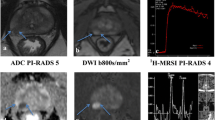
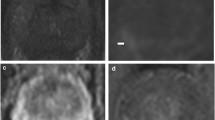
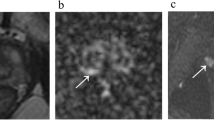
Abstract
Objectives
The aim of this study was to assess the diagnostic performance of pre-treatment 3-Tesla (3T) multiparametric magnetic resonance imaging (mpMRI) for predicting Gleason score (GS) downgrading after radical prostatectomy (RP) in patients with GS 3 + 4 prostate cancer (PCa) on biopsy.
Methods
We retrospectively reviewed 304 patients with biopsy-proven GS 3 + 4 PCa who underwent mpMRI before RP. On T2-weighted imaging and three mpMRI combinations (T2-weighted imaging + diffusion-weighted imaging [DWI], T2-weighted imaging + dynamic contrast-enhanced-MRI [DCE-MRI], and T2-weighted imaging + DWI + DCE-MRI), two radiologists (R1/R2) scored the presence of a dominant tumour using a 5-point Likert scale (1 = definitely absent to 5 = definitely present). Diagnostic performance in identifying downgrading was evaluated via areas under the curves (AUCs). Predictive accuracies of multivariate models were calculated.
Results
In predicting downgrading, T2-weighted imaging + DWI (AUC = 0.89/0.85 for R1/R2) performed significantly better than T2-weighted imaging alone (AUC = 0.72/0.73; p < 0.001/p = 0.02 for R1/R2), while T2-weighted imaging + DWI + DCE-MRI (AUC = 0.89/0.84 for R1/R2) performed no better than T2-weighted imaging + DWI (p = 0.48/p > 0.99 for R1/R2). On multivariate analysis, the clinical + mpMRI model incorporating T2-weighted imaging + DWI (AUC = 0.92/0.88 for R1/R2) predicted downgrading significantly better than the clinical model (AUC = 0.73; p < 0.001 for R1/R2).
Conclusion
mpMRI improves the ability to identify a subgroup of patients with Gleason 3 + 4 PCa on biopsy who are candidates for active surveillance. DCE-MRI (compared to T2 + DWI) offered no additional benefit to the prediction of downgrading.
Key Points
• Diagnostic performance of T2-weighted-imaging + DWI was better than T2-weighted-imaging alone.
• Diagnostic performance of T2-weighted-imaging + DWI was similar to T2-weighted-imaging + DWI + DCE-MRI.
• Combining clinical and T2-weighted-imaging + DWI features best predicted GS downgrading.
• mpMRI might prevent overtreatment by increasing eligibility for PCa active surveillance.


Similar content being viewed by others
Abbreviations
- mpMRI:
-
Multiparametric magnetic resonance imaging
- GS:
-
Gleason score
- RP:
-
Radical prostatectomy
- PCa:
-
Prostate cancer
- PSA:
-
Prostate-specific antigen
- DWI:
-
Diffusion-weighted imaging
- DCE:
-
Dynamic contrast-enhanced
- AUC:
-
Area under the curve
- ADC:
-
Apparent diffusion coefficient
- OR:
-
Odds ratio
- CI:
-
Confidence interval
References
Fridlyand J, Simon RM, Walrath JC et al (2013) Considerations for the successful co-development of targeted cancer therapies and companion diagnostics. Nat Rev Drug Discov 12:743–755
Klotz L (2013) Prostate cancer overdiagnosis and overtreatment. Curr Opin Endocrinol Diabetes Obes 20:204–209
Eggener SE, Mueller A, Berglund RK et al (2013) A multi-institutional evaluation of active surveillance for low risk prostate cancer. J Urol 189:S19–S25
Tosoian JJ, Trock BJ, Landis P et al (2011) Active surveillance program for prostate cancer: an update of the Johns Hopkins experience. J Clin Oncol 29:2185–2190
Lawrentschuk N, Klotz L (2011) Active surveillance for low-risk prostate cancer: an update. Nat Rev Urol 8:312–320
Bul M, van den Bergh RC, Zhu X et al (2012) Outcomes of initially expectantly managed patients with low or intermediate risk screen-detected localized prostate cancer. BJU Int 110:1672–1677
Cooperberg MR, Cowan JE, Hilton JF et al (2011) Outcomes of active surveillance for men with intermediate-risk prostate cancer. J Clin Oncol 29:228–234
Epstein JI, Feng Z, Trock BJ, Pierorazio PM (2012) Upgrading and downgrading of prostate cancer from biopsy to radical prostatectomy: incidence and predictive factors using the modified Gleason grading system and factoring in tertiary grades. Eur Urol 61:1019–1024
Turkbey B, Mani H, Aras O et al (2013) Prostate cancer: can multiparametric MR imaging help identify patients who are candidates for active surveillance? Radiology 268:144–152
Mullins JK, Bonekamp D, Landis P et al (2013) Multiparametric magnetic resonance imaging findings in men with low-risk prostate cancer followed using active surveillance. BJU Int 111:1037–1045
Margel D, Yap SA, Lawrentschuk N et al (2012) Impact of multiparametric endorectal coil prostate magnetic resonance imaging on disease reclassification among active surveillance candidates: a prospective cohort study. J Urol 187:1247–1252
Donati OF, Jung SI, Vargas HA et al (2013) Multiparametric prostate MR imaging with T2-weighted, diffusion-weighted, and dynamic contrast-enhanced sequences: are all pulse sequences necessary to detect locally recurrent prostate cancer after radiation therapy? Radiology 268:440–450
Hambrock T, Somford DM, Huisman HJ et al (2011) Relationship between apparent diffusion coefficients at 3.0-T MR imaging and Gleason grade in peripheral zone prostate cancer. Radiology 259:453–461
Verma S, Rajesh A, Morales H et al (2011) Assessment of aggressiveness of prostate cancer: correlation of apparent diffusion coefficient with histologic grade after radical prostatectomy. AJR Am J Roentgenol 196:374–381
Borofsky MS, Rosenkrantz AB, Abraham N, Jain R, Taneja SS (2013) Does suspicion of prostate cancer on integrated T2 and diffusion-weighted MRI predict more adverse pathology on radical prostatectomy? Urology 81:1279–1283
Lee DH, Koo KC, Lee SH et al (2013) Low-risk prostate cancer patients without visible tumor (T1c) on multiparametric MRI could qualify for active surveillance candidate even if they did not meet inclusion criteria of active surveillance protocol. Jpn J Clin Oncol 43:553–558
Vargas HA, Akin O, Afaq A et al (2012) Magnetic resonance imaging for predicting prostate biopsy findings in patients considered for active surveillance of clinically low risk prostate cancer. J Urol 188:1732–1738
Bjurlin MA, Carter HB, Schellhammer P et al (2013) Optimization of initial prostate biopsy in clinical practice: sampling, labeling and specimen processing. J Urol 189:2039–2046
Cohen J (1968) Weighted kappa: nominal scale agreement with provision for scaled disagreement or partial credit. Psychol Bull 70:213–220
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Lesaffre E, Willems JL, Albert A (1989) Estimation of error rate in multiple group logistic discrimination. The approximate leaving-one-out method. communications in statistics. Theory Methods 18:2989–3007
Whitson JM, Porten SP, Cowan JE, Simko JP, Cooperberg MR, Carroll PR (2013) Factors associated with downgrading in patients with high grade prostate cancer. Urol Oncol 31:442–447
Donohue JF, Bianco FJ Jr, Kuroiwa K et al (2006) Poorly differentiated prostate cancer treated with radical prostatectomy: long-term outcome and incidence of pathological downgrading. J Urol 176:991–995
Moussa AS, Li J, Soriano M, Klein EA, Dong F, Jones JS (2009) Prostate biopsy clinical and pathological variables that predict significant grading changes in patients with intermediate and high grade prostate cancer. BJU Int 103:43–48
Freedland SJ, Kane CJ, Amling CL et al (2007) Upgrading and downgrading of prostate needle biopsy specimens: risk factors and clinical implications. Urology 69:495–499
Vargas HA, Akin O, Shukla-Dave A et al (2012) Performance characteristics of MR imaging in the evaluation of clinically low-risk prostate cancer: a prospective study. Radiology 265:478–487
Van der Kwast TH, Roobol MJ (2013) Defining the threshold for significant versus insignificant prostate cancer. Nat Rev Urol 10:473–482
Ross HM, Kryvenko ON, Cowan JE, Simko JP, Wheeler TM, Epstein JI (2012) Do adenocarcinomas of the prostate with Gleason score (GS) </=6 have the potential to metastasize to lymph nodes? Am J Surg Pathol 36:1346–1352
Tosoian JJ, Johnbull E, Trock BJ et al (2013) Pathologic outcomes in men with low-risk and very low-risk prostate cancer: implications on the practice of active surveillance. J Urol 190:1218–1222
Adam M, Hannah A, Budäus L et al (2014) A tertiary gleason pattern in the prostatectomy specimen and its association to adverse outcome after radical prostatectomy; results of a large contemporary prostatectomy series. J Urol
Barentsz JO, Richenberg J, Clements R et al (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22:746–757
Rosenkrantz AB, Lim RP, Haghighi M, Somberg MB, Babb JS, Taneja SS (2013) Comparison of interreader reproducibility of the prostate imaging reporting and data system and likert scales for evaluation of multiparametric prostate MRI. AJR Am J Roentgenol 201:W612–W618
Acknowledgments
The guarantor of this study is Hebert Alberto Vargas. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article. The authors state that this work has not received any funding. Two of the authors (Junting Zheng and Chaya S. Moskowitz) have significant statistical expertise. Institutional Review Board approval was obtained (WA0297-13). Written informed consent was waived by the Institutional Review Board. Methodology: retrospective, diagnostic or prognostic study, performed at one institution.
This work was supported by the Sidney Kimmel Center for Prostate and Urologic Cancers. We are grateful to Ada Muellner, M.S., for editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Table 1
(DOCX 23 kb)
Rights and permissions
About this article
Cite this article
Gondo, T., Hricak, H., Sala, E. et al. Multiparametric 3T MRI for the prediction of pathological downgrading after radical prostatectomy in patients with biopsy-proven Gleason score 3 + 4 prostate cancer. Eur Radiol 24, 3161–3170 (2014). https://doi.org/10.1007/s00330-014-3367-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-014-3367-7