Abstract
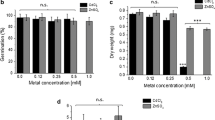
Responses induced by Cd exposure were assessed in tobacco seedlings (Nicotiana tabacum L.) using macro and molecular indices. The 100 μM of Cd exposure reduced the total dry weight and chlorophyll index of the seedlings as much as the genuine Fe-deficiency. Concentration of Fe in the shoots decreased, whereas that in the roots increased by the Cd exposure, especially in the apoplasmic space. It is probable that Cd interferes mainly with the step of Fe-translocation from the roots to shoots and this sets the upper-part of the plant in a state of Fe-deficiency. The Cd exposure coordinately increased the expressions of the exogenous and the endogenous Fe-deficiency responsive genes, HvIDS2 pro ::GUS, NtFRO1 and NtIRT1 in the roots. This is the first data to demonstrate the responses of Cd-inducible Fe-deficiency at a molecular level.








Similar content being viewed by others
Abbreviations
- CaMV:
-
Cauliflower Mosaic Virus
- GUS:
-
β-glucuronidase
- ICP:
-
inductively coupled plasma atomic emission spectrometer
- MU:
-
methylumbelliferone
- 4-MUG:
-
4-methylumbelliferyl-d-glucuronide
References
Bienfait HF, Van den Briel W, Mesland-Mul NT (1985) Free space iron pools in roots. Generation and mobilization. Plant Physiol 78:596–600
Cataldo DA, Garland TR, Wildumg RE (1983) Cadmium uptake kinetics in intact soybean plants. Plant Physiol 73:844–848
Chaney RL (1989) Kinetic of ferric chelate reduction by roots of iron-deficient peanut (Arachis hypogea). Acta Bot Neerl 38:155–163
Chaney RL, White MC, Simon PW (1975) Plant uptake of heavy metals from sewage sludge applied to land. In: Proc. Int Natl Conf Munic Sludge Manage, Rockville, USA, pp 167–178
Christensen TH (1984) Cadmium soil absorption at low concentrations: I. Effect of time, cadmium load, pH and calcium. Water, Air Soil Pollut 21:105–114
Clemens S (2001) Molecular mechanisms of plant metal tolerance and homeostasis. Planta 212:475–486
Cohen CK, Norvell WA, Kochian LV (1997) Induction of the root cell plasma membrane ferric reductase. Plant Physiol 114:1061–1069
Cohen CK, Fox TC, Garvin DF, Kochian LV (1998) The role of iron-deficiency stress responses in stimulating heavy metal transport in plants. Plant Physiol 116:1063–1072
Cohen CK, Garvin DF, Kochian LV (2004) Kinetic properties of a micronutrient transporter from Pisum sativum indicate a primary function in Fe uptake from the soil. Planta 218:784–792
Connolly EL, Fett JP, Guerinot ML (2002) Expression of the IRT1 metal transporter is controlled by metals at the levels of transpcript and protein accumulation. Plant Cell 14:1347–1357
Das P, Samantaray S, Rout GR (1997) Studies on cadmium toxicity in plants: A review. Environ Pollut 98:29–36
Eckhardt U, Mas Marques A, Buckhout TJ (2001) Two iron-regulated cation transporters from tomato complement metal uptake-deficient yeast mutants. Plant Mol Biol 45:437–448
Gallego SM, Benavides MP, Tomaro ML (1996) Effect of heavy metal ion excess on sunflower leaves: evidence for involvement of oxidative stress. Plant Sci 121:151–159
Garty J, Karay Y, Harel J (1992) Effect of low pH, heavy metals and anions an chlorophyll degradation in the lichen Ramalina duriaei (de not) Bagl. Environ Exp Bot 32:229–241
Gothberg A, Greger M, Holm K, Bengtsson BE (2004) Influence of nutrient levels on uptake and effects of mercury, cadmium, and lead in water spinach. J Environ Qual 33:1247–1255
Greger M, Örgren E (1991) Direct and indirect effects of Cd2+ on photosysnthesis in suger beet (Beta Vulgaris). Physiol Plant 83:129–135
Grusak MA (1995) Whole-root iron(III)-reductase activity throughout the life cycle of iron-grown Pisum sativum L. (Fabaceae): Relevance to iron nutrition of developing seeds. Planta 197:111–117
Grusak MA, Kochian LV, Welch RM (1993) Spatial and temporal development of iron(III)-reductase activity through out the life cycle of iron-grown Pisum sativum L. (Fabaceae): Relevance to iron nutrition of developing seeds. Planta 197:111–117
Grusak MA, Pezeshgi S (1996) Shoot-to-root signal transmission regulates root Fe(III) reductase activity in the dgl mutant of pea. Plant Physiol 110:329–334
Haghiri F (1973) Cadmium uptake by plants. J Environ Qual 2:93–96
Hanikenne M, Kramer U, Demoulin V, Baurain D (2005) A comparative inventory of metal transporters in the green alga Chlamydomonas reinhardtii and the red alga Cyanidioschizon merolae. Plant Physiol 137:428–46
Jefferson RA (1987) Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol Biol Rep 5:387–405
John MK (1976) Interrelationship between plant cadmium and uptake of some other elements from culture solutions by oat and lettuce. Environ Pollut 11:85–95
Kahle H (1993) Response of roots of trees to heavy metals. Environ Exp Bot 33:99–119
Kim Y-Y, Yang Y-Y, Lee Y (2002) Pb and Cd uptake in rice roots. Physiol Plant 116:368–372
Kobayashi T, Yoshihara T, Jiang T, Goto F, Nakanishi H, Mori S, Nishizawa NK (2003a) Combined deficiency of iron and other divalent cations mitigates the symptoms of iron deficiency in tobacco plants. Physiol Plant 119:400–408
Kobayashi T, Nakayama Y, Itai RN, Nakanishi H, Yoshihara T, Mori S, Nishizawa NK (2003b) Identification of novel cis-acting elements, IDE1 and IDE2, pf the barley IDS2 gene promoter conferring iron-deficiency-inducible, root-specific expression in heterogeneous tobacco plants. Plant J 36:780–793
Kobayashi T, Suzuki M, Inoue H, Nakanishi IR, Takahashi M, Nakanishi H, Mori S, Nishizawa NK (2005) Expression of iron-acquisition-related genes in iron-deficient rice is co-ordinately induced by partially conserved iron-deficiency-responsive elements. J Exp Bot [e-pub, Mar 21, 2005]
Kochian LV (1991) Mechanisms of micronutrient uptake and translocation in plants. In: Mortvert JJ, Cox FR, Shuman LM, Welch RM (eds) Micronutrients in agriculture, 2nd edn., Soil Science Society of America, Madison, WI, pp 229–296
Korshnova YO, Eide D, Clark WG, Guerinot ML, Pakrasi HB (1999) The IRT1 protein from Arabidopsis thaliana is a metal transporter with a broad substrate range. Plant Mol Biol 40:37–44
Krupa Z, Baszynski T (1995) Some aspects of heavy metals toxicity towards photosynthetic apparatus—direct and indirect effect on light and dark reactions. Acta Physiol Plant 17:177–190
Lee S, Moon JS, Ko TS, Petros D, Goldsbrough PB, Korban SS (2003) Overexpression of Arabidopsis phytochelatin synthase paradoxically leads to hypersensitivity to cadmium stress. Plant Physiol 131:656–63
Liu JG, Liang JS, Li KQ, Zhang ZJ, Yu BY, Lu XL, Yang JC, Zhu QS (2003) Correlations between cadmium and mineral nutrients in absorption and accumulation in various genotypes of rice under cadmium stress. Chemosphere 52:1467–1473
Lombi E, Tearall KL, Howarth JR, Zhao F-J, Hawkesford MJ, McGrath SP (2002) Influence of iron status on cadmium and zinc uptake by different ecotypes of the hyperaccumulator Thlaspi caerulescens. Plant Physiol 128:1359–1367
Marschner H (1995) Mineral Nutrition of Higher Plants, 2nd edn., Academic press, London, UK
Michalska M, Asp H (2001) Influence of lead and cadmium on growth, heavy metal uptake and nutrient concentration of three lettuce cultivars grown in hydroponic culture. Commun Soil Sci Plant Anal 32:571–583
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nakanishi H, Yamaguchi H, Sasakuma T, Nishizawa NK, Mori S (2000) Two dioxygenase genes, Ids3 and IDS2, from Hordeum vulgare are involved in the biosynthesis of mugineic acid family phytosiderophores. Plant Mol Biol 44:199–207
Okumura N, Nishizawa NK, Umehara Y, Ohata T, Nakanishi H, Yamaguchi H, Chino M, Mori S (1994) A dioxygenase gene (IDS2) expressed under Fe deficiency conditions in the roots of Hordeum vulgare. Plant Mol Biol 25:705–719
Olmos E, Martinez-Solano JR, Piqueras A, Hellin E (2003) Early steps in the oxidative burst induced by cadmium in cultured tobacco cells (BY-2 line). J Exp Bot 54:291–301
Perfus-Barbeoch L, Leonhardt N, Vavasseur A, Forestier C (2002) Heavy metal toxicity: cadmium permeates through calcium channels and disturbs the plant water status. Plant J 32:539–548
Römheld V (1987) Existence of two different strategies for the acquisition of iron in higher plants. In: Winelmann G, Van der Helm D, Neilands JB (eds) Iron transports in microbes, plants and animals. VCH Verlag, Weinheim, FRG pp 353–374
Schützendübel A, Polle A (2002) Plant responses to abiotic stresses: heavy metal-induced oxidative stress and protection by mycorrhization. J Exp Bot 53:1351–1365
Siegel SM (1977) The cytotoxic response of `Nicotiana' protoplasts to metal ions: A survey of the chemical elements. Water Air Soil Pollut 8:293–304
Somashekaraiah BV, Padmaja K, Prasad ARK (1992) Phytotoxicity of cadmium ions on germinating seedlings of mung bean (Phaseolus vulgaris): Involvement of lipid peroxidases in chlorophyll degradation. Physiol Plant 85:85–89
Thomine S, Wang R, Ward JM, Crawford NM, Schroeder JI (2000) Cadmium and iron transport by members of a plant metal transporter family in Arabidopsis with homology to Nramp genes. Proc Natl Acad Sci USA 97:4991–4996
Vert G, Grotz N, Dedaldechamp F, Gaymard F, Guerinot ML, Briat JF, Curie C (2002) IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth. Plant Cell 14:1223–1233
Vert GA, Briat JF, Curie C (2003) Dual regulation of the Arabidopsis high-affinity root iron uptake system by local and long-distance signals. Plant Physiol 132:796–804
Wagner GJ, Sutton TG, Yeargan R (1988) Root control of leaf cadmium accumulation in tobacco. Tob Sci 32:88–91
Wallace A, Romney EM, Alexander GV, Soufi SM, Patel PM (1977) Some interactions in plants among cadmium other heavy metals and chelating agents. Agron J 69:18–20
Welch RM (1995) Micronutrient nutrition of plants. Crit Rev Plant Sci 14:49–82
Welch RM, Norvell WA, Schaefer SC, Shaff JE, Kochian LV (1993) Induction of Fe(III) and copper(II) reduction in pea (Pisum sativum) roots by Fe and Cu status: Does the root plasmalemma Fe(III)-reductase perform a general role in regulating cation uptake? Planta 190:555–561
Yoshihara T, Kobayashi T, Goto F, Masuda T, Higuchi K, Nakanishi H, Nishizawa NK, Mori S (2003) Regulation of the Fe-deficiency responsive gene, IDS2 of barley in tobacco, Plant Biotech 20:33–41
Acknowledgements
We thank Prof. N.K. Nishizawa and S. Mori (The University of Tokyo) for providing the HvIDS2 gene promoter. This research was supported in part by a research grant from the Grant-in-aid for Scientific Research from the Japan Society for the Promotion of Science (JSPS) to TY.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P.P. Kumar
Rights and permissions
About this article
Cite this article
Yoshihara, T., Hodoshima, H., Miyano, Y. et al. Cadmium inducible Fe deficiency responses observed from macro and molecular views in tobacco plants. Plant Cell Rep 25, 365–373 (2006). https://doi.org/10.1007/s00299-005-0092-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-005-0092-3