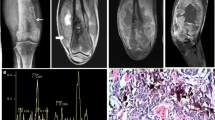
Abstract
Objectives
To determine whether proton magnetic resonance spectroscopy (1H-MRS) can help differentiate between benign and malignant soft tissue lesions, and to assess if there is a correlation between 1H-MRS data and the mitotic index.
Methods
MR measurements were performed in 43 patients with soft tissue tumours >15 mm in diameter. Six cases were excluded for technical failure. Examinations were performed at 1.5 T using a single-voxel point resolved spectroscopy sequence (PRESS) with TR/TE = 2000/150 ms. The volume of interest was positioned within the lesion avoiding inclusion of necrotic regions. In all patients, a histological diagnosis was obtained and the corresponding mitotic index was also computed. 1H-MRS results and histopathological findings were compared using the chi-squared test and correlation coefficient.
Results
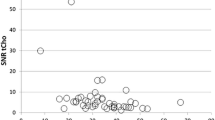
Choline was detected in 18/19 patients with malignant tumours and in 3/18 patients with benign lesions. The three benign lesions included one desmoid tumour, one ossificans myositis and one eccrine spiradenoma. Choline was not detected in 15 patients with benign lesions or in one patient with dermatofibrosarcoma protuberans. Resulting 1H-MRS sensitivity and specificity were 95% and 83% respectively.
Conclusions
Absence of choline peak is highly predictive of benign tumours suggesting that 1H-MRS can help to differentiate malignant from benign tumours.
Key Points
• 1H-MRS may allow differentiation between benign and malignant soft tissue lesions
• Absence of choline peak is highly predictive of benign soft tissue lesions
• Malignant tumours with a mitotic index >2/10 HPF had a positive choline peak
• A choline peak may still be identified in some benign tumours



Similar content being viewed by others
References
Weiss SW, Goldblum JR (2001) Enzinger and Weiss’s soft tissue tumors, 4th edn. Mosby-Harcourt Brace Company, St. Louis, MO, USA
Papagelopoulos PJ, Mavrogenis AF, Badekas A et al (2003) Foot malignancies: a multidisciplinary approach. Foot Ankle Clin 8:751–763
Moulton JS, Blebea JS, Dunco DM et al (1995) MR imaging of soft-tissue masses: diagnostic efficacy and value of distinguishing between benign and malignant lesions. AJR Am J Roentgenol 164:1191–1199
Campanacci M (1999) Bone and soft tissue tumors: clinical features, imaging, pathology and treatment, 2nd edn. Springer-Verlag, New York
Berquist TH, Ehman AL, Richardson ML (1987) Magnetic resonance of the musculoskeletal system. Raven, New York
Wetzel LH, Levine E (1990) Soft tissue tumors of the foot: value of MR imaging for specific diagnosis. AJR Am J Roentgenol 155:1025–1030
Berquist TH, Ehman RL, King BF et al (1990) Value of MR imaging in differentiating benign from malignant soft-tissue masses: study of 95 lesions. AJR Am J Roentgenol 155:1251–1255
Arkun R, Memis A, Akalin T et al (1997) Liposarcoma of soft tissue: MRI findings with pathologic correlation. Skeletal Radiol 26:167–172
Crim JR, Seeger LL, Yao L et al (1992) Diagnosis of soft-tissue masses with MR imaging: can benign masses be differentiated from malignant ones? Radiology 185:581–586
Verstraete KL, De Deene Y, Roels H et al (1994) Benign and malignant musculoskeletal lesions: dynamic contrast-enhanced MR imaging-parametric “first-pass” images depict tissue vascularization and perfusion. Radiology 192:835–843
Fletcher BD, Reddick WE, Taylor JS et al (1996) Dynamic MR imaging of musculoskeletal neoplasms. Radiology 200:869–872
Verstraete KL, Van der Woude HJ, Hogendoorn PC et al (1996) Dynamic contrast-enhanced MR imaging of musculoskeletal tumors: basic principles and clinical applications. J Magn Reson Imaging 6:311–321
Verstraete KL, Van der Woude HJ (2001) Dynamic contrast-enhanced magnetic resonance imaging. In: De Schepper AM (ed) Imaging of soft tissue tumors, 2nd edn. Springer, Berlin, pp 83–104
Van Rijswijk CSP, Geirnaerdt MJA, Hogendoorn PCW et al (2004) Soft-tissue tumors: value of static and dynamic gadopentetate dimeglumine-enhanced MR imaging in prediction of malignancy. Radiology 233:493–502
Verstraete KL, Dierick A, De Deene Y et al (1994) First-pass images of musculoskeletal lesions: a new and useful diagnostic application of dynamic contrast-enhanced MRI. Magn Reson Imaging 12:687–702
Payne GS, Leach MO (2006) Applications of magnetic resonance spectroscopy in radiotherapy treatment planning. Br J Radiol 79:S16–S26
Gupta RK, Cloughesy TF, Sinha U et al (2000) Relationships between choline magnetic resonance spectroscopy, apparent diffusion coefficient and quantitative histopathology in human glioma. J Neuro-Oncol 50:215–226
Doganay S, Altinok T, Alkan A et al (2011) The role of MRS in the differentiation of benign and malignant soft tissue and bone tumors. Eur J Radiol 79:e33–e37
Wang CK, Li CW, Hsieh TJ et al (2004) Characterization of bone and soft-tissue tumors with in vivo 1H MR spectroscopy: initial results. Radiology 232:599–605
Delorme S, Weber MA (2006) Applications of MRS in the evaluation of focal malignant brain lesions. Cancer Imaging 6:95–99
Swindle P, McCredie S, Russell P et al (2003) Pathologic characterization of human prostate tissue with proton MR spectroscopy. Radiology 228:144–151
Cheng LL, Burns MA, Taylor JL et al (2005) Metabolic characterization of human prostate cancer with tissue magnetic resonance spectroscopy. Cancer Res 65:3030–3034
Ronen SM, Leach MO (2001) Breast imaging technology: imaging biochemistry - applications to breast cancer. Breast Cancer Res 3:36–40
Katz-Brull R, Seger D, Rivenson-Segal D et al (2002) Enhanced choline metabolism and reduced choline-ether-phospholipid synthesis. Cancer Res 62:1966–1970
Fayad LM, Bluemke DA, McCarthy EF et al (2006) Musculoskeletal tumors: use of proton MR spectroscopic imaging for characterization. J Magn Reson Imaging 23(1):23–28
Fayad LM, Barker PB, Jacobs MA et al (2007) Characterization of musculoskeletal lesions on 3-T proton MR spectroscopy. AJR Am J Roentgenol 188:1513–1520
World Medical Association (2000) Declaration of Helsinki: Ethical Principles for Medical Research Involving Human Subjects, 5th rev. Edinburgh, Scotland
Bartella L, Morris EA, Dershaw DD et al (2006) Proton MR spectroscopy with choline peak as malignancy marker improves positive predictive value for breast cancer diagnosis: preliminary study. Radiology 239:686–692
Sardanelli F, Fausto A, Di Leo G et al (2009) In vivo proton MR spectroscopy of the breast using the total choline peak integral as a marker of malignancy. AJR Am J Roentgenol 192:1608–1617
Fayed N, Olmos S, Morales H et al (2006) Physical basis of magnetic resonance spectroscopy and its application to central nervous system diseases. Am J Appl Sci 3:1836–1845
Sijens PE, van den Bent MJ, Nowak PJ et al (1997) 1H chemical shift imaging reveals loss of brain tumor choline signal after administration of Gd-contrast. Magn Reson Med 37:222–225
Roebuck JR, Cecil KM, Schnall MD et al (1998) Human breast lesions: characterization with proton MR spectroscopy. Radiology 209:269–275
Baek HM, Chen JH, Yu HJ et al (2008) Detection of choline signal in human breast lesions with chemical-shift imaging. J Magn Reson Imaging 27:1114–1121
Huang W, Fisher PR, Dulaimy K et al (2004) Detection of breast malignancy: diagnostic MR protocol for improved specificity. Radiology 232:585–591
WHO Classification of Tumours (2002) Pathology & genetics—Tumours of soft tissue and bone. IARC, Lyon
Newcombe RG (1998) Two-sided confidence intervals for the single proportion: comparison of seven methods. Statist Med 17:857–872
Royal College of Obstetricians and Gynaecologists (2002) The investigation and management of the small-for-gestational-age fetus, guideline no.31. RCOG, London
Dowdy S, Weardon S, Chilko D (2004) Statistics for research, 3rd edn. Wiley, New Jersey
Hashimoto K, Brownstein MH, Jakobiec FA (1974) Dermatofibrosarcoma protuberans. A tumor with perineural and endoneural cell features. Arch Dermatol 110:874–885
Brenner W, Schaefler K, Chhabra H et al (1975) Dermatofibrosarcoma protuberans metastatic to a regional lymph node. Report of a case and review. Cancer 36:1897–1902
Herminghaus S, Pilatus U, Möller-Hartmann W et al (2002) Increased choline levels coincide with enhanced proliferative activity of human neuroepithelial brain tumors. NMR Biomed 15:385–392
Krouwer HGJ, Kim TA, Rand SD et al (1998) Single-voxel proton MR spectroscopy of nonneoplastic brain lesions suggestive of a neoplasm. AJNR Am J Neuroradiol 19:1695–1703
Bitsch A, Bruhn H, Vougioukas V et al (1999) Inflammatory CNS demyelination: histopathologic correlation with in vivo quantitative proton MR spectroscopy. AJNR Am J Neuroradiol 20:1619–1627
Parikh J, Hyare H, Saifuddin A (2002) The imaging features of post-traumatic Myositis Ossificans, with emphasis on MRI. Clin Radiol 57:1058–1066
Manaster BJ, May DA, Disler DG (2007) Musculoskeletal Imaging, 3rd edn. Mosby Elsevier, Philadelphia
Blamire AM (2008) The technology of MRI - the next 10 years? Br J Radiol 81:601–617
Qayyum A (2009) MR spectroscopy of the liver: principles and clinical applications. RadioGraphics 29:1653–1664
Erturk SM, Alberich-Bayarri A, Herrmann KA et al (2009) Use of 3.0-T MR imaging for evaluation of the abdomen. RadioGraphics 29:1547–1563
Ricci E, Pitt A, Keller PJ et al (2000) Effect of voxel position on single-voxel MR spectroscopy findings. AJNR Am J Neuroradiol 21:367–37
Bolan PJ, Nelson MT, Yee D et al (2005) Imaging in breast cancer: magnetic resonance spectroscopy. Research 7:149–152
Li BSY, Regal J, Gonen O (2001) SNR versus resolution in 3D 1H MRS of the human brain at high magnetic fields. Magn Reson Med 46:1049–1053
Usenius JPR, Kauppinen RA, Vainio PA et al (1994) Quantitative metabolite patterns of human brain tumors: detection by 1H NMR spectroscopy in vivo and in vitro. J Comput Assist Tomo 18:705–713
Manton DJ, Lowry M, Blackband SJ et al (1995) Determination of proton metabolite concentrations and relaxation parameters in normal human brain and intracranial tumours. NMR Biomed 8:104–112
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Russo, F., Mazzetti, S., Grignani, G. et al. In vivo characterisation of soft tissue tumours by 1.5-T proton MR spectroscopy. Eur Radiol 22, 1131–1139 (2012). https://doi.org/10.1007/s00330-011-2350-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-011-2350-9