Abstract
Radiologists have a special role in the management of neutropenic patients. The appropriate investigational technique, frequently targeted differential diagnosis, and the special needs of these patients, need to be understood. Early detection of a focus is the major goal in febrile neutropenic patients. As pneumonia is the most common focus, chest imaging is a special radiological task. The sensitivity of chest X-ray, especially in supine position, is known to be low; therefore, the very sensitive high-resolution CT (HRCT) became gold standard in neutropenic hosts and will probably be replaced by thin-section multislice CT (MSCT) in the near future. Costs of high-resolution CT are low in comparison to antibiotics. An infiltrate needs to be localised, so that a physician can utilise this information as a guidance for invasive procedures for further microbiological work-up. The radiological characterisation of infiltrates gives a first and rapid hint to differentiate between different sorts of infectious (typical bacterial, atypical bacterial, fungal) and non-infectious aetiologies. Follow-up investigations need careful interpretation according to disease and concomitant treatment. Due to an increased incidence of fungal infiltrates even with appropriate therapy, follow-up of an infiltrate must use further parameters in addition to lesion size. Temporary exclusion of infectious involvement of the lung with high accuracy remains of special interest for clinicians.
Similar content being viewed by others
Introduction
Modern tumour therapy utilises numerous high-dose chemotherapy protocols. This induces an increasing number of patients with long-term neutropenia (>10 days) with definitive immunodeficiency [1, 2]. In long-term neutropenia the risk for infections rises to more than 85% [3]. Furthermore, after initially successful empirical antibiotic treatment, an infectious relapse occurs in approximately 50% of the patients [4, 5]; thus, physicians are confronted with an increasing number of immunocompromised hosts showing non-specific clinical signs of infection. Therefore, an empirical antibiotic strategy will be started initially, which covers the most frequent types of infections. This approach is very successful [4, 6, 7] but non-targeted. Because the underlying micro-organism remains unknown, empirical antibiotic strategy has several disadvantages.
-
A de-escalation of a broad spectrum to narrow antibiotic usage, at least at the end of the infectious course, remains troublesome. This enhances costs for antibiotics as well as the rates of adverse effects, possibly overutilised drugs, and bacterial resistance.
-
Beside infections, non-infectious inflammatory diseases, such as relapse of haematological disease, graft-vs-host disease (GvHD), etc., might mimic infection; therefore, non-infectious differential diagnoses remain underestimated.
-
After an empirically treated episode of infection, the plan for the next chemotherapeutic course usually includes an antibiotic prophylaxis. The so-called secondary prophylaxis is again usually non-specific.
-
The local epidemiology remains unknown without appropriate diagnostic procedures; thus, patients will profit from identification of the underlying disease, and are sent to a radiologist for identification of the focus of infection. This early identification of the infection site is a major task for clinicians taking care for neutropenic patients. Frequently, fever is the only sign for infection and different aetiologies have to be considered in this setting (Table 1).
Table 1. Overview of frequent infectious and infectious-like diagnoses in neutropenia and thereafter
The major role of the radiologist focuses on the detection of the focus of infection or non-infectious disease. If an organ is affected, invasive diagnostic procedures can be undertaken for identification of the underlying micro-organism. The acquired information helps to localise the most suspicious region within this organ, e.g. the selection of a certain segment helps in guiding bronchoalveolar lavage or biopsy [10]. Furthermore, characterisation of the detected focus may give the clinician a clue for the underlying disease (Table 2) [10, 11]. The detected focus might additionally serve as a practicable follow-up parameter to document the course of the infection and therapeutic success.
Besides detecting infectious sites, the technical procedure should also present with a high negative predictive value to exclude the infectious involvement temporarily.
Risk and epidemiology
Bacterial infections are responsible for approximately 90% of infections in the early phase of neutropenia (Fig. 1; Table 1) [12]. In an allogeneous transplantation setting, gram-negative bacteria are documented in 16–31% of cases, whereas in 65–75% gram-positive bacteria are found [12]. On the other hand, the gram-negative bacteria lead to a significantly higher morbidity [12].
Infectious and infectious-like syndromes as wells as major risk factors at various times after bone marrow transplantation. Day 0 day of transplantation, HSV herpes simplex virus, CMV cytomegalovirus, Adeno adenovirus, VZV varicella zoster virus, GvHD graft-vs-host disease. (Adapted from [43])
Risk stratification and pharmacological improvements have enhanced the rational of empirical antibiotic strategy (Table 3). Bacteria are covered sufficiently by antibiotic (t.m. antibacterial) therapy [4]. If this approach fails or an infection breaks through, an antibacterial second-line treatment has usual limited success. The low success rate of 30% in second line demonstrates the limited role of antibiotic switching. Antifungal supplementation at this time point, however, reaches success rates of up to 78% [13]. This underlines the point that the early detection of non-bacterial pneumonia is the major task. From a clinical point of view, the detection of an ongoing bacterial infection appears less important than detecting non-bacterial pneumonia, e.g. fungal pneumonia; therefore, making a further characterisation of a bacterial pneumonia becomes less desirable for the clinician.
Besides prophylaxis and efficient initial broad-spectrum treatment, interventional therapy regimens for second- and third line are formulated [12]. In the treatment of pneumonia, prospective investigations demonstrate a major limitation of the empirical therapy due to fungal organisms [13]. Due to the recommendation of the European Organization for Research and Treatment of Cancer (EORTC) / Invasive Fungal Infections Cooperative Group and Bacterial and Mycosis Study Group (BAMSG), every new infiltrate is a minor criterion for fungal pneumonia and typical signs are a major criterion of fungal pneumonia [14]. This classification cannot be transferred to other immunodeficiencies such as AIDS [15].
Early detection
The necessity for an early detection of the focus of infection is based on a high mortality rate of infections in immunocompromised hosts and high costs of prolonged hospitalisation. The costs for broad-spectrum antibiotics and conventional amphotericin B varies from € 100 to € 140 per day and rises up to € 750 per day if the liposomal preparation of amphotericin B is used. (AmBisome is amphotericin B in liposomal preparation. Due to the sometimes life-threatening side effects of the conventional amphotericin B, the liposomal preparation is used in special cases only. However, the expense is enormous; therefore, a definite fungal diagnosis would be ideal for a targeted antifungal treatment, i.e. AmBisome at 3 mg/kg body weight or higher.) This is a relevant high cost in comparison with the costs of a non-enhanced CT scan of € 230 (in Germany, inpatient, including report and comparison with previous scans). Other empirical studies investigating new antifungal agents did not reach the study end point, so that the approval of the medical authorities was denied, thus making expensive methods more cost-effective in early detection. Usually, the search for the focus of infection consists of a physical examination and laboratory findings. Besides epidemiological knowledge, the results should be taken into account to identify the organ system which is most likely affected. After identification of the most suspected organ system(s), select the appropriate imaging technique for investigation. A high sensitivity and useful negative predictive value are needed.
Exact frequencies of organ infections are difficult to determine and differ from clinical (i.e. patients alive) and pathological evaluation (i.e. autopsies). Clinically, lungs are affected in 30%, paranasal sinuses in 3% of neutropenic patients, and 30% in an allogeneous transplantation setting (concomitant to pneumonia). Gastrointestinal tract, liver, spleen, central nervous system especially after allogeneous transplantation, and kidneys are rarely affected [6]. Due to the tremendously higher frequency of pneumonia in comparison with all other organs systems, this article focuses on early detection of pneumonia. A detailed discussion of the other organ systems and techniques is far beyond the scope of this article. The reader is referred to other publications in the literature [16, 17].
Chest X-ray
Chest X-ray (CXR) is widely performed when pneumonia is suspected or should be excluded [18, 19]. The CXR has several advantages such as being quick, widely available (even on the ward), inexpensive and low radiation dose. Some referrers prefer CXR in supine position done on the ward to keep the neutropenic patients isolated. But especially supine CXR has the crucial disadvantage of superimposition and, therefore, a limited sensitivity for the detection of pneumonia (Figs. 2, 3) [18, 20]. In a recent study with 40 patients suffering from fever of unknown origin (FUO) after bone marrow transplantation (BMT), digital CXR in supine position achieved a sensitivity for the early detection of pneumonia of only 46% [21]. Although CXR provides relevant clinical information concerning central venous catheters (CVC), pleural effusion and pulmonary congestion [21], it fails in the early detection or even exclusion of pneumonia, which is a major task in immunodeficient patients. The CXR in supine position alone is not recommended for the early detection of pneumonia in immunocompromised hosts [7].
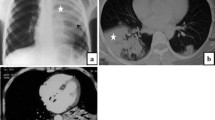
Neutropenic febrile patient receiving broad spectrum antibiotic therapy. a, b Chest X-ray (CXR) was normal at day 3 of fever. c High-resolution computed tomography (HRCT) performed the same day demonstrates bilateral infiltrates, which were hidden behind the heart in posterior--anterior and the spine in lateral projection
The c small ill-defined nodule in the right upper lobe of the 34-year-old neutropenic AML patient was even retrospectively not visible at a, b chest X-ray done the same day. d Amphotericin-B treatment was started due to suspicion of fungal pneumonia; however, the nodule size increased during haematological reconstitution 2 weeks later. In preparation of bone marrow transplantation, the lesion was resected to prevent from septical spread. Aspergillus pneumonia was verified
On the other hand, if an infiltrate is apparent at CXR, the options for pneumonia characterisation are very limited; thus, if pneumonia is in question in these hosts, HRCT or thin-section MSCT should be preferred at any time point if available [22].
High-resolution CT
The effective radiation dose of CXR is approximately 0.2 mSv but can be ten times higher depending on the equipment used [23]. In low-dose multislice CT of the chest, an effective radiation dose of 1.1 mSv is reported, whereas the gap in single-slice HRCT can reduce the dose to approximately 10% of this value [24]. Radiation dose is not a real limitation in the investigation of neutropenic patients because they frequently receive radiation for conditioning therapy for transplantation (total body irradiation, TBI) in more than 1000 times higher dosages than for diagnostic purposes. Furthermore, chemotherapy has similar cytotoxic effects on the patient; thus, high-resolution or thin-section CT has been introduced as the standard technique in neutropenic patients.
After previous studies describing a limited use of CXR in these patients [20], a prospective study investigated the benefit of HRCT in comparison with CXR in the early detection of pneumonia: 188 febrile neutropenic patients who did not defervest after 48 h on empirical antibiotic therapy [25] were included. If CXR was normal at this time, HRCT was done. In approximately 60% of the patients with normal CXR, HRCT demonstrated infiltrates (Fig. 4). During the following days, in approximately 50% of the cases (total 30%) the pneumonia seen at HRCT was verified either by microbiology or an infiltrate became visible on CXR. Another 40% had a normal chest X-ray and a normal HRCT when entering the study. In these patients, pneumonia occurred in only 10% during follow-up [25]. Methodological limitations are: (a) a mixed immune status due to inclusion of patients after conventional chemotherapy or transplantation setting; and (b) the verification of underlying micro-organism, which is either uncertain, or when taking only certain identifications into account, with a selection bias resulting [25]. Also the efforts in this trial (bronchoalveolar lavage, interdisciplinary clinical conference required) had limited effect. For the interpretation of microbiological results, superinfection, non-relevant isolates and contamination always have to be considered.
Number of HRCT demonstrating an infiltrate (shaded) or no infiltrate (white) with normal CXR the same day in neutropenia and empirical antibiotic therapy. The verification of pneumonia was done either by detection of an infiltrate on CXR or evidence of a relevant micro-organism during follow-up after HRCT. Very few verifications occur after normal HRCT (white), whereas many verifications are done after HRCT demonstrating infiltrate (shaded)
Besides the detection of pneumonia, the exclusion of pneumonia is relevant information for the referring physician; therefore, the time point of pneumonia verification (by CXR or microbiology) has been evaluated by [25] to assess the negative predictive value of HRCT. In patients with normal HRCT pneumonia verification happened rarely, slowly, and continuously during the whole follow-up, but never during the first 5 days (Fig. 5). In patients with infiltrates at HRCT, pneumonia was verified during the next 5–10 days in most cases (Fig. 5) [25].
Probability of having no pneumonia by either detection of an infiltrate on a CXR or evidence of a relevant micro-organism during follow-up after HRCT. Kaplan-Meier analysis for patients with normal HRCT scans (grey line) and patients with pneumonia on HRCT scans (black line). The difference was highly significant (p<0.0001). Very few verifications occur very late after a normal HRCT, whereas most verifications after HRCT demonstrating infiltrate take place during the first 5–10 days
Thus, HRCT yielded very promising results to be used as a screening technique with good sensitivity (87%) and negative predictive value (88%). The gap to 100% was caused mainly by later occurring pneumonia leading to a false-negative result, and minor infiltrates which were only detected at HRCT but, due to early detection and early treatment, did not progress to become visible on CXR. The additional and early use of HRCT achieved a time gain of approximately 5 days during which HRCT was able to exclude pneumonia [21]. This fact is essential in the management of immunodeficient hosts [7].
CT technique
Besides HRCT, which is established as the technique of choice for detailed investigation of the lung parenchyma, spiral CT and recently multislice CT are available for lung imaging. Limitation of spiral CT is especially in detection of inflammatory lung disease, especially ground-glass opacification [26]; therefore, HRCT should be performed as a standard. No additional information is expected from supplemental spiral CT as shown in AIDS patients [27]; however, the non-contiguous scanning using HRCT involves limitations in nodule detection and quantification. This topic is most relevant in follow-up scans and will be solved by usage of multislice CT (MSCT), whereas thin slices (approximately 1 mm) will combine the advantages of HRCT and spiral CT as well [28, 29]. It will certainly find a place as the method of choice when scanners become more widely employed [28, 29, 30].
Contrast enhancement is generally not necessary for detecting and characterising pneumonia [10, 22]. Only in special situations, such as pulmonary embolism or bleeding, e.g. due to vessel erosion by aspergillosis, is CT angiography beneficial [31]. In an allogeneous setting, bronchiolitis obliterans has to be considered [32, 33]. Air trapping is a relevant finding in this respect; therefore, an additional expiratory CT scan is helpful [32, 33].
MRI
Magnetic resonance imaging has been evaluated for the investigation of pulmonary disease since it has a known benefit in lesion characterisation [34, 35]; however, there are no studies that demonstrate the benefit of MRI in the early detection of pneumonia, where a high sensitivity is required (Fig. 6). In advanced stages, CT and MRI are comparable in the visualisation of infiltrates [35]. But CT is highly available, easier, and faster to perform as well as less susceptible to breathing artefacts. The MRI is superior to CT in the detection of abscesses due to a clearer detection of central necrosis in T2-weighted images and rim enhancement after contrast application in T1-weighted images [34]; however, this fact has limited clinical impact and duration of MRI and required compliance is substantially higher compared with CT.
Standard recommendation
In contrast to systemic infections, identification of the underlying organism in pneumonia is more difficult and complex. Trials to enforce this identification did not improve the therapeutic outcome significantly [13]; therefore, an empirical therapy in febrile immunodeficient patients based on imaging results also is widely used.
The use of HRCT is recommended for early detection of pneumonia [13]. The crucial fact is that HRCT allows for an optimisation for the indication and localisation of invasive diagnostic procedure, e.g. bronchoalveolar lavage (BAL). On the other hand, the exclusion of pneumonia can be obtained with a higher confidence compared with the exclusive use of CXR. The sequential cascade as shown in Fig. 7 can be modified if the CT capacity allows for the skipping of CXR.
Recommendations of the Guidelines of the Infectious Diseases Working Party of the German Society of Haematology and Oncology [13]
On the other hand, our own experience demonstrates the known limited success rate of invasive procedures. From 183 BAL specimens derived from January 2002 until November 2002, 71 had a positive bacterial/fungal result (39%). Only 9 of the 71 isolates were considered to be relevant for the suspected infection (8%), which results in a detection rate of 5% for the whole BAL and microbiological approach (virological evaluation was not considered).
Follow-up
The observation of growing infiltrates during haematological reconstitution has been quantified and documented recently [36]. Caillot et al. [36] performed HRCT at a standard interval in 25 neutropenic patients with proven pulmonary aspergillosis once a week. They documented the time point of different patterns and evaluated the size of the infiltrate. They frequently found the halo sign (Fig. 8) in their first CT and report a low sensitivity of this well-described pattern (68%). During follow-up this pattern disappeared. In contrast, the more specific air-crescent sign (Fig. 9) became more frequent during follow-up (increased from 8% to 63%). The size of the infiltrate increased four times despite of successful treatment and haematological reconstitution. In this approach, the first detection of pneumonia was at day 19 of neutropenia. This is late compared with day 11 in the study for early detection [25]. The increasing infiltrate is an immunological phenomenon due to invasion of newly appearing neutrophile granulocytes at the beginning of haematological reconstitution. In critically ill patients, this is a known risk factor to develop acute respiratory distress syndrome [18].
Neutropenic febrile patient who underwent autologous stem-cell transplantation due to non-Hodgkin's lymphoma. At day 2 after transplantation, neutropenia and fever occurred; therefore, antifungal treatment (amphotericin B) was started. Ill-defined pulmonary nodules were diagnosed at day 7. Haematological reconstitution took place at day 13; simultaneously, the nodule size reached its maximum during this course. Under continuously antifungal treatment and nearly normal leukocytes, the halo disappeared slowly, the lesions shrunk and a central cavitation occurred. Finally, the lesions almost disappeared
Characterisation
A radiologist's dream is to be capable of identifying the underlying micro-organism in pneumonia of immunocompromised hosts with a sufficient specificity. In clinical routine, however, one has to wait for the results of microbiological and pathological analysis of samples. This requires several days to be obtained and it will only be feasible in some cases [37]. Furthermore, the isolated organism is not necessarily the underlying problem: surface colonisation provides difficulties in the correct interpretation of microbiological results and superinfection with an additional organism takes place in approximately 20% [13, 38].
In some cases, imaging can give more or less useful clues, instead of verifications, for the underlying disease. The quality of these clues depends on the interdisciplinary co-operation between clinician and radiologist and the radiologists experience with these diseases. This requires an informational exchange concerning relevant patient data such as standard neutropenia, and allogeneous or autologous transplantation setting. Furthermore, the positivity for viral disease in graft and host is an essential information for correct interpretation of HRCT. Also the applied chemotherapeutic substances or the conditioning regimen need to be discussed (Table 2).
There are several differential diagnoses of FUO in immunocompromised hosts, which might appear clinically similar and where HRCT gives valuable hints for the differential diagnosis [10, 11, 39, 40]. The most useful clues are listed in Table 2.
Bacterial pneumonia
Since bacterial infections are responsible for approximately 90% of infections during the early phase of neutropenia (Fig. 1; Tables 1, 3) [12], their empirical treatment has been optimised during the past decades.
The radiological appearance of bacterial pneumonia includes consolidation, especially bronchopneumonia, and positive pneumobronchogram (Fig. 2) [40, 41, 42]. In contrast to immunocompetent patients, ground-glass opacification is found more often and remains non-specific.
Fungal pneumonia
Continuous febrile neutropenia is associated with invasive fungal infection [5]. In Europe, mainly Aspergillus species are the underlying organism. Mucormycosis seems to increase, but besides the "bird's-nest" sign, it is clinically and radiologically similar to aspergillosis. Ante mortem, Candida species are a rare pathogen entailing pneumonia (Fig. 10) [7]. Most isolates represent contamination due to surface colonisation. To describe typical findings of fungal pneumonia caused by different pathogens, a dedicated review is necessary [43]. This article focuses on the most relevant information for haematological patients. The appearance of pulmonary infiltrates with fungus are typical patterns in the early phase ill-defined nodules (Figs. 6, 8, 10) [42] in combination with the halo sign (Figs. 8, 10) [42], which is non-specific, late-phase, air-crescent-sign [44] and cavitations (Fig. 10).
Bilateral ill-defined nodules made the suspected diagnosis of a fungal infection which was treated accordingly. Candida spp. were identified from blood culture and suspected to be involved with this pneumonia. The small lesions developed into cavitations at haematological reconstitution and decreased. Due to increasing liver enzymes and because of known hepatospenic candidiasis after candidaemia, contrast-enhanced CT scan was done. Biopsy from the detected lesions revealed Candida spp. once again
For use in the context of clinical and epidemiological research in neutropenic patients, the EORTC and BAMSG have defined standards for the interpretation of radiological findings in invasive fungal infections [14]. The new occurrence of these "typical" CT patterns (halo sign, air-crescent sign, or cavity within area of consolidation) are classified as a major clinical criterion for fungal pneumonia. Furthermore, if a new infiltrate is observed even without a typical fungal pattern, it is classified as a minor clinical criterion for fungal pneumonia [14].
Air-crescent sign and cavitation occur simultaneously with haematological reconstitution during the late phase of infection (Fig. 10) [44]. Therefore, air-crescent and cavitation sign are known to have a positive prognosis; however, the specificities of these findings are limited and relevant differential diagnoses have to be considered (Fig. 9) [45]. The histopathological work-up verified fungal pneumonia only in 56% of cases [45]. Relevant differential diagnosis for the halo sign, such as cryptogenic organising pneumonia (formerly bronchiolitis obliterans organising pneumonia), pulmonary haemorrhage, and other infections (e.g. CMV, TBC, abscesses: Fig. 9; Candida: Fig. 10) have to be considered [45].
There are other useful patterns in the identification of fungal pneumonia: distribution along the bronchovascular bundle resulting in the feeding vessel sign with an angiotropic location.
The ongoing development of antifungal drugs may affect also the radiological procedures in the near future. Currently, conventional preparation of amphotericin B has a limited tolerability; however, liposomal amphotericin B is very costly. New Azoles and the new group of Echinocandins are being evaluated in clinical trials or are already on the market. These new agents have an improved safety record, but their efficiency ranges around 50%. This might change the question for fungal pneumonia for radiologists.
Pneumocystis jiroveci pneumonia
Pneumocystis jiroveci pneumonia (formerly P. carinii; the abbreviation PcP continues for Pneumocystis pneumonia) [46] is not a typical finding in haematological patients except in the late phase after allogeneous transplantation together with chronic GvHD [12]. Under the standard trimethoprim/sulfamethoxazol prophylaxis, 8% of the patients develop PcP, and without prophylaxis, 29% [12]. Mortality is 4–15% in these cases [12].
High-resolution CT provides a valuable characterisation for this micro-organism [11, 22, 10, 40] and is a reliable method for differentiating PcP from other infectious processes [42, 47]. A combination of ground-glass opacities and intralobular septa sparing out the subpleural space (i.e. perihilar distribution) are very suggestive for PcP (Fig. 11) [40, 42, 47, 48].
Bilateral pneumonia caused by Pneumocystis jiroveci (PcP) at different stages of immunosuppression. The subpleural space is typically left out. a Diffuse ground-glass opacification appears typically in the early phase of infection, whereas b consolidations appear at a fulminant course. c The predominance of intralobular linear patterns takes place during a later and treated stage of PcP
Tuberculosis
Tuberculosis (TB) has always to be considered as a rare but relevant differential diagnosis. In immunocompromised host, TB appears different compared to immunocompetent hosts (e.g. gangliopulmonary, i.e. primary, forms) [49]. More widespread lymphogenic and haematogeneous dissemination can occur and, therefore, the clinical course might be fulminant [49, 50]. On the other hand, TB might mimic or come along with other infections such as pulmonary aspergillosis or systemic candidiasis [50].
In immunocompromised hosts a peribronchial distribution (resulting in a "tree-in-bud" sign) of small, sometimes cavitated ill-defined nodules can be obtained due to miliary distribution [49, 50]. Gangliopulmonary (primary) forms, however, present with inhomogeneous consolidation and necrotic mediastinal/hilar lymphadenopathy [49].
Viral pneumonia
Atypical pneumonia in neutropenic patients and especially after haematological reconstitution is frequently caused by virus infection. Viral pneumonia is associated with a mortality of approximately 50% in neutropenic hosts. The most frequent suspected virus is CMV; furthermore, herpes, influenza, parainfluenza, adenovirus and respiratory syncytial (RSV) viruses have to be considered. There are no radiological patterns available to differentiate various forms of viral pneumonia; however, even the information that there is viral pneumonia is very valuable to the clinicians. Appropriate drug regimens are available for many of these viruses. The typical appearance of viral pneumonia in the early stage is ground-glass opacification [40, 42] and mosaic pattern with affected and non-affected secondary lobules lying adjacent to one another (Fig. 12).
Non-infectious disease
Certain non-infectious diseases have to be considered in haematological patients: GvHD; radiation or drug toxicity; pulmonary congestion; bleeding; or early tumour recurrence. Fever, dyspnoea or lab findings (C-reactive protein, transaminases) might be caused by some of these diseases and obscure the differentiation from infection. For instance, in GvHD, the therapeutic approach to non-infectious caused infiltrates is in contrast to infection: further suppression of the immune system. This differential diagnosis is very helpful for clinicians. The HRCT is able to assist in the detection and characterisation of these diseases [10, 11, 39, 22].
Graft-vs-host disease
Pulmonary manifestation of chronic GvHD occurs in approximately up to 10% of patients usually 9 months after allogeneous transplantation (Fig. 13) [51]. Bronchiolitis obliterans is the pulmonary manifestation of this reaction [32]. Unfortunately, the radiological appearance is similar to viral pneumonia, and to make things more complicated, clinical appearance and time point for both diseases are often similar (Fig. 1).
A 28-year-old man after allogeneic re-transplantation due to CMV. The HRCT was performed due to fever, cough and dyspnoea. Peripheral intralobular septa (arrows) and ground-glass opacification was determined at HRCT at day 91 after transplantation. Tree-in-bud pattern (arrowheads) points to bronchiolitis obliterans. Acute GvHD was diagnosed from transbronchial biopsy. After increasing immunosuppression, the clinical symptoms and the radiological signs disappeared. Note the similarity to Fig. 15
Ground-glass opacification and mosaic pattern, as well as signs of bronchiolitis obliterans, such as air trapping [32, 33], and bronchus wall thickening occur during the early stage of pulmonary GvHD (Fig. 13), whereas intralobular septa and tree-in-bud follow in later stages [11, 51, 52].
Radiation toxicity
An incidence of 5–25% even after total body irradiation (TBI) is reported, which is applied for conditioning therapy prior to bone-marrow or stem-cell transplantation [53]. One problem in detecting radiation toxicity is the time delay after radiation, which is approximately 3 weeks, but can also occur several months later [52, 53].
At HRCT, it is characterised by ground-glass opacities with transition to consolidations (Fig. 14) [52, 53]. The key finding is the limitation of these patterns to the parenchyma within the radiation field. Even in TBI, lung parenchyma is blocked out; thus, paramediastinal and apical lung parenchyma suffers mainly from radiation toxicity.
Three weeks after local radiation for a tumorous spine destruction, this patient suffered from fever and dyspnoea. Perihilar infiltrates appeared suddenly. Intralobular septa, consolidation and ground-glass opacification were determined at HRCT. Especially the paramediastinal distribution of the infiltrates led to the differential diagnosis of radiation pneumonitis. After failure of antibiotic escalation (chosen because of a concomitant abscess), steroids were applied additionally. This led to a quick improvement of the symptoms as well as reduction of infiltrates
Drug toxicity
Especially high-dose chemotherapy protocols are used for conditioning therapy which results in pulmonary drug toxicity. Some of the frequently used agents are bleomycin, methotrexate (MTX), cytarabine (Ara-C), carmustine (BCNU), and many more (Fig. 15) [54]. Radiologists have to consider a treatment with these drugs in their evaluation and request this information.
A 40-year-old man received chemotherapy including bleomycin (PEB protocol) for testicular cancer. An HRCT was performed because of fever, cough and dyspnoea. The HRCT revealed peripheral intralobular septa and ground-glass opacification. Due to the known pulmonary toxicity of the applied bleomycin, a pulmonary drug toxicity was suspected and verified by open-lung biopsy. Symptoms disappeared and findings decreased after application of steroids. Note the similarity to Fig 13
The term drug-induced pneumonitis includes mainly diffuse alveolar damage, non-specific interstitial pneumonia and bronchiolitis obliterans organising pneumonia [54]. The HRCT appearance consists of ground-glass opacities with transition to consolidations, intralobular septa, air trapping, and possibly the non-specific "crazy-paving" pattern [52, 54]. This is quite similar to radiation toxicity but without being limited to the radiation field.
Pulmonary congestion
Dyspnoea and infiltration are frequent in patients suffering from pulmonary congestion. Due to CVC, extensive hydration for renal protection during chemotherapy, frequent temporary renal impairment, hypoproteinosis, or pulmonary congestion appear even in younger patients. It is one of the most frequent disorders in intensively treated patients.
At CXR, pulmonary congestion might be combined with infiltration. Computed tomography demonstrates a thickening of the lymphatic vessels, which corresponds to the well-known Kerley lines (Fig. 16).
Leukaemic infiltration
Leukaemic pulmonary infiltration is a less common clinical finding. Especially the perilymphatic pulmonary interstitium is involved [55]. This can be visualised at CT as thickening of the bronchovascular bundles and interlobular septa. Besides this, non-lobular and non-segmental ground-glass opacifications can be seen [39]. This pattern arrangement might mimic pulmonary congestion (Fig. 16).
Pulmonary haemorrhage
In pancytopenia, pulmonary bleeding occurs spontaneously, after interventions (e.g. BAL), or during haematological reconstitution after fungal pneumonia [31].
Pulmonary haemorrhage might be a focal or diffuse pattern, and the phenomenon of sedimentation within the secondary lobules can sometimes be depicted (Fig. 17).
Intervention
Radiologically guided interventions in neutropenic patients are limited due to the coincidental thrombocytopenia. Therefore, interventions should be performed in patients with at least 50,000 platelet/µl ideally with a running substitution during the biopsy.
Biopsy
There is a large interest in organ specimens for microbiological or pathological investigations. In most cases of fungal pneumonia, BAL fails to detect the fungi; therefore, this frequent differential diagnosis is a special task for percutaneous intervention. There is no literature available that analyses risk and benefit in this population. Actually, neutropenic patients undergo biopsy rarely. On the other hand, the limited risk of radiological interventions in lungs or liver are known to radiologists: Risk for pneumothorax requiring therapy is less than 3% [56], and for bleeding in liver lesions less than 2%. The probability to hit pulmonary nodules under CT guidance is approximately 95% [56]. Using CT fluoroscopy, investigation time and sensitivity are improved especially in small lesions. New non-culture detection tests, on the other hand, are becoming widely available (Galactomannan antigen test, Platelia, Aspergillus--PCR) reducing the necessity to perform invasive diagnostics procedures.
Local drug instillation
As mentioned previously, the response of fungal pneumonia to antifungal drugs is limited. Only when reconstitution of the leukocytes emerges can a substantial response be achieved. Dose escalation has been evaluated, but costs and adverse effects increase without significant improvement. Several groups have evaluated repeated local instillation of amphotericin-B preparation into the fungal pneumonia under CT guidance. They had an improved outcome: 8 lesions completely resolved, 4 greatly improved, and 1 was without change [57]. Thus, local therapy seems to be a promising tool in special cases; however, new antifungal agents are very promising and will become more available in the near future.
Conclusion
Several chest complications occur in patients suffering from neutropenia, after bone-marrow or stem-cell transplantation. Due to the clinical risk, CXR in supine position is not recommended for early detection of pneumonia in these hosts. If pneumonia is suspected, HRCT or thin-section MSCT is suggested to identify the focus of fever or even to exclude pneumonia for some days.
In addition, characterisation of the infiltrate is a relevant topic in thoracic imaging; therefore, close interdisciplinary co-operation as well as careful image interpretation may deliver rapidly a clear number of valid differential diagnoses.
References
Chanock S (1993) Evolving risk factors for infectious complications of cancer. Hematol Oncol Clin North Am 7:771–793
Höffken K (1995) Antibiotische therapie bei neutropenischem fieber. Onkologe 1:503–510
Hiddemann W, Maschmeyer G, Runde V, Einsele H (1996) Prevention, diagnosis and therapy of infections in patients with malignant diseases. Internist 37:1212–1224
Maschmeyer G, Link H, Hiddemann W, Meyer P, Helmerking M, Eisenmann E, Schmitt J, Adam D (1994) Empirical antimicrobial therapy in neutropenic patients: results of a multicenter study by the infections in Hematology Study Group of the Paul Ehrlich Society. Med Klin 89:114–123
Pizzo PA, Robichaud KJ, Gill FA, Witebsky FG (1982) Empirical antibiotic and antifungal therapy for cancer patients with prolonged fever and granulocytopenia. Am J Med 72:101–111
Maschmeyer G, Link H, Hiddemann W, Meyer P, Helmerking M, Eisenmann E, Schmitt J, Adam D (1994) Pulmonary infiltrations in febrile neutropenic patients: risk factors and outcome under empirical antimicrobial therapy in a randomized multicenter trial. Cancer 73:2296–2304
Maschmeyer G (2001) Pneumonia in febrile neutropenic patients: radiologic diagnosis. Curr Opin Oncol 13:229–235
Kolbe K, Domkin D, Derigs HG, Bhakdi S, Huber C, Aulitzky WE (1997) Infectious complications during neutropenia subsequent to peripheral blood stem cell transplantation. Bone Marrow Transpl 19:143–147
Uzun O, Anaissie EJ (1995) Antifungal prophylaxis in patients with hematologic malignancies: a reappraisal. Blood 86:2063–2072
Schaefer-Prokop C, Prokop M, Fleischmann D, Herold C (2001) High-resolution CT of diffuse interstitial lung disease: key findings in common disorders. Eur Radiol 11:373–392
Tanaka N, Matsumoto T, Miura G, Emoto T, Matsunaga N (2002) HRCT findings of chest complications in patients with leukemia. Eur Radiol 12:1512–1522
Einsele H, Bertz H, Beyer J, Kiehl MG, Runde V, Kolb H-J, Holler E, Beck R, Schwertfeger R, Schumacher U, Hebart H, Martin H, Kienast J, Ullmann AJ, Maschmeyer G, Krüger W, Link H, Schmidt CA, Oettle H, Klingebiel T (2001) Epidemiologie und interventionelle therapiestrategien infektiöser komplikationen nach allogener stammzelltransplantation. Dtsch Med Wochenschr 126:1278–1284
Maschmeyer G, Buchheidt D, Einsele H, Heussel CP, Holler E, Lorenz J, Schweigert M (1999) Diagnostik und therapie von lungeninfiltraten bei febrilen neutropenischen patienten: leitlinien der arbeitsgemeinschaft infektionen in der hämatologie und onkologie der deutschen gesellschaft für hämatologie und onkologie. Dtsch Med Wochenschr 9 (Suppl 1):S18–S23 Update: http://www.dgho-infektionen.de/agiho/content/e125/e670/lugeninfiltrate.pdf (26.03.03)
Ascioglu S, Rex JH, de Pauw B, Bennett JE, Bille J, Crokaert F, Denning DW, Donnelly JP, Edwards JE, Erjavec Z, Fiere D, Lortholary O, Maertens J, Meis JF, Patterson TF, Ritter J, Selleslag D, Shah PM, Stevens DA, Walsh TJ (2002) Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: an international consensus. Clin Infect Dis 34:7–14
Edinburgh KJ, Jasmer RM, Huang L, Reddy GP, Chung MH, Thompson A, Halvorsen RA Jr, Webb RA (2000) Multiple pulmonary nodules in AIDS: usefulness of CT in distinguishing among potential causes. Radiology 214:427–432
Heussel CP, Kauczor H-U, Heussel G, Poguntke M, Schadmand-Fischer S, Mildenberger P, Thelen M (1998) Magnetic resonance imaging of liver and brain in hematologic/oncologic patients with fever of unknown origin. Fortschr Roentgenstr 169:128–134
Heussel CP, H-U Kauczor, Heussel G, Derigs HG, Thelen M (2000) Looking for the cause in neutropenic fever: imaging diagnostics. Radiologe 40:88–101
Azoulay E, Darmon M, Delclaux C, Fieux F, Bornstain C, Moreau D, Attalah H, Le Gall Jr, Schlemmer B (2002) Deterioration of previous acute lung injury during neutropenia recovery. Crit Care Med 30:781–786
Navigante AH, Cerchietti LC, Costantini P, Salgado H, Castro MA, Lutteral MA, Cabalar ME (2002) Conventional chest radiography in the initial assessment of adult cancer patients with fever and neutropenia. Cancer Control 9:346–351
Barloon TJ, Galvin JR, Mori M, Stanford W, Gingrich RD (1991) High-resolution ultrafast chest CT in the clinical management of febrile bone marrow transplant patients with normal or nonspecific chest roentgenograms. Chest 99:928–933
Weber C, Maas R, Steiner P, Kramer J, Bumann D, Zander AR, Bucheler E (1999) Importance of digital thoracic radiography in the diagnosis of pulmonary infiltrates in patients with bone marrow transplantation during aplasia. Fortschr Röntgenstr 171:294–301
McLoud TC, Naidich DP (1992) Thoracic disease in the immunocompromised patient. Radiol Clin North Am 30:525–554
Cardillo I, Boal TJ, Einsiedel PF (1997) Patient doses from chest radiography in Victoria Australas. Phys Eng Sci Med 20:92–101
Schöpf UJ, Becker CXR, Obuchowski NA, Rust G-F, Ohnesorge BM, Kohl G, Schaller S, Modic MT, Reiser MF (2001) Multi-slice computed tomography as a screening tool for colon cancer, lung cancer and coronary artery disease. Eur Radiol 11:1975–1985
Heussel CP, Kauczor H-U, Heussel G, Fischer B, Begrich M, Mildenberger P, Thelen M Pneumonia in febrile neutropenic patients, bone-marrow and blood stem-cell recipients: use of high-resolution CT. J Clin Oncol 17:796–805
Remy-Jardin M, Giraud F, Remy J, Copin MC, Gosselin B, Duhamel A (1993) Computed tomography assessment of ground-glass opacity: semiology and significance. J Thorac Imaging 8:249–264
Kauczor HU, Schnütgen M, Fischer B, Schwickert HC, Hartel S, Schadmand-Fischer S, Gerken G, Schweden F (1995) Pulmonary manifestations in HIV patients: the role of chest films, CT and HRCT. Fortschr Roentgenstr 162:282–287
Grenier PA, Beigelman-Aubry C, Fetita C, Preteux F, Brauner MW, Lenoir S (2002) New frontiers in CT imaging of airway disease. Eur Radiol 12:1022–1044
Flohr T, Stierstorfer K, Bruder H, Simon J, Schaller S (2002) New technical developments in multislice CT. Part 1: approaching isotropic resolution with sub-millimeter 16-slice scanning. Fortschr Röntgenstr 174:839–845
Eibel R, Ostermann H, Schiel X (2003) Thorakale Computertomographie von Lungeninfiltraten. http://www.dgho-infektionen.de/agiho/content/e134/e619/e657 (26.03.03)
Heussel CP, Kauczor HU, Heussel G, Mildenberger P, Dueber C (1997) Aneurysms complicating inflammatory diseases in immunocompromised hosts: value of contrast-enhanced CT. Eur Radiol 7:316–319
Conces DJ (1999) Noninfectious lung disease in immunocompromised patients. J Thorac Imaging 14:9–24
Grenier PA, Beigelman-Aubry C, Fetita C, Preteux F, Brauner MW, Lenoir S (2002) New frontiers in CT imaging of airway disease. Eur Radiol 12:1022–1044
Leutner CC, Gieseke J, Lutterbey G, Kuhl CK, Glasmacher A, Wardelmann E, Theisen A, Schild HH (2000) MR imaging of pneumonia in immunocompromised patients: comparison with helical CT. AJR 175:391–397
Leutner C, Schild H (2001) MRI of the lung parenchyma. Fortschr Röntgenstr 173:168–175
Caillot D, Couaillier JF, Bernard A, Casasnovas O, Denning DW, Mannone L, Lopez J, Couillault G, Piard F, Vagner O, Guy H (2001) Increasing volume and changing characteristics of invasive pulmonary aspergillosis on sequential thoracic computed tomography scans in patients with neutropenia. J Clin Oncol 19:253–259
Davies SF (1994) Fungal pneumonia. Med Clin North Am 78:1049–1065
Serra P, Santini C, Venditti M, Mandelli F, Martino P (1985) Superinfections during antimicrobial treatment with betalactam-aminoglycoside combinations in neutropenic patients with hematologic malignancies. Infection 13 (Suppl 1):S115–S122
Tanaka N, Matsumoto T, Miura G, Emoto T, Matsunaga N, Satoh Y, Oka Y (2002) CT findings of leukemic pulmonary infiltration with pathologic correlation. Eur Radiol 12:166–174
Reittner P, Ward S, Heyneman L, Johkoh T, Müller NL (2003) Pneumonia: high-resolution CT findings in 114 patients. Eur Radiol 13:515–521
Conces DJ (1998) Bacterial pneumonia in immunocompromised patients. J Thorac Imaging 13:261–270
Reittner P, Ward S, Heyneman L, Johkoh T, Muller NL (2003) Pneumonia: high-resolution CT findings in 114 patients. Eur Radiol 13:515–521
Heussel CP, Ullmann AJ, Kauczor H-U (2000) Fungal pneumonia. Radiologe 40:518–529
Kim MJ, Lee KS, Kim J, Jung KJ, Lee HG, Kim TS (2001) Crescent sign in invasive pulmonary aspergillosis: frequency and related CT and clinical factors. J Comput Assist Tomogr 25:305–310
Kim K, Lee MH, Kim J, Lee KS, Kim SM, Jung MP, Han J, Sung KW, Kim WS, Jung CW, Yoon SS, Im YH, Kang WK, Park K, Park CH (2002) Importance of open lung biopsy in the diagnosis of invasive pulmonary aspergillosis in patients with hematologic malignancies. Am J Hematol 71:75–79
Stringer JR, Beard CB, Miller RF, Wakefield AE (2002) A new name (Pneumocystis jiroveci) for pneumocystis from humans. Emerg Infect Dis Sep 8 http://www.cdc.gov/ncidod/EID/vol8no9/02–0096.htm (26.03.03)
Hidalgo A, Falcó V, Mauleón S, Andreu J, Crespo M, Ribera E, Pahissa A, Cáceres J (2003) Accuracy of high-resolution CT in distinguishing between Pneumocystis carinii pneumonia and non-Pneumocystis carinii pneumonia in AIDS patients. Eur Radiol 13:1179–1184
McGuinness G, Gruden JF (1999) Viral and Pneumocystis carinii infections of the lung in the immunocompromized host. J Thorac Imaging 14:25–36
van Dyck P, Vanhoenacker FM, van den Brande P, de Schepper AM (2003) Imaging of pulmonary tuberculosis. Eur Radiol 13:1771–1785
Goo JM, Im JG (2002) CT of tuberculosis and nontuberculous mycobacterial infections. Radiol Clin North Am 40:73–87
Leblond V, Zouabi H, Sutton L, Guillon JM, Mayaud CM, Similowski T, Beigelman C, Autran B (1994) Late CD8+ lymphocytic alveolitis after allogeneic bone marrow transplantation and chronic graft-vs-host disease. Am J Respir Crit Care Med 150:1056–1061
Oikonomou A, Hansell DM (2002) Organizing pneumonia: the many morphological faces. Eur Radiol 12:1486–1496
Monson JM, Stark P, Reilly JJ, Sugarbaker DJ, Strauss GM, Swanson SJ, Decamp MM, Mentzer SJ, Baldini EH (1998) Clinical radiation pneumonitis and radiographic changes after thoracic radiation therapy for lung carcinoma. Cancer 82:842–850
Erasmus JJ, McAdams HP, Rossi SE (2002) High-resolution CT of drug-induced lung disease. Radiol Clin North Am 40:61–72
Heyneman LE, Johkoh T, Ward S, Honda O, Yoshida S, Muller NL (2000) Pulmonary leukemic infiltrates: high-resolution CT findings in 10 patients. Am J Roentgenol 174:517–521
Froelich JJ, Wagner HJ (2001) CT fluoroscopy: tool or gimmick? Cardiovasc Intervent Radiol 24:297–305
Veltri A, Anselmetti GC, Bartoli G, Martina MC, Regge D, Galli J, Bertini M (2000) Percutaneus treatment with amphotericin B of mycotic lung lesions from invasive aspergillosis: results in 10 immunocompromised patients. Eur Radiol 10:1939–1944
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Heussel, C.P., Kauczor, HU. & Ullmann, A.J. Pneumonia in neutropenic patients. Eur Radiol 14, 256–271 (2004). https://doi.org/10.1007/s00330-003-1985-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-003-1985-6