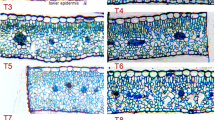
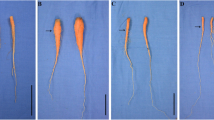
Abstract
Key message
Hypoxia enhances lignification of carrot root.
Abstract
Hypoxia stress was thought to be one of the major abiotic stresses that inhibiting the growth and development of higher plants. The genes encoding the plant alcohol dehydrogenase (ADH-P) were induced when suffering hypoxia. To investigate the impact of hypoxia on the carrot root growth, carrot plants were cultivated in the hydroponics with or without aeration. Morphological characteristics, anatomical structure, lignin content, and the expression profiles of DcADH-P genes and lignin biosynthesis-related genes were measured. Six DcADH-P genes were identified from the carrot genome. The expression profiles of only three (DcADH-P1, DcADH-P2, and DcADH-P3) genes could be detected and the other three (DcADH-P4, DcADH-P5, and DcADH-P6) could not be detected when carrot cultivated in the solution without aeration. In addition, carrot roots had more lignin content, aerenchyma and less fresh weight when cultivated in the solution without aeration. These results suggested that hypoxia could enhance the lignification and affect anatomical structure of the carrot root. However, the expression levels of the genes related to lignin biosynthesis were down-regulated under the hypoxia. The enhancement of lignification may be the consequence of the structure changes in the carrot root. Our work was potentially helpful for studying the effect of hypoxia on carrot growth and may provide useful information for carrot hydroponics.








Similar content being viewed by others
References
Albrecht G, Mustroph A (2003) Localization of sucrose synthase in wheat roots: increased in situ activity of sucrose synthase correlates with cell wall thickening by cellulose deposition under hypoxia. Planta 217:252–260
Alvarez S, Marsh EL, Schroeder SG, Schachtman DP (2008) Metabolomic and proteomic changes in the xylem sap of maize under drought. Plant Cell Environ 31:325–340
Bacanamwo M, Purcell LC (1999) Soybean root morphological and anatomical traits associated with acclimation to flooding. Crop Sci 39:143–149
Bai T, Yin R, Li C, Ma F, Yue Z, Shu H (2011) Comparative analysis of endogenous hormones in leaves and roots of two contrasting Malus species in response to hypoxia stress. J Plant Growth Regul 30:119–127
Baxter-Burrell A, Chang R, Springer P, Bailey-Serres J (2003) Gene and enhancer trap transposable elements reveal oxygen deprivation-regulated genes and their complex patterns of expression in Arabidopsis. Ann Bot 91:129–141
Boerjan W, Ralph J, Baucher M (2003) Lignin biosynthesis. Annu Rev Plant Biol 54:519–546
Bonawitz ND, Chapple C (2010) The genetics of lignin biosynthesis: connecting genotype to phenotype. Annu Rev Genet 44:337–363
Boudet AM (2000) Lignins and lignification: selected issues. Plant Physiol Biochem 38:81–96
Cai C, Xu C, Li X, Ferguson I, Chen K (2006) Accumulation of lignin in relation to change in activities of lignification enzymes in loquat fruit flesh after harvest. Postharvest Biol Technol 40:163–169
Cavagnaro PF, Chung SM, Manin S, Yildiz M, Ali A, Alessandro MS, Iorizzo M, Senalik DA, Simon PW (2011) Microsatellite isolation and marker development in carrot - genomic distribution, linkage mapping, genetic diversity analysis and marker transferability across Apiaceae. BMC Genom 12:386
Cervilla L, Rosales M, Rubio-Wilhelmi M, Sánchez-Rodríguez E, Blasco B, Ríos J, Romero L, Ruiz J (2009) Involvement of lignification and membrane permeability in the tomato root response to boron toxicity. Plant Sci 176:545–552
Chang C, Meyerowitz EM (1986) Molecular cloning and DNA sequence of the Arabidopsis thaliana alcohol dehydrogenase gene. Proc Natl Acad Sci 83:1408–1412
Christianson JA, Llewellyn DJ, Dennis ES, Wilson IW (2009) Global gene expression responses to waterlogging in roots and leaves of cotton (Gossypium hirsutum L.). Plant Cell Physiol 51:21–37
Christianson JA, Llewellyn DJ, Dennis ES, Wilson IW (2010) Comparisons of early transcriptome responses to low-oxygen environments in three dicotyledonous plant species. Plant Signal Behav 5:1006–1009
Costa MA, Bedgar DL, Moinuddin SG, Kim KW, Cardenas CL, Cochrane FC, Shockey JM, Helms GL, Amakura Y, Takahashi H (2005) Characterization in vitro and in vivo of the putative multigene 4-coumarate: CoA ligase network in Arabidopsis: syringyl lignin and sinapate/sinapyl alcohol derivative formation. Phytochemistry 66:2072–2091
Dixon RA, Achnine L, Kota P, Liu CJ, Reddy M, Wang L (2002) The phenylpropanoid pathway and plant defence—a genomics perspective. Mol Plant Pathol 3:371–390
Donaldson LA, Knox JP (2012) Localization of cell wall polysaccharides in normal and compression wood of radiata pine: relationships with lignification and microfibril orientation. Plant Physiol 158:642–653
Drew MC, He CJ, Morgan PW (1989) Decreased ethylene biosynthesis, and induction of aerenchyma, by nitrogen- or phosphate-starvation in adventitious roots of Zea mays L. Plant Physiol 91:266–271
Drew MC, He CJ, Morgan PW (2000) Programmed cell death and aerenchyma formation in roots. Trends Plant Sci 5:123–127
Eddy SR (2011) Accelerated profile HMM searches. PLoS Comput Biol 7:e1002195
Finn RD, Bateman A, Clements J, Coggill P, Eberhardt RY, Eddy SR, Heger A, Hetherington K, Holm L, Mistry J (2013) Pfam: the protein families database. Nucleic Acids Res 42:D222-D230
Finn RD, Clements J, Arndt W, Miller BL, Wheeler TJ, Schreiber F, Bateman A, Eddy SR (2015) HMMER web server: 2015 update. Nucleic Acids Res 43:W30-W38
Fukao T, Bailey-Serres J (2004) Plant responses to hypoxia—is survival a balancing act? Trends Plant Sci 9:449–456
Gabaldón T, Koonin EV (2013) Functional and evolutionary implications of gene orthology. Nat Rev Genet 14:360–366
Geigenberger P (2003) Response of plant metabolism to too little oxygen. Curr Opin Plant Biol 6:247–256
Hickman G (2011) Greenhouse vegetable production statistics: a review of current data on the international production of vegetables in greenhouses. Cuesta Roble greenhouse consultants, Mariposa
Hu WJ, Harding SA, Lung J, Popko JL, Ralph J, Stokke DD, Tsai C-J, Chiang VL (1999) Repression of lignin biosynthesis promotes cellulose accumulation and growth in transgenic trees. Nat Biotechnol 17:808–812
Hu B, Jin J, Guo AY, Zhang H, Luo J, Gao G (2014) GSDS 2.0: an upgraded gene feature visualization server. Bioinformatics 31:1296–1297
Iorizzo M, Ellison S, Senalik D, Zeng P, Satapoomin P, Huang J, Bowman M, Iovene M, Sanseverino W, Cavagnaro P, Yildiz M, Macko-Podgórni A, Moranska E, Grzebelus E, Grzebelus D, Ashrafi H, Zheng Z, Cheng S, Spooner D, Van Deynze A, Simon P (2016) A high-quality carrot genome assembly provides new insights into carotenoid accumulation and asterid genome evolution. Nat Genet 48:657–666
Ismond KP, Dolferus R, De Pauw M, Dennis ES, Good AG (2003) Enhanced low oxygen survival in Arabidopsis through increased metabolic flux in the fermentative pathway. Plant Physiol 132:1292–1302
Jia XL, Wang GL, Xiong F, Yu XR, Xu ZS, Wang F, Xiong AS (2015) De novo assembly, transcriptome characterization, lignin accumulation, and anatomic characteristics: novel insights into lignin biosynthesis during celery leaf development. Sci Rep 5:8259
Jung H, Casler M (2006) Maize stem tissues: impact of development on cell wall degradability. Crop Sci 46:1801
Kao YY, Harding SA, Tsai CJ (2002) Differential expression of two distinct phenylalanine ammonia-lyase genes in condensed tannin-accumulating and lignifying cells of quaking aspen. Plant Physiol 130:796–807
Kawai M, Samarajeewa PK, Barrero RA, Nishiguchi M, Uchimiya H (1998) Cellular dissection of the degradation pattern of cortical cell death during aerenchyma formation of rice roots. Planta 204:277–287
Kimura M, Rodriguez-Amaya DB (2003) Carotenoid composition of hydroponic leafy vegetables. J Agric Food Chem 51:2603–2607
Kürsteiner O, Dupuis I, Kuhlemeier C (2003) The pyruvate decarboxylase1 gene of Arabidopsis is required during anoxia but not other environmental stresses. Plant Physiol 132:968–978
Lalitha S (2000) Primer premier 5. Biotech Softw Internet Rep 1:270–272
Lee D, Ellard M, Wanner LA, Davis KR, Douglas CJ (1995) The Arabidopsis thaliana 4-coumarate: CoA ligase (4CL) gene: stress and developmentally regulated expression and nucleotide sequence of its cDNA. Plant Mol Biol 28:871–884
Morgan PW (1994) Induction of enzymes associated with lysigenous aerenchyma formation in roots of Zea mays during hypoxia or nitrogen starvation. Plant Physiol 105:861–865
Moura JCMS., Bonine CAV, De Oliveira Fernandes Viana J, Dornelas MC, Mazzafera P (2010) Abiotic and biotic stresses and changes in the lignin content and composition in plants. J Integr Plant Biol 52:360–376
Müse G, Schindler T, Bergfeld R, Ruel K, Jacquet G, Lapierre C, Speth V, Schopfer P (1997) Structure and distribution of lignin in primary and secondary cell walls of maize coleoptiles analyzed by chemical and immunological probes. Planta 201:146–159
Narsai R, Rocha M, Geigenberger P, Whelan J, van Dongen JT (2011) Comparative analysis between plant species of transcriptional and metabolic responses to hypoxia. N Phytol 190:472–487
Nobel PS (2005) Physicochemical and environmental plant physiology. Q Rev Biol 54:507–543
Ober ES, Sharp RE (1996) A microsensor for direct measurement of O2 partial pressure within plant tissues3. J Exp Bot 47:447–454
Park J, Yoon JH, Depuydt S, Oh JW, Jo Y, Kim K, Brown MT, Han T (2016) The sensitivity of an hydroponic lettuce root elongation bioassay to metals, phenol and wastewaters. Ecotoxicol Environ Saf 126:147–153
Peng HP, Chan CS, Shih MC, Yang SF (2001) Signaling events in the hypoxic induction of alcohol dehydrogenase gene in Arabidopsis. Plant Physiol 126:742–749
Ratsch H (1983) Interlaboratory root elongation testing of toxic substances on selected plant species. Environ Prot Agency Carvallis Environ Res Lab Coevallis OR EPA 600:3–85
Rozema J, van de Staaij J, Björn LO, Caldwell M (1997) UV-B as an environmental factor in plant life: stress and regulation. Trends Ecol Evol 12:22–28
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3:1101–1108
Schussler EE, Longstreth DJ (1996) Aerenchyma develops by cell lysis in roots and cell separation in leaf petioles in Sagittaria lancifolia (Alismataceae). Am J Bot 83:1266–1273
Shen H, Fu C, Xiao X, Ray T, Tang Y, Wang Z, Chen F (2009) Developmental control of lignification in stems of lowland switchgrass variety Alamo and the effects on saccharification efficiency. BioEnergy Res 2:233–245
Suralta RR, Yamauchi A (2008) Root growth, aerenchyma development, and oxygen transport in rice genotypes subjected to drought and waterlogging. Environ Exp Bot 64:75–82
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Thomashow MF (1999) Plant cold acclimation: freezing tolerance genes and regulatory mechanisms. Annu Rev Plant Biol 50:571–599
Tian C, Jiang Q, Wang F, Wang GL, Xu ZS, Xiong AS (2015) Selection of suitable reference genes for qPCR normalization under abiotic stresses and hormone stimuli in carrot leaves. PLoS One 10:e0117569
Tuberosa R, Sanguineti MC, Landi P, Giuliani MM, Salvi S, Conti S (2002) Identification of QTLs for root characteristics in maize grown in hydroponics and analysis of their overlap with QTLs for grain yield in the field at two water regimes. Plant Mol Biol 48:697–712
Vanholme R, Demedts B, Morreel K, Ralph J, Boerjan W (2010) Lignin biosynthesis and structure. Plant Physiol 153:895–905
Wang GL, Xiong F, Que F, Xu ZS, Wang F, Xiong AS (2015) Morphological characteristics, anatomical structure, and gene expression: novel insights into gibberellin biosynthesis and perception during carrot growth and development. Hortic Res 2:e0134166
Wang GL, Huang Y, Zhang XY, Xu ZS, Wang F, Xiong AS (2016) Transcriptome-based identification of genes revealed differential expression profiles and lignin accumulation during root development in cultivated and wild carrots. Plant Cell Rep 35:1743–1755
Wang F, Wang GL, Hou XL, Li MY, Xu ZS, Xiong AS (2018) The genome sequence of ‘Kurodagosun’, a major carrot variety in Japan and China, reveals insights into biological research and carrot breeding. Mol Genet Genom 2018:1–11
Weng JK, Chapple C (2010) The origin and evolution of lignin biosynthesis. N Phytol 187:273–285
Xu ZS, Tan HW, Wang F, Hou XL, Xiong AS (2014a) CarrotDB: a genomic and transcriptomic database for carrot. Database J Biol Databases Curation 2014:1229–1245
Xu ZS, Ying H, Feng W, Xiong S, Wang GL, Xiong AS (2014b) Transcript profiling of structural genes involved in cyanidin-based anthocyanin biosynthesis between purple and non-purple carrot (Daucus carota L.) cultivars reveals distinct patterns. BMC Plant Biol 14:1–10
Yu JQ, Matsui Y (1993) Extraction and identification of phytotoxic substances accumulated in nutrient solution for the hydroponic culture of tomato. Soil Sci Plant Nutr 39:691–700
Zhao Q, Dixon RA (2011) Transcriptional networks for lignin biosynthesis: more complex than we thought? Trends Plant Sci 16:227–233
Acknowledgements
The research was supported by the New Century Excellent Talents in University (NCET-11-0670); Jiangsu Natural Science Foundation (BK20130027); Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Maike Petersen.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Que, F., Wang, GL., Feng, K. et al. Hypoxia enhances lignification and affects the anatomical structure in hydroponic cultivation of carrot taproot. Plant Cell Rep 37, 1021–1032 (2018). https://doi.org/10.1007/s00299-018-2288-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-018-2288-3