Abstract
Purpose
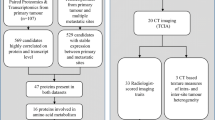
To evaluate the associations between clinical outcomes and radiomics-derived inter-site spatial heterogeneity metrics across multiple metastatic lesions on CT in patients with high-grade serous ovarian cancer (HGSOC).
Methods
IRB-approved retrospective study of 38 HGSOC patients. All sites of suspected HGSOC involvement on preoperative CT were manually segmented. Gray-level correlation matrix-based textures were computed from each tumour site, and grouped into five clusters using a Gaussian Mixture Model. Pairwise inter-site similarities were computed, generating an inter-site similarity matrix (ISM). Inter-site texture heterogeneity metrics were computed from the ISM and compared to clinical outcomes.
Results
Of the 12 inter-site texture heterogeneity metrics evaluated, those capturing the differences in texture similarities across sites were associated with shorter overall survival (inter-site similarity entropy, similarity level cluster shade, and inter-site similarity level cluster prominence; p ≤ 0.05) and incomplete surgical resection (similarity level cluster shade, inter-site similarity level cluster prominence and inter-site cluster variance; p ≤ 0.05). Neither the total number of disease sites per patient nor the overall tumour volume per patient was associated with overall survival. Amplification of 19q12 involving cyclin E1 gene (CCNE1) predominantly occurred in patients with more heterogeneous inter-site textures.
Conclusion
Quantitative metrics non-invasively capturing spatial inter-site heterogeneity may predict outcomes in patients with HGSOC.
Key Points
• Calculating inter-site texture-based heterogeneity metrics was feasible
• Metrics capturing texture similarities across HGSOC sites were associated with overall survival
• Heterogeneity metrics were also associated with incomplete surgical resection of HGSOC.



Similar content being viewed by others
Abbreviations
- CCNE1:
-
Cyclin E1 gene
- CLOVAR:
-
Classification of Ovarian Cancer transcriptomic profiles
- CSR:
-
Complete surgical resection
- CT:
-
Computed tomography
- EM:
-
Expectation-Maximization algorithm
- FIGO:
-
The International Federation of Gynecology and Obstetrics
- GLCM:
-
Gray-level correlation matrix
- GMM:
-
Gaussian Mixture Model
- HGSOC:
-
High-grade serous ovarian cancer
- IQR:
-
Interquartile range
- ISM:
-
Inter-site similarity matrix
- KM:
-
Kaplan-Meier
- ROI:
-
Region of interest
- SCP:
-
Inter-site similarity level cluster prominence
- SCS:
-
Similarity level cluster shade
- SE:
-
Inter-site similarity entropy
- SLV:
-
Inter-site similarity level variance
- TCGA:
-
The Cancer Genome Atlas
- VOI:
-
Volume of interest
References
Engel J, Eckel R, Schubert-Fritschle G, Kerr J, Kuhn W, Diebold J et al (2002) Moderate progress for ovarian cancer in the last 20 years: prolongation of survival, but no improvement in the cure rate. Eur J Cancer 38:2435–2445
Hyman DM, Zhou Q, Iasonos A, Grisham RN, Arnold AG, Phillips MF et al (2012) Improved survival for BRCA2-associated serous ovarian cancer compared with both BRCA-negative and BRCA1-associated serous ovarian cancer. Cancer 118:3703–3709
Tothill RW, Tinker AV, George J, Brown R, Fox SB, Lade S et al (2008) Novel molecular subtypes of serous and endometrioid ovarian cancer linked to clinical outcome. Clin Cancer Res 14:5198–5208
Verhaak RG, Tamayo P, Yang JY, Hubbard D, Zhang H, Creighton CJ et al (2013) Prognostically relevant gene signatures of high-grade serous ovarian carcinoma. J Clin Invest 123:517–525
Soslow RA (2008) Histologic subtypes of ovarian carcinoma: an overview. Int J Gynecol Pathol 27:161–174
Khalique L, Ayhan A, Weale ME, Jacobs IJ, Ramus SJ, Gayther SA (2007) Genetic intra-tumour heterogeneity in epithelial ovarian cancer and its implications for molecular diagnosis of tumours. J Pathol 211:286–295
Kumar V, Gu Y, Basu S, Berglund A, Eschrich SA, Schabath MB et al (2012) Radiomics: the process and the challenges. Magn Reson Imaging 30:1234–1248
Haralick RM, Shanmugam K, Dinstein IH (1973) Textural features for image classification. IEEE Trans Syst Man Cyber SMC-3:610–621
Aerts HJ, Velazquez ER, Leijenaar RT, Parmar C, Grossmann P, Carvalho S et al (2014) Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach. Nat Commun 5:4006
Segal E, Sirlin CB, Ooi C, Adler AS, Gollub J, Chen X et al (2007) Decoding global gene expression programs in liver cancer by noninvasive imaging. Nat Biotechnol 25:675–680
Vargas HA, Micco M, Hong SI, Goldman DA, Dao F, Weigelt B et al (2015) Association between morphologic CT imaging traits and prognostically relevant gene signatures in women with high-grade serous ovarian cancer: a hypothesis-generating study. Radiology 274:742–751
Veeraraghavan H, Schrater P, Papanikolopoulos NP (2006) Adaptive templates for feature matching. Proc IEEE Int Conf Robot Autom 3393–3398
Tibshirani R (1996) Regression shrinkage and selection via the lasso. J R Stat Soc Ser B Methodol 58:267–288
Ture MTF, Kurt I (2009) Using Kaplan-Meier analysis together with decision tree methods in determining recurrence-free survival of breast cancer patients. Expert Syst Appl 36:2017–2026
Etemadmoghadam D, deFazio A, Beroukhim R, Mermel C, George J, Getz G et al (2009) Integrated genome-wide DNA copy number and expression analysis identifies distinct mechanisms of primary chemoresistance in ovarian carcinomas. Clin Cancer Res Off J Am Assoc Cancer Res 15:1417–1427
Patch AM, Christie EL, Etemadmoghadam D, Garsed DW, George J, Fereday S et al (2015) Whole-genome characterization of chemoresistant ovarian cancer. Nature 521:489–494
Gerlinger M, Rowan AJ, Horswell S, Larkin J, Endesfelder D, Gronroos E et al (2012) Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med 366:883–892
Juric D, Castel P, Griffith M, Griffith OL, Won HH, Ellis H et al (2015) Convergent loss of PTEN leads to clinical resistance to a PI(3)Kalpha inhibitor. Nature 518:240–244
Yu HA, Arcila ME, Rekhtman N, Sima CS, Zakowski MF, Pao W et al (2013) Analysis of tumor specimens at the time of acquired resistance to EGFR-TKI therapy in 155 patients with EGFR-mutant lung cancers. Clin Cancer Res 19:2240–2247
Schwarz RF, Ng CK, Cooke SL, Newman S, Temple J, Piskorz AM et al (2015) Spatial and temporal heterogeneity in high-grade serous ovarian cancer: a phylogenetic analysis. PLoS Med 12:e1001789
Mittempergher L (2016) Genomic characterization of high-grade serous ovarian cancer: dissecting its molecular heterogeneity as a road towards effective therapeutic strategies. Curr Oncol Rep 18:44
Paracchini L, Mannarino L, Craparotta I, Romualdi C, Fruscio R, Grassi T et al (2016) Regional and temporal heterogeneity of epithelial ovarian cancer tumor biopsies: implications for therapeutic strategies. Oncotarget. doi:10.18632/oncotarget.10505
Rizvi NA, Hellmann MD, Snyder A, Kvistborg P, Makarov V, Havel JJ et al (2015) Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science (New York, NY) 348:124–128
Rosenberg JE, Hoffman-Censits J, Powles T, van der Heijden MS, Balar AV, Necchi A et al (2016) Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: a single-arm, multicentre, phase 2 trial. Lancet (London, England) 387:1909–1920
Hugo W, Zaretsky JM, Sun L, Song C, Moreno BH, Hu-Lieskovan S et al (2016) Genomic and transcriptomic features of response to anti-PD-1 therapy in metastatic melanoma. Cell 165:35–44
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Evis Sala.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Funding
This study has received funding by NIH grant P30 CA008748.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Ethical approval
Institutional Review Board approval was obtained.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Study subjects or cohorts overlap
All of the study subjects or cohorts have been previously reported in a prior study that investigated the associations between qualitative CT imaging features, CLOVAR gene signatures and survival in women with HGSOC (paper also uploaded through editorial manager).
Vargas HA, Miccò M, Hong SI, Goldman DA, Dao F, Weigelt B, Soslow RA, Hricak H, Levine DA, Sala E. Association between Morphologic CT Imaging Traits and Prognostically Relevant Gene Signatures in Women with High-Grade Serous Ovarian Cancer: A Hypothesis-generating Study. Radiology. 2015 Mar;274(3):742-51. doi: 10.1148/radiol.14141477 Epub 2014 Nov 10.
Methodology
• retrospective
• cross sectional study
• performed at one institution
Additional information
Hebert Alberto Vargas and Harini Veeraraghavan contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
(DOCX 18 kb)
ESM 1
(DOCX 20 kb)
Rights and permissions
About this article
Cite this article
Vargas, H.A., Veeraraghavan, H., Micco, M. et al. A novel representation of inter-site tumour heterogeneity from pre-treatment computed tomography textures classifies ovarian cancers by clinical outcome. Eur Radiol 27, 3991–4001 (2017). https://doi.org/10.1007/s00330-017-4779-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-4779-y