédinghen V, Wong GL, Vergniol J et al (2016) Controlled attenuation parameter for the diagnosis of steatosis in non-alcoholic fatty liver disease. J Gastroenterol Hepatol 31:848–855
Chan WK, Nik Mustapha NR, Mahadeva S (2014) Controlled attenuation parameter for the detection and quantification of hepatic steatosis in nonalcoholic fatty liver disease. J Gastroenterol Hepatol 29:1470–1476
Kumar M, Rastogi A, Singh T et al (2013) Controlled attenuation parameter for non-invasive assessment of hepatic steatosis: does etiology affect performance? J Gastroenterol Hepatol 28:1194–1201
Lin SC, Heba E, Wolfson T et al (2015) Noninvasive diagnosis of nonalcoholic fatty liver disease and quantification of liver fat using a new quantitative ultrasound technique. Clin Gastroenterol Hepatol 13:1337–1345.e1336
Paige JS, Bernstein GS, Heba E et al (2017) A pilot comparative study of quantitative ultrasound, conventional ultrasound, and MRI for predicting histology-determined steatosis grade in adult nonalcoholic fatty liver disease. AJR Am J Roentgenol 208:W168–w177
Friedrich-Rust M, Nierhoff J, Lupsor M et al (2012) Performance of acoustic radiation force impulse imaging for the staging of liver fibrosis: a pooled meta-analysis. J Viral Hepat 19:e212–e219
Friedrich-Rust M, Ong MF, Martens S et al (2008) Performance of transient elastography for the staging of liver fibrosis: a meta-analysis. Gastroenterology 134:960–974
Oelze ML, Mamou J (2016) Review of quantitative ultrasound: envelope statistics and backscatter coefficient imaging and contributions to diagnostic ultrasound. IEEE Trans Ultrason Ferroelectr Freq Control 63:336–351
Mamou J, Oelze ML (2013) Quantitative ultrasound in soft tissues. Springer, Dordrecht
Kazemirad S, Zhang E, Nguyen BN et al (2017) Detection of steatohepatitis in a rat model by using spectroscopic shear-wave US elastography. Radiology 282:726–733
Rinella ME, Elias MS, Smolak RR, Fu T, Borensztajn J, Green RM (2008) Mechanisms of hepatic steatosis in mice fed a lipogenic methionine choline-deficient diet. J Lipid Res 49:1068–1076
Hebbard L, George J (2011) Animal models of nonalcoholic fatty liver disease. Nat Rev Gastroenterol Hepatol 8:35–44
Kazemirad S, Bernard S, Hybois S, Tang A, Cloutier G (2016) Ultrasound shear wave viscoelastography: model-independent quantification of the complex shear modulus. IEEE Trans Ultrason Ferroelectr Freq Control 63:1399–1408
Bercoff J, Tanter M, Fink M (2004) Supersonic shear imaging: a new technique for soft tissue elasticity mapping. IEEE Trans Ultrason Ferroelectr Freq Control 51:396–409
Garcia D, Le Tarnec L, Muth S, Montagnon E, Porée J, Cloutier G (2013) Stolt’s f-k migration for plane wave ultrasound imaging. IEEE Trans Ultrason Ferroelectr Freq Control 60:1853–1867
Ophir J, Céspedes I, Ponnekanti H, Yazdi Y, Li X (1991) Elastography: a quantitative method for imaging the elasticity of biological tissues. Ultrason Imaging 13:111–134
Bernal M, Nenadic I, Urban MW, Greenleaf JF (2011) Material property estimation for tubes and arteries using ultrasound radiation force and analysis of propagating modes. J Acoust Soc Am 129:1344–1354
Destrempes F, Meunier J, Giroux MF, Soulez G, Cloutier G (2011) Segmentation of plaques in sequences of ultrasonic B-mode images of carotid arteries based on motion estimation and a Bayesian model. IEEE Trans Biomed Eng 58:2202–2211. https://doi.org/10.1109/TBME.2011.2127476
Destrempes F, Porée J, Cloutier G (2013) Estimation method of the homodyned K-distribution based on the mean intensity and two log-moments. SIAM J Imaging Sci 6:1499–1530
Destrempes F, Franceschini E, Yu FT, Cloutier G (2016) Unifying concepts of statistical and spectral quantitative ultrasound techniques. IEEE Trans Med Imaging 35:488–500
Bigelow TA, Labyed Y (2013) Attenuation compensation and estimation. In: Mamou J, Oelze ML (eds) Quantitative ultrasound in soft tissues. Springer, Dordrecht, pp 71–93
Montaldo G, Tanter M, Bercoff J, Benech N, Fink M (2009) Coherent plane-wave compounding for very high frame rate ultrasonography and transient elastography. IEEE Trans Ultrason Ferroelectr Freq Control 56:489–506
Breiman L (2001) Random forests. Mach Learn 45:5–32
He H, Garcia EA (2009) Learning from imbalanced data. IEEE Trans Knowl Data Eng 21:1263–1284
Efron B, Tibshirani R (1997) Improvements on cross-validation: the 632+ bootstrap method. J Am Stat Assoc 92:548–560
DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837–845
Davis J, Goadrich M (2006) The relationship between precision-recall and ROC curves. Proceedings of the 23rd international conference on machine learning. ACM, pp 233–240
Parker KJ, Doyley MM, Rubens DJ (2011) Imaging the elastic properties of tissue: the 20 year perspective. Phys Med Biol 56:R1–R29
Barr RG (2014) Elastography in clinical practice. Radiol Clin North Am 52:1145–1162
Iredale JP (2007) Models of liver fibrosis: exploring the dynamic nature of inflammation and repair in a solid organ. J Clin Invest 117:539–548
Zeng X, Xu C, He D et al (2015) Influence of hepatic inflammation on FibroScan findings in diagnosing fibrosis in patients with chronic hepatitis B. Ultrasound Med Biol 41:1538–1544
Dong DR, Hao MN, Li C et al (2015) Acoustic radiation force impulse elastography, FibroScan(®), Forns’ index and their combination in the assessment of liver fibrosis in patients with chronic hepatitis B, and the impact of inflammatory activity and steatosis on these diagnostic methods. Mol Med Rep 11:4174–4182
Chen J, Talwalkar JA, Yin M, Glaser KJ, Sanderson SO, Ehman RL (2011) Early detection of nonalcoholic steatohepatitis in patients with nonalcoholic fatty liver disease by using MR elastography. Radiology 259:749–756
Yoneda M, Suzuki K, Kato S et al (2010) Nonalcoholic fatty liver disease: US-based acoustic radiation force impulse elastography. Radiology 256:640–647
Mohareri O, Ruszkowski A, Lobo J et al (2014) Multi-parametric 3D quantitative ultrasound vibro-elastography imaging for detecting palpable prostate tumors. Med Image Comput Comput Assist Interv 17:561–568
Roy-Cardinal M, Destrempes F, Soulez G, Cloutier G (2016) Homodyned K-distribution parametric maps combined with elastograms for carotid artery plaque assessment. 2016 IEEE International Ultrasonics Symposium (IUS) pp 1–4
Wang QB, Zhu H, Liu HL, Zhang B (2012) Performance of magnetic resonance elastography and diffusion-weighted imaging for the staging of hepatic fibrosis: a meta-analysis. Hepatology 56:239–247
Acknowledgments
We thank Mr. Jamal Ait Ichou for his assistance in the literature review.
Funding
This study has received funding by Fonds de Recherche du Québec—Nature et Technologies (FRQNT) (PR-174387), Canadian Institutes of Health Research, Institute of Nutrition, Metabolism, and Diabetes (grant nos. 273738 and 301520), Quebec Bio-imaging Network (QBIN/RBIQ #5886), New Researcher Startup Grant from the Centre de Recherche du Centre Hospitalier de l’Université de Montréal, and by a Chercheur-Boursier Junior 2 from the Fonds de Recherche du Québec en Santé and Fondation de l’association des radiologistes du Québec (FRQS-ARQ #34939) to An Tang.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor if this publication is An Tang.
Conflict of interest
The authors declare that they have no conflict of interest.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Approval from the institutional animal care committee was obtained.
Ethical approval
Institutional review board approval was obtained.
Study subjects or cohorts overlap
The study cohorts have been previously reported in [24]. That prior article focused on a comparison between low-frequency versus high-frequency ultrasonographic shear wave elastography for the detection of steatohepatitis. In contrast, the present article investigates a machine learning model based on quantitative ultrasound parameters to improve classification of steatohepatitis compared to shear wave elastography alone.
Methodology
• Diagnostic study
• Experimental
• Performed at one institution
Electronic supplementary material
ESM 1
(DOCX 44 kb)
Rights and permissions
About this article
Cite this article
Tang, A., Destrempes, F., Kazemirad, S. et al. Quantitative ultrasound and machine learning for assessment of steatohepatitis in a rat model. Eur Radiol 29, 2175–2184 (2019). https://doi.org/10.1007/s00330-018-5915-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-018-5915-z