Abstract
Objective
This study investigated the correlation of oestrogen receptor (ER) and human epidermal growth factor receptor 2 (HER2) status with the probability of malignancy (POM) of mammographic calcifications in ductal carcinoma in situ (DCIS).
Methods
A total of 101 women (age range, 27–83 years) with pure DCIS that presented as mammographic calcifications were included. Three radiologists independently reviewed mammograms according to the BI-RADS lexicon and provided 100-point POM scores and a BI-RADS category. ER, HER2 and breast cancer subtypes were determined using immunohistochemistry (IHC) and fluorescence in situ hybridisation. Pairwise correlations between POM and IHC biomarker scores were calculated, and mammographic features were compared between breast cancer subtypes.
Results
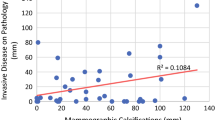
HER2 level positively correlated with the POM score (P < 0.0001) and BI-RADS category (P < 0.0001), and ER level inversely correlated with the POM score (P < 0.013) and BI-RADS category (P < 0.010). Fine linear branching (P = 0.004) and segmental (P = 0.014) calcifications were significantly associated with HER2-positive cancers, and clustered calcifications were more frequently observed in ER-positive cancers (P = 0.014).
Conclusion
HER2 status in DCIS correlated positively with the POM of mammographic calcifications, as determined by radiologists on the basis of the BI-RADS lexicon.
Key Points
• Prediction of malignancy on mammographic ductal carcinoma in situ is difficult.
• HER2 level correlated positively with the probability of malignancy assigned by radiologists.
• ER level correlated inversely with the probability of malignancy assigned by radiologists.
• HER2-positive DCIS more frequently exhibited fine linear branching or segmental calcifications.
• ER-positive DCIS more frequently exhibited clustered calcifications.


Similar content being viewed by others
References
Kerlikowske K (2010) Epidemiology of ductal carcinoma in situ. J Natl Cancer Inst Monogr 41:139–141
Allred DC (2010) Ductal carcinoma in situ: terminology, classification, and natural history. J Natl Cancer Inst Monogr 41:134–138
Burstein HK, Polyak K, Wong JS et al (2004) Ductal carcinoma in situ of the breast. N Engl J Med 350:1430–1441
Erbas B, Provenzano E, Armes J et al (2006) The natural history of ductal carcinoma in situ of the breast: a review. Breast Cancer Res Treat 97:135–144
D’Orsi CJ (2010) Imaging for the diagnosis and management of ductal carcinoma in situ. J Natl Cancer Inst Monogr 41:214–217
Holmes P, Lloyd J, Chervoneva I et al (2011) Prognostic markers and long-term outcomes in ductal carcinoma in situ of the breast treated with excision alone. Cancer 117:3650–3657
Han K, Nofech-Mozes S, Narod S et al (2012) Expression of HER2neu in ductal carcinoma in situ is associated with local recurrence. Clin Oncol (R Coll Radiol) 24:183–189
Shao W, Brown M (2012) Advances in estrogen receptor biology: prospects for improvements in targeted breast cancer therapy. Breast Cancer Res 6:39–52
Provenzano E, Hopper JL, Biles GG et al (2003) Biological markers that predict clinical recurrence in ductal carcinoma in situ of the breast. Eur J Cancer 39:622–630
Rubin I, Yarden Y (2001) The basic biology of HER2. Ann Oncol 12:S3–S8
Leonard GD, Swain SM (2004) Ductal carcinoma in situ, complexities and challenges. J Natl Cancer Inst 96:906–920
Polyak K (2010) Molecular markers for the diagnosis and management of ductal carcinoma in situ. J Natl Cancer Inst Monogr 41:210–213
Perou CM, Sorlie T, Eisen MB et al (2000) Molecular portraits of human breast tumours. Nature 406:747–752
Weigel S, Decker T, Korsching E, Hungermann D, Böcker W, Heindel W (2010) Calcifications in digital mammographic screening: improvement of early detection of invasive breast cancers? Radiology 255:738–745
Holland R, Schuurmans Stekhoven JH, Hendriks JHCL, Vebeek ALM, Mravunac M (1990) Extent, distribution, and mammographic/histologic correlations of breast ductal carcinoma in situ. Lancet 335:519–522
Dershaw DD, Giess CS, McCormick B et al (1997) Patterns of mammographically detected calcifications after breast-conserving therapy associated with tumor recurrence. Cancer 79:1355–1361
Antonio AL, Crespi CM (2010) Predictors of interobserver agreement in breast imaging using the Breast Imaging Reporting and Data System. Breast Cancer Res Treat 120:539–546
Badra FA, Karamouzis MV, Ravazoula P et al (2006) Non-palpable breast carcinomas: correlation of mammographically detected malignant-appearing microcalcifications and epidermal growth factor receptor (EGFR) family expression. Cancer Lett 244:34–41
Karamouzis MV, Likaki-Karatza E, Ravazoula P et al (2002) Non-palpable breast carcinomas: correlation of mammographically detected malignant-appearing microcalcifications and molecular prognostic factors. Int J Cancer 102:86–90
Gajdos C, Tartter PI, Bleiweiss IJ et al (2002) Mammographic appearance of nonpalpable breast cancer reflects pathologic characteristics. Ann Surg 235:246–251
Seo BK, Pisano ED, Kuzimak CM et al (2006) Correlation of HER-2/neu overexpression with mammography and age distribution in primary breast carcinomas. Acad Radiol 13:1211–1218
Evans AJ, Pinder SE, Ellis IO et al (1994) Correlations between the mammographic features of ductal carcinoma in situ (DCIS) and C-erbB-2 oncogene expression. Clin Radiol 49:559–562
Wang Y, Ikeda DM, Narasimhan B et al (2008) Estrogen receptor-negative invasive breast cancer: imaging features of tumors with and without human epidermal growth factor receptor type 2 overexpression. Radiology 246:367–375
American College of Radiology (2003) Breast Imaging Reporting and Data System (BI-RADS) (4th edition): mammography. American College of Radiology, Reston
Ohlschlegel C, Zahel K, Kradolfer D et al (2011) HER2 genetic heterogeneity in breast carcinoma. J Clin Pathol 64:1112–1116
Wolff AC, Hammond ME, Schwartz JN et al (2007) American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol 25:118–145
Allred DC, Wu Y, Mao S et al (2008) Ductal carcinoma in situ and emergence of diversity during breast cancer evolution. Clin Cancer Res 14:370–378
Hannemann J, Velds A, Halfwerk JB, Kreike B, Peterse JL, van de Vijver MJ (2006) Classification of ductal carcinoma in situ by gene expression profiling. Breast Cancer Res 8:R61
Bent CK, Bassett LW, D’Orsi CJ, Sayre JW (2010) The positive predictive value of BI-RADS microcalcification descriptors and final assessment categories. AJR Am J Roentgenol 194:1378–1383
Wiechmann L, Kuerer HM (2008) The molecular journey from ductal carcinoma in situ to invasive breast cancer. Cancer 112:2130–2142
Tsikitis VL, Chung MA (2006) Biology of ductal carcinoma in situ classification based on biologic potential. Am J Clin Oncol 29:305–310
Evans A, Pinder S, Wilson R et al (1994) Ductal carcinoma in situ of the breast: correlation between mammographic and pathologic findings. AJR Am J Roentgenol 162:1307–1311
Dinkel HP, Gassel AM, Tschammler A (2000) Is the appearance of microcalcifications on mammography useful in predicting histological grade of malignancy in ductal cancer in situ? Br J Radiol 73:938–944
McCaskill-Stevens W (2010) National Institutes of Health state-of-the-science conference on the management and diagnosis of ductal carcinoma in situ: a call to action. J Natl Cancer Inst Monogr 2010:111–112
Wang SY, Shamliyan T, Virnig BA, Kane R (2011) Tumor characteristics as predictors of local recurrence after treatment of ductal carcinoma in situ: a meta-analysis. Breast Cancer Res Treat 127:1–14
Kerlikowske K, Molinaro AM, Gauthier ML et al (2010) Biomarker expression and risk of subsequent tumors after initial ductal carcinoma in situ diagnosis. J Natl Cancer Inst 102:627–637
Rakovitch E, Nofech-Mozes S, Hanna W et al (2012) HER2/neu and Ki67 expression predict non-invasive recurrence following breast-conserving therapy for ductal carcinoma in situ. Br J Cancer 106:1160–1165
Allred DC, Anderson SJ, Paik S et al (2012) Adjuvant tamoxifen reduces subsequent breast cancer in women with estrogen receptor-positive ductal carcinoma in situ: a study based on NSABP protocol B-24. J Clin Oncol 30:1268–1273
Kane RL, Virnig BA, Shamliyan T et al (2010) The impact of surgery, radiation, and systemic treatment on outcomes in patients with ductal carcinoma in situ. J Natl Cancer Inst Monogr 41:130–133
Sharma A, Koldovsky U, Xu S et al (2012) HER-2 pulsed dendritic cell vaccine can eliminate HER-2 expression and impact ductal carcinoma in situ. Cancer 118:4354–4362
Tamimi RM, Baer HJ, Marotti J et al (2008) Comparison of molecular phenotypes of ductal carcinoma in situ and invasive breast cancer. Breast Cancer Res 10:R67–R76
Kojima Y, Tsunoda H, Honda S et al (2011) Radiographic features for triple negative ductal carcinoma in situ of the breast. Breast Cancer 18:213–220
Yang WT, Dryden M, Broglio K et al (2008) Mammographic features of triple receptor-negative primary breast cancers in young premenopausal women. Breast Cancer Res Treat 111:405–410
Acknowledgments
This study was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2012R1A1A2009414 and 2012R1A2A1A01010846), by the Health and Medical Technology R&D Programs through the Korea Health Industry Development Institute supervised by the Ministry for Health, Welfare and Family Affairs (A110433) and by a grant from the Seoul National University Hospital (SNUH) Research Fund (03-2012-0400). We thank the Medical Research Collaborating Center (MRCC) of SNUH for the statistical analysis. The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bae, M.S., Moon, W.K., Chang, J.M. et al. Mammographic features of calcifications in DCIS: correlation with oestrogen receptor and human epidermal growth factor receptor 2 status. Eur Radiol 23, 2072–2078 (2013). https://doi.org/10.1007/s00330-013-2827-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-013-2827-9