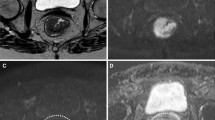
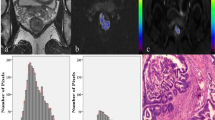
Abstract
Objective
To investigate potential relationships between diffusion kurtosis imaging (DKI)-derived parameters using whole-tumour volume histogram analysis and clinicopathological prognostic factors in patients with rectal adenocarcinoma.
Material and methods
79 consecutive patients who underwent MRI examination with rectal adenocarcinoma were retrospectively evaluated. Parameters D, K and conventional ADC were measured using whole-tumour volume histogram analysis. Student’s t-test or Mann-Whitney U-test, receiver operating characteristic curves and Spearman’s correlation were used for statistical analysis.
Results
Almost all the percentile metrics of K were correlated positively with nodal involvement, higher histological grades, the presence of lymphangiovascular invasion (LVI) and circumferential margin (CRM) (p<0.05), with the exception of between K10th, K90th and histological grades. In contrast, significant negative correlations were observed between 25th, 50th percentiles and mean values of ADC and D, as well as ADC10th, with tumour T stages (p< 0.05). Meanwhile, lower 75th and 90th percentiles of ADC and D values were also correlated inversely with nodal involvement (p< 0.05). Kmean showed a relatively higher area under the curve (AUC) and higher specificity than other percentiles for differentiation of lesions with nodal involvement.
Conclusion
DKI metrics with whole-tumour volume histogram analysis, especially K parameters, were associated with important prognostic factors of rectal cancer.
Key Points
• K correlated positively with some important prognostic factors of rectal cancer.
• K mean showed higher AUC and specificity for differentiation of nodal involvement.
• DKI metrics with whole-tumour volume histogram analysis depicted tumour heterogeneity.



Similar content being viewed by others
Abbreviations
- ADC:
-
Apparent diffusion coefficient
- CEA:
-
Carcinoembryonic antigen
- CRM:
-
Circumferential margin
- CRT:
-
Chemoradiotherapy
- D:
-
Diffusivity
- DKI:
-
Diffusion kurtosis imaging
- DWI:
-
Diffusion weighted imaging
- FOV:
-
Field of view
- ICC:
-
Interclass correlation coefficient
- K:
-
Kurtosis
- LVI:
-
Lymphangiovascular invasion
- MRI:
-
Magnetic resonance imaging
- ROC:
-
Receiver operating characteristic
- ROI:
-
Region of interest
- SS-EPI:
-
Single shot echo-planar imaging
References
Lee YC, Hsieh CC, Chuang JP (2013) Prognostic significance of partial tumor regression after preoperative chemoradiotherapy for rectal cancer: a meta-analysis. Dis Colon Rectum 56:1093–1101
Boras Z, Kondza G, Sisljagic V et al (2012) Prognostic factors of local recurrence and survival after curative rectal cancer surgery: a single institution experience. Coll Antropol 36:1355–1361
Cienfuegos JA, Rotellar F, Baixauli J et al (2015) Impact of perineural and lymphovascular invasion on oncological outcomes in rectal cancer treated with neoadjuvant chemoradiotherapy and surgery. Ann Surg Oncol 22:916–923
Lee JH, Jang HS, Kim JG et al (2012) Lymphovascular invasion is a significant prognosticator in rectal cancer patients who receive preoperative chemoradiotherapy followed by total mesorectal excision. Ann Surg Oncol 19:1213–1221
Peng J, Sheng W, Huang D et al (2011) Perineural invasion in pT3N0 rectal cancer: the incidence and its prognostic effect. Cancer 117:1415–1421
Madbouly KM, Abbas KS, Hussein AM (2014) Metastatic lymph node ratio in stage III rectal carcinoma is a valuable prognostic factor even with less than 12 lymph nodes retrieved: a prospective study. Am J Surg 207:824–831
Zhang XM, Zhang HL, Yu D et al (2008) 3-T MRI of rectal carcinoma: preoperative diagnosis, staging, and planning of sphincter-sparing surgery. AJR Am J Roentgenol 190:1271–1278
Blazic IM, Lilic GB, Gajic MM (2017) Quantitative Assessment of Rectal Cancer Response to Neoadjuvant Combined Chemotherapy and Radiation Therapy: Comparison of Three Methods of Positioning Region of Interest for ADC Measurements at Diffusion-weighted MR Imaging. Radiology 282:418–428
Choi MH, Oh SN, Rha SE et al (2016) Diffusion-weighted imaging: Apparent diffusion coefficient histogram analysis for detecting pathologic complete response to chemoradiotherapy in locally advanced rectal cancer. J Magn Reson Imaging 44:212–220
Cai PQ, Wu YP, An X et al (2014) Simple measurements on diffusion-weighted MR imaging for assessment of complete response to neoadjuvant chemoradiotherapy in locally advanced rectal cancer. Eur Radiol 24:2962–2970
Curvo-Semedo L, Lambregts DM, Maas M et al (2012) Diffusion-weighted MRI in rectal cancer: apparent diffusion coefficient as a potential noninvasive marker of tumor aggressiveness. J Magn Reson Imaging 35:1365–1371
Akashi M, Nakahusa Y, Yakabe T et al (2014) Assessment of aggressiveness of rectal cancer using 3-T MRI: correlation between the apparent diffusion coefficient as a potential imaging biomarker and histologic prognostic factors. Acta Radiol 55:524–531
Tong T, Yao Z, Xu L et al (2014) Extramural depth of tumor invasion at thin-section MR in rectal cancer: associating with prognostic factors and ADC value. J Magn Reson Imaging 40:738–744
Sun Y, Tong T, Cai S et al (2014) Apparent Diffusion Coefficient (ADC) value: a potential imaging biomarker that reflects the biological features of rectal cancer. PloS one 9:e109371
Lambregts DM, Beets GL, Maas M et al (2011) Tumor ADC measurements in rectal cancer: effect of ROI methods on ADC values and interobserver variability. Eur Radiol 21:2567–2574
Rosenkrantz AB, Padhani AR, Chenevert TL et al (2015) Body diffusion kurtosis imaging: Basic principles, applications, and considerations for clinical practice. J Magn Reson Imaging 42:1190–1202
Jensen JH, Helpern JA, Ramani A et al (2005) Diffusional kurtosis imaging: the quantification of non-gaussian water diffusion by means of magnetic resonance imaging. Magn Reson Med 53:1432–1440
Jensen JH, Helpern JA (2010) MRI quantification of non-Gaussian water diffusion by kurtosis analysis. NMR Biomedicine 23:698–710
Pang H, Ren Y, Dang X et al (2016) Diffusional kurtosis imaging for differentiating between high-grade glioma and primary central nervous system lymphoma. J Magn Reson Imaging 44:30–40
Bai Y, Lin Y, Tian J et al (2016) Grading of Gliomas by Using Monoexponential, Biexponential, and Stretched Exponential Diffusion-weighted MR Imaging and Diffusion Kurtosis MR Imaging. Radiology 278:496–504
Goshima S, Kanematsu M, Noda Y et al (2015) Diffusion kurtosis imaging to assess response to treatment in hypervascular hepatocellular carcinoma. AJR Am J Roentgenol 204:W543–W549
Wang Q, Li H, Yan X et al (2015) Histogram analysis of diffusion kurtosis magnetic resonance imaging in differentiation of pathologic Gleason grade of prostate cancer. Urol Oncol 33(337):e15–e24
Nogueira L, Brandao S, Matos E et al (2014) Application of the diffusion kurtosis model for the study of breast lesions. Eur Radiol 24:1197–1203
Yu J, Xu Q, Song JC, et al (2016) The value of diffusion kurtosis magnetic resonance imaging for assessing treatment response of neoadjuvant chemoradiotherapy in locally advanced rectal cancer. Eur Radiol 1–10
Yu J, Huang DY, Li Y et al (2016) Correlation of standard diffusion-weighted imaging and diffusion kurtosis imaging with distant metastases of rectal carcinoma. J Magn Reson Imaging 44:221–229
Zhu L, Pan Z, Ma Q, et al (2016) Diffusion Kurtosis Imaging Study of Rectal Adenocarcinoma Associated with Histopathologic Prognostic Factors: Preliminary Findings. Radiology 160094.
Edge SB, Compton CC (2010) The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Annals of surgical oncology 17:1471–1474
DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837–845
Li HM, Zhao SH, Qiang JW, et al (2017) Diffusion kurtosis imaging for differentiating borderline from malignant epithelial ovarian tumors: A correlation with Ki-67 expression. J Magn Reson Imaging
Jambor I, Merisaari H, Taimen P et al (2015) Evaluation of different mathematical models for diffusion-weighted imaging of normal prostate and prostate cancer using high b-values: a repeatability study. Magn Reson Med 73:1988–1998
Sun K, Chen X, Chai W et al (2015) Breast Cancer: Diffusion Kurtosis MR Imaging-Diagnostic Accuracy and Correlation with Clinical-Pathologic Factors. Radiology 277:46–55
Van Cauter S, Veraart J, Sijbers J et al (2012) Gliomas: diffusion kurtosis MR imaging in grading. Radiology 263:492–501
Al-Sukhni E, Milot L, Fruitman M et al (2012) Diagnostic accuracy of MRI for assessment of T category, lymph node metastases, and circumferential resection margin involvement in patients with rectal cancer: a systematic review and meta-analysis. Ann Surg Oncol 19:2212–2223
Attenberger UI, Pilz LR, Morelli JN et al (2014) Multi-parametric MRI of rectal cancer - do quantitative functional MR measurements correlate with radiologic and pathologic tumor stages? Eur J Radiol 83:1036–1043
Funding
This work was supported by the fund of Science and Technology Project of Shanxi Province (No. 20150313007-5).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Xiaotang Yang.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• diagnostic or prognostic study
• performed at one institution
Rights and permissions
About this article
Cite this article
Cui, Y., Yang, X., Du, X. et al. Whole-tumour diffusion kurtosis MR imaging histogram analysis of rectal adenocarcinoma: Correlation with clinical pathologic prognostic factors. Eur Radiol 28, 1485–1494 (2018). https://doi.org/10.1007/s00330-017-5094-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-5094-3