Abstract
Objectives
To diagnose Parkinson disease (PD) at the individual level using pattern recognition of brain susceptibility-weighted imaging (SWI).
Methods
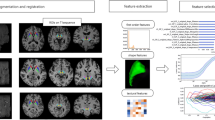
We analysed brain SWI in 36 consecutive patients with Parkinsonism suggestive of PD who had (1) SWI at 3 T, (2) brain 123I-ioflupane SPECT and (3) extensive neurological testing including follow-up (16 PD, 67.4 ± 6.2 years, 11 female; 20 OTHER, a heterogeneous group of atypical Parkinsonism syndromes 65.2 ± 12.5 years, 6 female). Analysis included group-level comparison of SWI values and individual-level support vector machine (SVM) analysis.
Results
At the group level, simple visual analysis yielded no differences between groups. However, the group-level analyses demonstrated increased SWI in the bilateral thalamus and left substantia nigra in PD patients versus other Parkinsonism. The inverse comparison yielded no supra-threshold clusters. At the individual level, SVM correctly classified PD patients with an accuracy above 86 %.
Conclusions
SVM pattern recognition of SWI data provides accurate discrimination of PD among patients with various forms of Parkinsonism at an individual level, despite the absence of visually detectable alterations. This pilot study warrants further confirmation in a larger cohort of PD patients and with different MR machines and MR parameters.
Key Points
• Magnetic resonance imaging data offers new insights into Parkinson’s disease
• Visual susceptibility-weighted imaging (SWI) analysis could not discriminate idiopathic from atypical PD
• However, support vector machine (SVM) analysis provided highly accurate detection of idiopathic PD
• SVM analysis may contribute to the clinical diagnosis of individual PD patients
• Such information can be readily obtained from routine MR data



Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer disease
- DaTScan:
-
123I-ioflupane SPECT
- DN:
-
dentate nucleus (of the cerebellum)
- DTI:
-
diffusion tensor imaging
- FA:
-
fractional anisotropy
- GM:
-
grey matter
- MCI:
-
mild cognitive impairment
- MRI:
-
magnetic resonance imaging
- MSA-P:
-
Parkinson variant of multiple system atrophy
- PD:
-
Parkinson disease
- PSP:
-
progressive supranuclear palsy
- RBF:
-
radial basis function
- RN:
-
red nucleus
- ROI:
-
region of interest
- SMO:
-
sequential minimal optimisation
- SN:
-
substantia nigra
- SPECT:
-
single-photon emission computed tomography
- SVM:
-
support vector machine
- SWI:
-
susceptibility-weighted imaging
- TBSS:
-
tract-based spatial statistics
- TFCE:
-
threshold free cluster enhancement
- VBM:
-
voxel-based morphometry
- WM:
-
white matter
References
Aquino D, Bizzi A, Grisoli M et al (2009) Age-related iron deposition in the basal ganglia: quantitative analysis in healthy subjects. Radiology 252:165–172
Thomas M, Jankovic J (2004) Neurodegenerative disease and iron storage in the brain. Curr Opin Neurol 17:437–442
Smith MA, Zhu X, Tabaton M et al (2010) Increased iron and free radical generation in preclinical Alzheimer disease and mild cognitive impairment. J Alzheimers Dis 19:363–372
Bishop GM, Robinson SR, Liu Q et al (2002) Iron: a pathological mediator of Alzheimer disease? Dev Neurosci 24:184–187
Haller S, Bartsch A, Nguyen D et al (2010) Cerebral microhemorrhage and iron deposition in mild cognitive impairment: susceptibility-weighted MR imaging assessment. Radiology 257:764–773
Dexter DT, Carayon A, Javoy-Agid F et al (1991) Alterations in the levels of iron, ferritin and other trace metals in Parkinson’s disease and other neurodegenerative diseases affecting the basal ganglia. Brain 114:1953–1975
Youdim MB, Ben-Shachar D, Riederer P (1993) The possible role of iron in the etiopathology of Parkinson’s disease. Mov Disord 8:1–12
Haacke EM, Xu Y, Cheng YC et al (2004) Susceptibility weighted imaging (SWI). Magn Reson Med 52:612–618
Oikawa H, Sasaki M, Tamakawa Y et al (2002) The substantia nigra in Parkinson disease: proton density-weighted spin-echo and fast short inversion time inversion-recovery MR findings. AJNR Am J Neuroradiol 23:1747–1756
Hughes AJ, Daniel SE, Ben-Shlomo Y et al (2002) The accuracy of diagnosis of parkinsonian syndromes in a specialist movement disorder service. Brain 125:861–870
Haller S, Lovblad KO, Giannakopoulos P (2011) Principles of classification analyses in mild cognitive impairment (MCI) and Alzheimer disease. J Alzheimers Dis 26:389–394
Noble WS (2006) What is a support vector machine? Nat Biotechnol 24:1565–1567
Hoehn MM, Yahr MD (1967) Parkinsonism: onset, progression and mortality. Neurology 17:427–442
Fazekas F, Chawluk JB, Alavi A et al (1987) MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging. AJR Am J Roentgenol 149:351–356
Gupta D, Saini J, Kesavadas C et al (2010) Utility of susceptibility-weighted MRI in differentiating Parkinson’s disease and atypical parkinsonism. Neuroradiology 52:1087–1094
Smith SM, Jenkinson M, Woolrich MW et al (2004) Advances in functional and structural MR image analysis and implementation as FSL. NeuroImage 23:S208–19
Smith SM, Nichols TE (2009) Threshold-free cluster enhancement: addressing problems of smoothing, threshold dependence and localisation in cluster inference. NeuroImage 44:83–98
Haller S, Nguyen D, Rodriguez C et al (2010) Individual prediction of cognitive decline in mild cognitive impairment using support vector machine-based analysis of diffusion tensor imaging data. J Alzheimers Dis 22:315–327
Kononenko I, Šimec E, Robnik-Šikonja M (1997) Overcoming the myopia of inductive learning algorithms with RELIEFF. Appl Intell 7:39–55
Platt J (1999) Sequential minimal optimization: A fast algorithm for training support vector machines. Advances in Kernel methods-support vector learning 208
Scholkopf B, Sung KK, Burges CJC et al (1997) Comparing support vector machines with Gaussian kernels to radialbasis function classifiers. IEEE Trans Signal Process 45:2758–2765
Wang Y, Butros S R, Shuai X et al (2011) Different iron-deposition patterns of multiple system atrophy with predominant parkinsonism and idiopathetic parkinson diseases demonstrated by phase-corrected susceptibility-weighted imaging. AJNR Am J Neuroradiol
Zhang J, Zhang Y, Wang J et al (2010) Characterizing iron deposition in Parkinson’s disease using susceptibility-weighted imaging: an in vivo MR study. Brain Res 1330:124–130
Huang XM, Sun B, Xue YJ et al (2010) Susceptibility-weighted imaging in detecting brain iron accumulation of Parkinson’s disease. Zhonghua Yi Xue Za Zhi 90:3054–3058
Rossi M, Ruottinen H, Elovaara I et al (2010) Brain iron deposition and sequence characteristics in Parkinsonism: comparison of SWI, T* maps, T-weighted-, and FLAIR-SPACE. Invest Radiol 45:795–802
Zhang W, Sun SG, Jiang YH et al (2009) Determination of brain iron content in patients with Parkinson’s disease using magnetic susceptibility imaging. Neurosci Bull 25:353–360
Grabner G, Haubenberger D, Rath J et al (2010) A population-specific symmetric phase model to automatically analyze susceptibility-weighted imaging (SWI) phase shifts and phase symmetry in the human brain. J Magn Reson Imaging 31:215–220
Pezzoli G, Canesi M, Galli C (2004) An overview of parkinsonian syndromes: data from the literature and from an Italian data-base. Sleep Med 5:181–187
Jeon HS, Han J, Yi WJ et al (2008) Classification of Parkinson gait and normal gait using spatial-temporal image of plantar pressure. Conf Proc IEEE Eng Med Biol Soc 2008:4672–4675
Brewer BR, Pradhan S, Carvell G et al (2009) Feature selection for classification based on fine motor signs of Parkinson’s disease. Conf Proc IEEE Eng Med Biol Soc 2009:214–217
Patel S, Lorincz K, Hughes R et al (2009) Monitoring motor fluctuations in patients with Parkinson’s disease using wearable sensors. IEEE Trans Inf Technol Biomed 13:864–873
Chan J, Leung H, Poizner H (2010) Correlation among joint motions allows classification of Parkinsonian versus normal 3-D reaching. IEEE Trans Neural Syst Rehabil Eng 18:142–149
Ozcift A (2011) SVM feature selection based rotation forest ensemble classifiers to improve computer-aided diagnosis of Parkinson disease. J Med Syst
Focke NK, Helms G, Scheewe S et al (2011) Individual voxel-based subtype prediction can differentiate progressive supranuclear palsy from idiopathic parkinson syndrome and healthy controls. Hum Brain Mapp 32:1905–1915
Groschel K, Kastrup A, Litvan I et al (2006) Penguins and hummingbirds: midbrain atrophy in progressive supranuclear palsy. Neurology 66:949–950
Seppi K, Schocke MF, Esterhammer R et al (2003) Diffusion-weighted imaging discriminates progressive supranuclear palsy from PD, but not from the Parkinson variant of multiple system atrophy. Neurology 60:922–927
Schocke MF, Seppi K, Esterhammer R et al (2002) Diffusion-weighted MRI differentiates the Parkinson variant of multiple system atrophy from PD. Neurology 58:575–580
Plant C, Teipel SJ, Oswald A et al (2010) Automated detection of brain atrophy patterns based on MRI for the prediction of Alzheimer’s disease. NeuroImage 50:162–174
Haacke EM, Mittal S, Wu Z et al (2009) Susceptibility-weighted imaging: technical aspects and clinical applications, part 1. AJNR Am J Neuroradiol 30:19–30
Hopp K, Popescu BF, McCrea RP et al (2010) Brain iron detected by SWI high pass filtered phase calibrated with synchrotron X-ray fluorescence. J Magn Reson Imaging 31:1346–1354
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haller, S., Badoud, S., Nguyen, D. et al. Differentiation between Parkinson disease and other forms of Parkinsonism using support vector machine analysis of susceptibility-weighted imaging (SWI): initial results. Eur Radiol 23, 12–19 (2013). https://doi.org/10.1007/s00330-012-2579-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-012-2579-y