Abstract
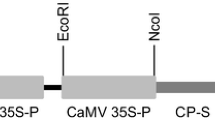
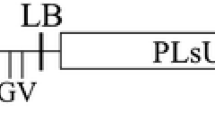
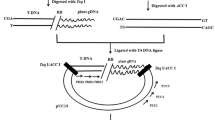
A collection of 127 putatively transgenic individuals of Vitis vinifera cv. Russalka was characterized by PCR and Southern hybridization. Six different constructs containing the neomycin phosphotransferase (nptII) marker gene and sequences of the Grapevine Fanleaf Virus Coat Protein (GFLV CP) gene including non-translatable and truncated forms were transferred via Agrobacterium-mediated transformation. Detection of transgenic sequences by PCR was positive in all lines. Southern blot analysis revealed that the number of inserted T-DNA copies ranged from 1 to 6. More than 46% of the tested transgenic lines contain one copy of the inserted T-DNA, qualifying them as interesting candidates for further breeding programs. Southern data of one line indicate the presence of an incomplete copy of the T-DNA, thus confirming previous PCR results. Since many putative transgenic lines shared identical hybridization patterns, they were clustered into 39 lines and considered as having originated from independent transformation events. The detection of the tetracycline (TET) resistance genes in 15% of the lines shows that an integration of plasmid backbone sequences beyond the T-DNA borders occurred. Enzyme-linked immunosorbent assay (ELISA) performed on leaf tissue did not show any accumulation of the GFLV CP in the 39 transgenic lines analyzed. Reverse transcription polymerase chain reaction (RT-PCR) and Northern blot were carried out; RT-PCR analyses showed that the GFLV CP mRNA was expressed at variable levels.




Similar content being viewed by others
References
Aaziz R, Tepfer M (1999) Recombination in RNA viruses and in virus-resistant transgenic plants. J Gen Virol 80:1339–1346
Ainley WM, Kumpatla SP (2004) Gene silencing in plant. In: Parekh SR (ed) The GMO handbook. Humana Press, Totowa, New Jersey, pp 243–262
An G, Ebert PR, Mitra A, Ha SB (1988) Binary vectors. In: Gelvin SB, Schilperoort RA (eds) Plant molecular biology manual. Kluwer, Dortrecht, pp A3/1–13
Andret-Link P, Schmitt-Kechinger C, Demangeat G, Komar V, Fuchs M (2004a) The specific transmission of Grapevine fanleaf virus by its nematode vector Xiphinema index is solely determined by the viral coat protein. Virology 320:12–22
Andret-Link P, Laporte C, Valat L, Laval V, Ritzenthaler C, Demangeat G,Vigne E, Pfeiffer P, Stussi-Garaud C, Fuchs M (2004b) Grapevine fanleaf virus: Still a major threat to the grapevine industry. J Plant Pathol 86:183–195
Baulcombe D (2004) RNA silencing in plants. Nature 431:356--363
Bertioli DJ, Harris DR, Edwards ML, Cooper JI, Hawes WS (1991) Transgenic plants and insect cells expressing the coat protein of Arabis mosaic virus produce empty virus-like particles. J Gen Virol 72:1801–1809
Bovey R, Gärtel W, Hewitt WB, Martelli GP, Vuittnez A (1980) Maladies à virus et affections similaires de la vigne. Atlas en couleur des symptômes. Eds Payot, Lausanne
Colova-Tsolova V, Perl A, Krastanova S, Tsvetkov I, Atanassov A (2001) Genetically engineered grape for disease and stress tolerance. In: Roubelakis-Angelakis K (ed) Molecular biology and biotechnology of grapevine, Kluwer, Dortrecht, pp 411–432
Dean JD, Goodwin PH, Hsiang T (2002) Comparison of relative RT-PCR and Northern blot analyses to measure expression of β-1,3-glucanase in Nicotiana benthamiana infected with Colltotrichum destructivum. Plant Mol Biol Rep 20:347–356
EC (2001) Directive 2001/18/EC of the European Parliament and of the Council of 12 March 2001 on the deliberate release into the environment and of genetically modified organisms and repealing Council Directive 90/220/EEC. Official Journal of the European Communities, L106-1-39. http://europa.eu.int/eur-lex/pri/dat/2001/1_106/1_10620010417en 00010038.pdf
Finnegan EJ, Wang MB, Waterhouse PM (2001) Gene silencing: Fleshing out the bones. Curr Biol 11:99–102
Flavell RB (1994) Inactivation of gene expression in plants as a consequence of specific sequence duplication. Proc Natl Acad Sci USA 91:3490–3496
Gambino G, Gribaudo I, Leopold S, Schartl A, Laimer M (2005) Molecular characterization of grapevine plants transformed with GFLV resistance genes: I. Plant Cell Rep 24:655--662
Gölles R (1994) Expression verschiedener Varianten des Hüllproteingens von Grapevine Fanleaf Virus in transgenen Pflanzen. Diploma Thesis, University of Agriculture, Vienna
Gölles R, da Câmara Machado A, Tsolova V, Bouquet A, Moser R, Katinger H, Laimer da Câmara Machado M (1998) Transformation of somatic embryos of Vitis sp. with different constructs containing nucleotide sequences from nepovirus coat protein genes. Acta Hort 447:265–270
Gölles R, da Câmara Machado A, Minafra A, Savino V, Saldarelli G, Martelli GP, Pühringer H, Katinger H, Laimer da Câmara Machado M (2000) Transgenic grapevines expressing coat protein gene sequences of grapevine fanleaf virus, arabis mosaic virus, grapevine virus A and grapevine virus B. Acta Hort 528:305–311
Krastanova S, Perrin M, Barbier P, Demangeat G, Cornuet P, Bardonnet N, Otten L, Pinck L, Walter B (1995) Transformation of grapevine rootstocks with the coat protein gene of grapevine fanleaf nepovirus. Plant Cell Rep 14:50–554
Le Gall O, Torregrosa L, Danglot Y, Candresse T, Bouquet A (1994) Agrobacterium-mediated genetic transformation of grapevine somatic embryos and regeneration of transgenic plants expressing the coat protein of grapevine chrome mosaic nepovirus (GCMV). Plant Sci 102:161–170
Lomonossoff GP (1995) Pathogen-derived resistance to plant viruses. Ann Rev Phytopathol 33:323–343
Mauro MC, Toutain S, Walter B, Pinck L, Otten L, Coutos-Thevenot P, Deloire A, Barbier P (1995) High efficiency regeneration of grapevine plants transformed with the GFLV coat protein gene. Plant Sci 112:97–106
Mayo MA, Robinson DJ (1996) Nepoviruses: molecular biology and replication. In: Harrison BD, Murant AF (eds) The plant viruses, Plenum, New York, pp 139–185
Minafra A, Gölles R, da Câmara Machado A, Saldarelli P, Savino V, Katinger H, Laimer da Câmara Machado M, Martelli GP (1997) Coat protein-mediated resistance against grapevine virus A and grapevine virus B in Nicotiana benthamiana and Nicotiana occidentalis. Extended abstracts 12th Meeting ICVG, Lisbon, Portugal, 29 September–2 October 1997, p 140
Ohba T, Yoshioka Y, Machida C, Machida Y (1995) DNA rearrangement associated with the integration of T-DNA in tobacco: an example for multiple duplications of DNA around the integration target. Plant J 7:157–164
Pinck L, Ritzenthaler C, Gaire F, Margis R, Bardonnet N, Walter B (1997) Protection against grapevine fanleaf nepovirus in transgenic tobacco expressing the Vpg-proteinase or the viral replicase gene. Extended abstracts 12th Meeting ICVG, Lisbon, Portugal, 29 September–2 October 1997, p 135
Quacquarelli A, Gallitelli D, Savino V, Martelli GP (1976) Properties of grapevine fannleaf virus. J Gen Virol 32:349
Radian-Sade S, Edelbaum O, Rubinstein Y, Gafni R, Sela I, Tanne E (1997) Transgenic Nicotiana benthamiana plants resistant to grapevine virus A. Extended abstracts 12th Meeting ICVG, Lisbon, Portugal, 29 September–2 October 1997, p 141
Robinson DJ (1996) Environmental risk assessment of releases of transgenic plants containing virus-derived inserts. Transgenic Res 5:359–562
Serghini MA, Fuchs M, Pinck M, Reinbolt J, Walter B, Pinck L (1990) RNA 2 of grapevine fanleaf virus: sequence analysis and coat protein cistron location. J Gen Viro l71:1433–1441
Spielmann A, Krastanova S, Douet-Orhant V, Gugerli P (2000a) Analysis of transgenic grapevine (Vitis rupestris) and Nicotiana benthamiana plants expressing an Arabis mosaic virus coat protein gene. Plant Sci 156:235–244
Spielmann A, Douet-Orhant V, Krastanova S, Gugerli P (2000b) Resistance to Nepoviruses in grapevine and Nicotiana benthamiana: expression of several putative resistance genes in transgenic plants. Acta Hort 528:373–378
Steinkellner H, Weinhäusl A, Laimer M, da Câmara Machado A, Katinger H (1991) Identification of the coat protein gene of arabis mosaic nepovirus and its expression in transgenic plants. Acta Hort 308:37–41
Tepfer M (1993) Viral genes and transgenic plants: what are the potential environmental risks? Biotechnology 11:1125–1132
Tepfer M (2002) Risk assessment of virus resistant transgenic plants. Annu Rev Phytopathol 40:467--491
Tsolova V, Antanassov A (1994) Induction of polyembryony and secondary embryogenesis in culture for embryo rescue of stenospermocarpic genotypes of Vitis vinifera L. Vitis 33:55–56
Vaucheret H, Beclin C, Elmayan T, Feuerbach F, Godon C, Morel JB, Mourrain P, Palauqui JC, Vernhettes S (1998) Transgene-induced gene silencing in plants. Plant J 16:651–659
Wellink J, Le Gall O, Sanfacon H, Ikegami M, Jones AT (2000) Family Comoviridae. In: Van Regenmortel MH, Fauquet CM, Bishop DHL, Carstens EB, Estes MK, Lemon SM, Maniloff J, Mayo MA, McGeoch DJ, Wickner RB (eds) Virus taxonomy: classification and nomenclature of viruses. 7th Report of the international committee on taxonomy of viruses. Academic Press, San Diego, pp 691–701
Xue B, Ling K-S, Reid CL, Krastanova S, Sekiya M, Momol EA, Süle S, Mozsar J, Gonsalves D, Burr TJ (1999) Transformation of five rootstocks with plant virus genes and a virE2 gene from Agrobacterium tumefaciens. In vitro Cell Dev Biol Plant 35:226–231
Acknowledgments
The authors express their gratitude to Richard Gölles; and John Hammond for primers JH006 and JH020. The financial support by the BMLFUW is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Lörz
Rights and permissions
About this article
Cite this article
Maghuly, F., Leopold, S., da Câmara Machado, A. et al. Molecular characterization of grapevine plants transformed with GFLV resistance genes: II. Plant Cell Rep 25, 546–553 (2006). https://doi.org/10.1007/s00299-005-0087-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-005-0087-0